Abstract
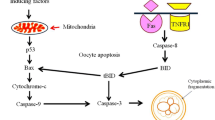
This study was designed to determine follicular atresia in the newborn and the prepubertal spiny mouse. We analyzed the processes of follicle loss using classical markers of apoptosis (TUNEL reaction, active caspase-3) and autophagy (Lamp1). Numerous small clear vacuoles and autophagosomes as well as strong Lamp1 staining were observed in dying oocytes of all follicle types, especially of the primordial and primary ones. Active caspase 3 and the TUNEL reaction were detected only in the granulosa cells of large secondary and antral follicles. The expression of apoptosis and autophagy markers was also changing during the prepubertal period. Western blot analysis indicated that at the moment of birth, females undergo an increased rate of follicular atresia mediated by autophagy, while apoptosis is the dominant form of ovarian atresia in consecutive postnatal days. On the basis of these observations, we concluded that apoptosis and autophagy are involved in follicular atresia and these processes are cell and developmental stage-specific.




Similar content being viewed by others
References
Oosterius WP, Mooren PG, Charles R, Lamers WH (1984) Perinatal development of the lung in rat and spiny mouse: its relation to altricial and precocial timing of birth. Biol Neonate 45:236–243
Hułas M, Gawron A, Orfin G (2003) A comparative study of ovary development in the precocial spiny mouse (Acomys cahirinus) and the altricial Norway rat (Rattus Norvegicus). Isr J Zool 49:307–313
Lamers WH, Mooren PG, De Graaf A, Charles R (1985) Perinatal development of the liver in rat and spiny mouse. Its relation to altricial and precocial timing of birth. Eur J Biochem 146:475–480
Dickinson H, Walker DW, Cullen-McEwen L, Wintour EM, Mortiz K (2005) The spiny mouse (Acomys cahirinus) completes nephrogenesis before birth. Am J Physiol Renal Physiol 289:F273–F279
Lamers WH, Mooren PG, Charles R (1985) Perinatal development of the small intestine and pancreas in rat and spiny mouse. Its relation to altricial and precocial timing of birth. Biol Neonate 47:153–162
Ghafari F, Gutierrez CG, Hartshorne GM (2007) Apoptosis in mouse fetal and neonatal oocytes during meiotic prophase one. BMC Dev Biol 7:87
Lobascio AM, Klinger FG, Scaldaferri ML, Farini D, De Felici M (2007) Analysis of programmed cell death in mouse fetal oocytes. Reproduction 134:241–252
Hartshorne GM, Lyrakou S, Hamoda H, Oloto E, Ghafari F (2009) Oogenesis and cell death in human prenatal ovaries: what are the criteria for oocyte selection. Mol Hum Reprod 15:805–819
Escobar ML, Echeverria OM, Ortiz R, Vasquez-Nin GH (2008) Combined apoptosis and autophagy, the process that eliminates the oocytes of atretic follicles in immature rats. Apoptosis 13:1253–1266
Tingen C, Kim A, Woodruff TK (2009) The primordial pool of follicle and nest breakdown in mammalian ovaries. Mol Hum Reprod 15:795–803
Takeo Y, Hokono M (1995) An electron microscopic study of apoptosis in the granulosa layer of ovarian follicles in rats treated with continuous illumination. Med Electron Microsc 28:38–44
Devine PJ, Payne CM, McCuskey MK, Hoyer PB (2000) Ultrastructural evaluation of oocytes during atresia in rat ovarian follicles. Biol Reprod 63:1245–1952
Irving-Rodgers HF, van Wezel IL, Mussard ML, Kinder JE, Rodgers RJ (2001) Atresia revisited: two basic patterns of atresia of bovine antral follicles. Reproduction 122:761–775
Hurst PR, Mora JM, Fenwick MA (2006) Caspase-3, TUNEL and ultrastructural studies of small follicles in adult human ovarian biopsis. Hum Reprod 21:1974–1980
Tingen CM, Bristol-Gould SK, Kiesewetter SE, Wellington JT, Shea L, Woodruff TK (2009) Prepubertal primordial follicle loss in mice is not due to classical apoptotic pathways. Biol Reprod 81:16–25
Swanson MS (2006) Autophagy: eating for good health. J Immunol 177:4945–4951
Oberstein A, Jeffry PD, Shi Y (2007) Crystal structure of the Bcl-XL-Beclin 1 peptide complex: beclin 1 is a novel BH3-only protein. J Biol Chem 27:13123–13132
Fukuda M (1991) Lysosomal membrane glycoproteins. Structure, biosynthesis and intracellular trafficking. J Biol Chem 266:21327
Bradford MM (1976) A rapid and sensitive method for quantification of microgram quantities of protein utilizing the principle of protein-die binding. Anal Biochem 72:248–254
Glamoclija V, Vilovic K, Saraga-Babic M, Baranovic A, Sapunar D (2005) Apoptosis and active caspase-3 expression in human granulosa cells. Fertil Steril 83:426–431
Mclaughlin EA, Mclver SC (2009) Awakening the oocyte: controlling primordial follicle development. Reproduction 137:1–11
Kezele P, Skinner MK (2003) Regulation of ovarian primordial follicle assembly and development by estrogen and progesterone: endocrine model of follicle assembly. Endocrinology 144:3329–3337
Hułas-Stasiak M, Gawron A (2007) Immunohistochemical localization of estrogen receptors ERα and ERβ in the spiny mouse (Acomys cahirinus) ovary during postnatal development. J Mol Histol 38:25–32
Hułas-Stasiak M, Gawron A (2010) Distribution of androgen and progesterone receptors in the spiny mouse (Acomys cahirinus) ovary during postnatal life. Reprod Biol 10:37–51
Rodrigues P, Limback D, McGinnis LK, Plancha CE, Albertini DF (2009) Multiple mechanisms of germ cell loss in the perinatal mouse ovary. Reproduction 137:709–720
Ortiz R, Echeverria OM, Salgado R, Escobar ML, Vasquez-Nin GH (2006) Fine structural and cytochemical analysis of the process of cell death of oocytes in atretic follicles in newborn and prepubertal rats. Apoptosis 11:25–37
Gonzales-Polo R, Boya P, Pauleau A, Jalil P, Larochette N, Souquere S, Eskelinen E, Pierron G, Saftig P, Kroemer G (2005) The apoptosis/autophagy paradox: autophagic vacuolization before apoptotic death. J Cell Sci 118:3091–3102
Pattingre S, Tassa A, Qu X, Garugi R, Liang XH, Mizushima N, Packer M, Schneider MD, Levine B (2005) Bcl-2 antiapoptotic proteins inhibit Beclin 1-dependent autophagy. Cell 122:927–939
Inoue S, Watanabe H, Saito H, Hiroi M, Tonosaki A (2000) Elimination of atretic follicles from the mouse ovary: a TEM and immunohistochemical study in mice. J Anat 196:103–110
Szołtys M, Słomczyńska M, Wojtowicz A, Pierścińska A (1999) Subpopulations of the granulosa layer differentiated by histo- and immunocystochemical methods in the preovulatory follicle of the rat. Folia Histochem Cytobiol 37:91–92
Orisaka M, Tajima K, Tsang BK, Kotsuji F (2009) Oocyte-granulosa-theca cell interactions during preantral follicular development. J Ovarian Res 2:9
Takagi K, Yamanda T, Miki Y, Umegaki T, Nishimura M, Sasaki J (2007) Histological observation of the development of follicles and follicular atresia in immature rat ovaries. Acta Med Okayama 61:283–298
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hułas-Stasiak, M., Gawron, A. Follicular atresia in the prepubertal spiny mouse (Acomys cahirinus) ovary. Apoptosis 16, 967–975 (2011). https://doi.org/10.1007/s10495-011-0626-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10495-011-0626-9