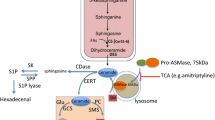
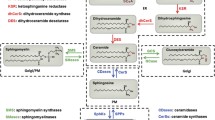
Abstract
Sphingolipids (SLs) have been considered for many years as predominant building blocks of biological membranes with key structural functions and little relevance in cellular signaling. However, this view has changed dramatically in recent years with the recognition that certain SLs such as ceramide, sphingosine 1-phosphate and gangliosides, participate actively in signal transduction pathways, regulating many different cell functions such as proliferation, differentiation, adhesion and cell death. In particular, ceramide has attracted considerable attention in cell biology and biophysics due to its key role in the modulation of membrane physical properties, signaling and cell death regulation. This latter function is largely exerted by the ability of ceramide to activate the major pathways governing cell death such as the endoplasmic reticulum and mitochondria. Overall, the evidence so far indicates a key function of SLs in disease pathogenesis and hence their regulation may be of potential therapeutic relevance in different pathologies including liver diseases, neurodegeneration and cancer biology and therapy.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Kolesnick R (2002) The therapeutic potential of modulating the ceramide/sphingomyelin pathway. J Clin Invest 110:3–8
Ogretmen B, Hannun YA (2004) Biologically active sphingolipids in cancer pathogenesis and treatment. Nat Rev Cancer 4:604–616
Hofmann K, Dixit VM (1998) Ceramide in apoptosis–does it really matter? Trends Biochem Sci 23:374–377
Kolesnick R, Hannun YA (1999) Ceramide and apoptosis. Trends Biochem Sci 24:224–227
Grullich C, Sullards MC, Fukz Z, Merrill AH Jr, Kolesnick R (2000) CD95(Fas/APO-1) signals ceramide generation independent of the effector stage of apoptosis. J Biol Chem 275:8650–8656
Kolesnick R, Kronke M (1998) Regulation of ceramide production and apoptosis. Annu Rev Physiol 60:643–665
Marchesini N, Hannun YA (2004) Acid and neutral sphingomyelinases: roles and mechanisms of regulation. Biochem Cell Biol 82:27–44
Morales A, Colell A, Mari M, Garcia-Ruiz C, Fernandez-Checa JC (2004) Glycosphingolipids and mitochondria: role in apoptosis and disease. Glyconjug J 20:579–588
Merrill AH (2002) De novo sphingolipid biosynthesis: a necessary, but dangerous, pathway. J Biol Chem 277:25843–25846
Pewzner-Jung Y, Ben-Dor S, Futerman AH (2006) When do Lasses (longevity assurance genes) become CerS (ceramide synthases)? Insights into the regulation of ceramide synthesis. J Biol Chem 281:25001–25005
Futerman AH, Rietzman H (2005) The ins and outs of sphingolipid synthesis. Trends Cell Biol 15:312–318
Lynch DV, Dunn TM (2004) An introduction to plant sphingolipids and a review of recent advances in understanding their metabolism and function. New Phytologist 161:677–702
Butron A, Santiago R, Mansilla P, Pintos-Varela C, Ordas A, Malvar RA (2006) Maize (Zea mays L.) genetic factors for preventing fumonisin contamination. J Agric Food Chem 54:6113–6117
Stockmann-Juvala H, Naarala J, Loikkanen J, Vahakangas K, Savolainen K (2006) Fumonisin B1-induced apoptosis in neuroblastoma, glioblastoma and hypothalamic cell lines. Toxicology 225:234–241
Clarke CJ, Snook CF, Tani M, Matmati N, Marchesini N, Hannun YA (2006) The extended family of neutral sphingomyelinases. Biochemistry 45:11247–11256
Tomiuk S, Hofmann K, Nix M, Zumbansen M, Stoffel W (1998) Cloned mammalian neutral sphingomyelinase: functions in sphingolipid signaling?. Proc Natl Acad Sci USA 95:3638–3643
Sawai H, Domae N, Nagan N, Hannun YA (1999) Function of the cloned putative neutral sphingomyelinase as lyso-platelet activating factor-phospholipase C. J Biol Chem 274: 38131–38139
Marchesini N, Luberto C, Hannun YA (2003) Biochemical properties of mammalian neutral sphingomyelinase 2 and its role in sphingolipid metabolism. J Biol Chem 278:13775–13783
Krut O, Wiegmann K, Kashkar H, Yazdanpanah B, Kronke M (2006) Novel tumor necrosis factor-responsive mammalian neutral sphingomyelinase-3 is a C-tail-anchored protein. J Biol Chem 281:13784–13793
Schissel SL, Jiang X, Tweedie-Hardman J et al (1998) Secretory sphingomyelinase, a product of the acid sphingomyelinase gene, can hydrolyze atherogenic lipoproteins at neutral pH. Implications for atherosclerotic lesion development. J Biol Chem 273:2738–2746
Liu P, Anderson RG (1995) Compartmentalized production of ceramide at the cell surface. J Biol Chem 270:27179–27185
Grassme H, Cremesti A, Kolesnick R, Gulbins E (2003) Ceramide-mediated clustering is required for CD95-DISC formation. Oncogene 22:5457–5470
Garcia-Ruiz C, Colell A, Mari M et al (2003) Defective TNF-alpha-mediated hepatocellular apoptosis and liver damage in acidic sphingomyelinase knockout mice. J Clin Invest 111:197–208
Llacuna L, Mari M, Garcia-Ruiz C, Fernandez-Checa JC, Morales A (2006) Critical role of acidic sphingomyelinase in murine hepatic ischemia-reperfusion injury. Hepatology 44:561–572
Garcia-Ruiz C, Mari M, Morales A, Colell A, Ardite E, Fernandez-Checa JC (2000) Human placenta sphingomyelinase, an exogenous acidic pH-optimum sphingomyelinase, induces oxidative stress, glutathione depletion, and apoptosis in rat hepatocytes. Hepatology 32:56–65
Mari M, Colell A, Morales A et al (2004) Acidic sphingomyelinase downregulates the liver-specific methionine adenosyltransferase 1A, contributing to tumor necrosis factor-induced lethal hepatitis. J Clin Invest 113:895–904
Valaperta R, Chigomo V, Basso L et al (2006) Plasma membrane production of ceramide from ganglioside GM3 in human fibroblasts. FASEB J 20:1227–1229
El Bawab S, Roddy P, Qian T, Bielawska A, Lemasterss JJ, Hannun YA (2000) Molecular cloning and characterization of a human mitochondrial ceramidase. J Biol Chem 275:21508–21513
Bionda C, Portoukalian J, Schmitt D, Rodriguez-Lafrasse C, Ardail D (2004) Subcellular compartmentalization of ceramide metabolism: MAM (mitochondria-associated membrane) and/or mitochondria? Biochem J 382:527–533
Taha TA, Hannun YA, Obeid LM (2006) Sphingosine kinase: biochemical and cellular regulation and role in disease. J Biochem Mol Biol 39:113–131
Allende ML, Sasaki T, Kawai H et al (2004) Mice deficient in sphingosine kinase 1 are rendered lymphopenic by FTY720. J Biol Chem 279:52487–52492
Dressler KA, Kolesnick RN (1990) Ceramide-1-phosphate, a novel phospholipid in human leukemia (HL-60) cells. J Biol Chem 265:14917–14921
Gómez-Muñoz A (2006) Ceramide-1-phosphate/ceramde: a switch between life and death. Biochim Biophys Acta in press
Bajjalieh SM, Martin TFJ, Floor E (1989) Synaptic vesicle ceramide kinase. A calcium-stimulated lipid kinase that copurifies with brain synaptic vesicles. J Biol Chem 264:14354–14360
Sugiura M, Kono K, Liu H et al (2002) Ceramide kinase, a novel lipid kinase. J Biol Chem 277:23294–23300
Perry RJ, Ridgway ND (2005) Molecular mechanisms and regulation of ceramide transport. Biochim Biophys Acta 1734:220–234
Hanada K, Kumagai K, Yasuda S et al (2003) Molecular machinery for non-vesicular trafficking of ceramide. Nature 426:803–809
Kumagai K, Yasuda S, Okemoto K, Nishijima M, Kobayashi S, Hanaka K (2005) CERT mediates intermembrane transfer of various molecular species of ceramides. J Biol Chem 280: 6488–6495
Perry RJ, Ridway ND (2006) Oxysterol-binding protein and vesicle-associated membrane protein-associated protein are required for sterol-dependent activation of the ceramide transport protein. Mol Biol Cell 17:2604–2616
Giussani P, Maceyka M, Le Stunff M et al (2006) Sphingosine-1-phosphate phosphohydrolase regulates endoplasmic reticulum-to-Golgi trafficking of ceramide. Mol Cell Biol 26:5055–5069
Basu S, Kaufman B, Roseman S (1973) Enzymatic synthesis of glucocerebroside by a glucosyltransferase from embryonic chicken brain. J Biol Chem 248:1388–1394
Basu S, Schutz AM, Basu M, Roseman S (1971) Enzymatic synthesis of galactosylcerebroside by a galactosyltransferase from endoplasmic chicken brain. J Biol Chem 246:4272–4279
Yamashita T, Wada R, Sasaki T et al (1999) A vital role for glycosphingolipid synthesis during development and differentiation. Proc Natl Acad Sci USA 96:9142–9147
Basu S (1991) The serendipity of ganglioside biosynthesis: pathway to CARS and HY-CARS glycosyltransferases. Glycobiology 1:469–475
Lovat PE, Corazzari M, DiSano F, Piacentini M, Redfern CP (2005) The role of gangliosides in fenretinide-induced apoptosis of neuroblastoma. Cancer Lett 228:105–110
Hail N, Kim HJ, Lotan R (2006) Mechanisms of fenretinide-induced apoptosis. Apoptosis 11:1677–1694
Blitterswijk WJ, Van Der Luit A, Veldman R, Verheij M, Borst J (2003) Ceramide: second messenguer or modulator of membrane structure and dynamics? Biochem J 369:199–211
Goñi FM, Alonso A (2006) Biophysics of sphingolipids I. Membrane properties of sphingosine, ceramides and other simple sphingolipids. Biochim Biophys Acta in press
Koiv A, Mustonen P, Kinnunen PKJ (1993) Influence of sphingosine on the thermal phase behaviour of neutral and acidic phospholipid liposomes. Chem Phys Lipids 66:123–134
Kinnunen PKJ, Rytomaa M, Koiv A, Lehtonen J, Mustonen P, Aro A (1993) Sphingosine-mediated membrane association of DNA and its reversal by phosphatidic-acid. Chem Phys Lipids 66:75–88
Siskind LJ, Fluss S, Bui M, Colombini M (2005) Sphingosine forms channels in membranes that differ greatly from those formed by ceramide. J Bioenerg Biomemb 37:227–236
Contreras FX, Sot J, Alonso A, Goñi FM (2006) Sphingosine increases the permeability of model and cell membranes. Biophys J 90:4085–4092
Holopainen JM, Lehtonen JY, Kinnunen PKJ (1997) Lipid microdomains in dimyristoylphosphatidylcholine-ceramide liposomes. Chem Phys Lipids 88:1–13
Holopainen JM, Subramanian M, Kinnunen PKJ (1998) Sphingomyelinase induces lipid microdomain formation in a fluid phosphatidylcholine/sphingomyelin membrane. Biochemistry 37:17562–17570
Huang HW, Goldberg EM, Zidovetzki Z (1996) Ceramides induces structural defects into phosphatidylcholine bilayers and activates phospholipase A2. Biochem Biophys Res Commun 220:834–838
Veiga MP, Arrondo JLR, Goñi FM, Alonso A (1999) Ceramides in Phospholipid Membranes: Effects on Bilayer Stability and Transition to Nonlamellar Phases. Biophys J 76:342–350
Fanani ML, Hartel S, Oliveira RG, Maggio B (2002) Bidirectional control of sphingomyelinase activity and surface topography in lipid monolayers. Biophys J 83:3416–3424
Härtel S, Fanani ML, Maggio B (2005) Shape transitions and lattice structuring of ceramide-enriched domains generated by sphingomyelinase in lipid monolayers. Biophys J 88:287–304
Sot J, Bagatoll LA, Goñi FM, Alonso A (2006) Detergent-resistant, ceramide-enriched domains in sphingomyelin/ceramide bilayers. Biophys J 90:903–914
Bai JN, Pagano (1997) Measurement of spontaneous transfer and transbilayer movement of BODIPY-labeled lipids in lipid vesicles. Biochemistry 36:8840–8848
Lopez-Montero I, Rodriguez N, Cribier S, Pohl A, Velez L, Devaux PF (2005) Rapid transbilayer movement of ceramides in phospholipid vesicles and in human erythrocytes. J Biol Chem 280:25811–25819
Contreras FX, Basañez G, Alonso A, Herrmann A, Goñi FM (2005) Asymmetric addition of ceramides but not dihydroceramides promotes transbilayer (flip-flop) lipid motion in membranes. Biophys J 88:348–359
Contreras FX, Villar AV, Alonso A, Kolesnick RN, Goñi FM (2003) Sphingomyelinase activity causes transbilayer lipid translocation in model and cell membranes. J Biol Chem 278:37169–37174
Pascher I (1976) Molecular arrangements in sphingolipids. Conformation and hydrogen bonding of ceramide and their implication on membrane stability and permeability. Biochim Biophys Acta 455:433–451
Ruiz-Arguello MB, Basañez G, Goñi FM, Alonso A (1996) Different effects of enzyme-generated ceramides and diacylglycerols in phospholipid membrane fusion and leakage. J Biol Chem 271:26616–26621
Montes LR, Ruiz-Arguello MB, Goñi FM, Alonso A (2002) Membrane restructuring via ceramide results in enhanced solute efflux. J Biol Chem 277:11788–11794
Siskind LJ, Colombini M (2000) The lipids C2- and C16-ceramide form large stable channels. Implications for apoptosis. J Biol Chem 275:38640–38644
Siskind LJ, Kolesnick R, Colombini M (2006) Ceramide forms channels in mitochondrial outer membranes at physiologically relevant concentrations. Mitochondrion 6:118–125
Hannun YA, Obeid LM (2002) The Ceramide-centric universe of lipid-mediated cell regulation: stress encounters of the lipid kind. J Biol Chem 277:25847–25850
Lozano J, Berra E, Municio MM et al (1994) Protein kinase C ζ isoform is critical for κB-dependent promoter activation by sphingomyelinase. J Biol Chem 269:19200–19202
Muller G, Ayoub M, Storz P, Rennecke J, Fabbro D, Pfizenmaier K (1995) PKCz is a molecular switch in signal transduction of TNF-α, bifunctionally regulated by ceramide and arachidonic acid. EMBO J 14:1961–1969
Zhang Y, Yao B, Delikat S et al (1997) Kinase suppressor of Ras is ceramide-activated protein kinase. Cell 89:63–72
Huwiler A, Brunner J, Hummel R et al (1996) Ceramide-binding and activation defines protein kinase c-Raf as a ceramide-activated protein kinase. Proc Natl Acad Sci USA 93:6959–6963
Dobrowsky RT, Kamibayashi C, Mumby MC, Hannun YA (1993) Ceramide activates heterotrimeric protein phosphatase 2A. J Biol Chem 268:15523–15530
Ruvolo PP, Clark W, Mumby M, Gao F, May WS (2002) A functional role for the B56 alpha-subunit of protein phosphatase 2A in ceramide-mediated regulation of Bcl2 phosphorylation status and function. J Biol Chem 277: 22847–22852
Verheij M, Bose R, Lin XH et al (1996) Requirement for ceramide-initiated SAPK/JNK signaling in stress-induced apoptosis. Nature 380:75–79
Xin M, Deng X (2006) Protein phosphatase 2A enhances the proapoptotic function of Bax through dephosphorylation. J Biol Chem 281:18859–18867
Heinrich M, Wickel M, Schneider-Brachert W et al (1999) Cathepsin D targeted by acid sphingomyelinase-derived ceramide. EMBO J 18:5252–5263
Heinrich M, Neumeyer J, Jakob M et al (2004) Cathepsin D links TNF-induced acid sphingomyelinase to Bid-mediated caspase-9 and -3 activation. Cell Death Differ 11:550–563
Simons K, Ehehalt R (2002) Cholesterol, lipid rafts, and disease. J Clin Invest 110:597–603
Rotolo JA, Zhang J, Donepudi M, Lee H, Fuks Z, Kolesnick R (2005) Caspase-dependent and -independent activation of acid sphingomyelinase signaling. J Biol Chem 280:26425–26434
Bollinger CR, Teichgraber V, Gulbins E (2005) Ceramide-enriched membrane domains. Biochim Biophys Acta 1746:284–294
Gulbins E, Kolesnick R (2003) Raft ceramide in molecular medicine. Oncogene 22:7070–7077
Morita Y, Perez GI, Paris F et al (2000) Oocyte apoptosis is suppressed by disruption of the acid sphingomyelinase gene or by sphingosine-1-phosphate therapy. Nat Med 6:1109–1114
Perez GI, Jurisicova A, Matikainen T et al (2005) A central role for ceramide in the age-related acceleration of apoptosis in the female germline. FASEB J 19:860–862
Potten CS (1990) A comprehensive study of the radiobiological response of the murine (BDF1) small intestine. Int J Radiat Biol 58:925–973
Potten CS, Booth C, Pritchard DM (1997) The intestinal epithelial stem cell: the mucosal governor. Int J Exp Pathol 78:219–243
Petrache I, Natarajan V, Zhen L et al (2005) Ceramide upregulation causes pulmonary cell apoptosis and emphysema-like disease in mice. Nat Med 11:491–498
Ch’ang HJ, Maj JG, Paris F et al (2005) ATM regulates target switching to escalating doses of radiation in the intestines. Nat Med 11:484–490
Lin T, Genestier L, Pinkosli MJ et al (2000) Role of acidic sphingomyelinase in Fas/CD95-mediated cell death. J Biol Chem 275:8657–8663
Luberto C (2002) Inhibition of tumor necrosis factor-induced cell death in MCF-7 by a novel inhibitor of neutral sphingomyelinase. J Biol Chem 277:41128–41139
Liu JJ, Wang JY, Hertervig E et al (2000) Activation of neutral sphingomyelinase participates in ethanol-induced apoptosis in HepG2 cells. Alcohol Alcohol 35:569–573
Scarlatti F, Bauvy C, Ventruti A et al (2004) Ceramide-mediated macroautophagy involves inhibition of protein kinase B and up-regulation of beclin 1. J Biol Chem 279:18384–18391
Daido S, Kanzawa T, Yamamoto A, Takeuchi H, Kondo Y, Kondo S (2004) Pivotal role of the cell death factor BNIP3 in ceramide-induced autophagic cell death in malignant glioma cells. Cancer Res 64:4286–4293
Dunn W (1994) Autophagy and related mechanisms of lysosome-mediated protein degradation. Trends Cell Biol 4:139–143
Levine B, Yuan J (2005) Autophagy in cell death: an innocent convict? J Clin Invest 115:2679–2688
Boya P, Gonzalez-Polo RA, Casares N et al (2005) Inhibition of macroautophagy triggers apoptosis. Mol Cell Biol 25:1025–1040
Leist M, Jäättelä M (2001) Four death and a funeral: from caspases to alternative mechanisms. Nat Rev Mol Cell Biol 2:589–598
Kroemer G, Martin SJ (2005) Caspase-independent cell death. Nat Med 11:725–730
Ferri K, Kroemer G (2001) Organelle-specific initiation of cell death pathways. Nat Cell Biol 3:E255–E263
Walter L, Hajnoczky G (2005) Mitochondria and endoplasmic reticulum: the lethal interorganelle cross-talk. J Bioenerg Biomem 37:191–206
Shiraishi H, Okamoto H, Yoshimura A, Yoshida H (2006) ER stress-induced apoptosis and caspase-12 activation occurs downstream of mitochondrial apoptosis involving Apaf-1. J Cell Sci 119:3958–3966
Boya P, Cohen I, Zamzami N, Vieira HL, Kroemer G (2002) Endoplasmic reticulum stress-induced cell death requires mitochondrial membrane permeabilization. Cell Death Differ 9: 465–467
Simmen T, Aslan JE, Blagoveschchenskaya AD et al (2005) PACS-2 controls endoplasmic reticulum-mitochondria communication and Bid-mediated apoptosis. EMBO J 24:717–729
Marciniak SJ, Ron D (2006) Endoplasmic reticulum stress signaling in disease. Physiol Rev 86:1133–1149
Wu J, Kaufman RJ (2006) From acute ER stress to physiological roles of the Unfolded Protein Response. Cell Death Differ 13:374–384
Hwang C, Sinskey AJ, Lodish HF (1992) Oxidized redox state of glutathione in the endoplasmic reticulum. Science 257:1496–1502
Zhao L, Ackerman SL (2006) Endoplasmic reticulum stress in health and disease. Curr Opin Cell Biol 18:444–452
Ron D (2006) Stressed cells cope with protein overload. Science 313:52–53
Hollien J, Weissmann JS (2006) Decay of Endoplasmic Reticulum-Localized mRNAs During the Unfolded Protein Response. Science 313:104–107
Harding HP, Zhang Y, Zeng H et al (2003) An integrated stress response regulates amino acid metabolism and resistance to oxidative stress. Mol Cell 11:619–633
Breckenridge DG, Germain M, Mathai JP, Nguyen M, Shore GC (2003) Regulation of apoptosis by endoplasmic reticulum pathways. Oncogene 22:8608–8618
Ledeen RW, Wu G (2002) Ganglioside function in calcium homeostasis and signaling. Neurochem Res 27:637–647
d’Azzo A, Tessitore A, Sano R (2006) Gangliosides as apoptotic signals in ER stress response. Cell Death Differ 13:404–414
Pelled D, Lloyd-Evans E, Riebeling C, Jeyakumar M, Platt FM, Futerman AH (2003) Inhibition of calcium uptake via the sarco/endoplasmic reticulum Ca2+-ATPase in a mouse model of Sandhoff disease and prevention by treatment with N-butyldeoxynojirimycin. J Biol Chem 278: 29496–29501
Lloyd-Evans E, Pelled D, Riebeling C et al (2003) Glucosylceramide and glucosylsphingosine modulate calcium mobilization from brain microsomes via different mechanisms. J Biol Chem 278:23594–23599
Tessitore A, del P Martin M, Sano R et al (2004) GM1-ganglioside-mediated activation of the unfolded protein response causes neuronal death in a neurodegenerative gangliosidosis. Mol Cell 10:753–766
Carracedo A, Lorente M, Egia A et al (2006) The stress-regulated protein p8 mediates cannabinoid-induced apoptosis of tumor cells. Cancer Cell 9:301–312
Green DR, Kroemer G (2004) The pathophysiology of mitochondrial cell death. Science 305:626–629
Gogvadze V, Orrenius S, Zhivotovsky B (2006) Multiple pathways of cytochrome c release from mitochondria in apoptosis. Biochim Biophys Acta 1757:639–647
Zamzami N, Kroemer G (2001) The mitochondrion in apoptosis: how Pandora's box opens. Nat Rev Mol Cell Biol 2:67–71
Martinou JC, Green DR (2001) Breaking the mitochondrial barrier. Nat Rev Mol Cell Biol 2:63–67
Garcia-Ruiz C, Colell A, Mari M, Morales A, Fernandez-Checa JC (1997) Direct effect of ceramide on the mitochondrial electron transport chain leads to generation of reactive oxygen species. Role of mitochondrial glutathione. J Biol Chem 272:11369–11377
Dai Q, Liu J, Chen J, Durrant D, McIntyre TM, Lee RM (2004) Mitochondrial ceramide increases in UV-irradiated HeLa cells and is mainly derived from hydrolysis of sphingomyelin. Oncogene 23:3650–3658
Birbes H, Luberto C, Hsu YT, El Bawab S, Hannun YA, Obeid LM (2005) A mitochondrial pool of sphingomyelin is involved in TNFalpha-induced Bax translocation to mitochondria. Biochem J 386:445–451
Dindo D, Dahm F, Szluc Z et al (2006) Cationic long-chain ceramide LCL-30 induced cell death by mitochondrial targeting in SW403 cells. Mol Cancer Ther 5:1520–1529
Gudz TI, Tserng KY, Hoppel Cl (1997) Direct inhibition of mitochondrial respiratory chain complex III by cell-permeable ceramide. J Biol Chem 272:24154–24158
Quillet-Mary A, Jaffrezou JP, Mansat V, Bordier C, Naval J, Laurent G (1997) Implication of mitochondrial hydrogen peroxide generation in ceramide-induced apoptosis. J Biol Chem 272:21388–21395
Birbes H, El Bawab S, Hannun YA, Obeid LM (2001) Selective hydrolysis of a mitochondrial pool of sphingomyelin induces apoptosis. FASEB J 15:2669–2679
Novgorodov SA, Szule ZM, Luberto C et al (2005) Positively charged ceramide is a potent inducer of mitochondrial permeabilization. J Biol Chem 280:16096–16105
Rippo MR, Malisan F, Ravagnan L et al (2000) GD3 ganglioside directly targets mitochondria in a bcl-2-controlled fashion. FASEB J 14:2047–2054
García-Ruiz C, Colell A, París R, Fernández-Checa JC (2000) Direct interaction of GD3 ganglioside with mitochondria generates reactive oxygen species followed by mitochondrial permeability transition, cytochrome c release and caspase activation. FASEB J 14:847–858
Garcia-Ruiz C, Colell A, Morales A, Calvo M, Enrich C, Fernandez-Checa JC (2002) Trafficking of ganglioside GD3 to mitochondria by tumor necrosis factor-alpha. J Biol Chem 277:36443–36448
DeMaria R, Lenti T, Malissan F et al (1997) Requirement for GD3 ganglioside in ceramide-induced apoptosis. Science 277:1652–1655
Colell A, Morales A, Fernandez-Checa JC, Garcia-Ruiz C (2002) Ceramide generated by acidic sphingomyelinase contributes to tumor necrosis factor-mediated apoptosis in HT-29 cells through glycosphingolipid generation. Possible role of ganglioside GD3. FEBS Lett 526:135–141
Copani A, Melchiorri D, Caricasole A et al (2002) Beta-amyloid-induced synthesis of ganglioside GD3 is a requisite for cell cycle reactivation and apoptosis in neurons. J Neurosci 22:3963–3968
Garofalo T, Giammarioli AM, Misasi R et al (2005) Lipid microdomains contribute to apoptosis-associated modifications of mitochondria in T cells. Cell Death Differ 12:1378–1389
Braun T, Carvalho G, Fabre C, Grosjean J, Fenaux P, Kroemer G (2006) Targeting NF-kappaB in hematologic malignancies. Cell Death Differ 13:748–758
Colell A, Garcia-Ruiz C, Roman J, Ballesta, Fernandez-Checa JC (2001) Ganglioside GD3 enhances apoptosis by suppressing the nuclear factor-kappaB-dependent survival pathway. FASEB J 15:1068–1070
Paris R, Morales A, Coll O, Sanchez-Reyes A, Garcia-Ruiz C, Fernandez-Checa JC (2002) Ganglioside GD3 sensitizes human hepatoma cells to cancer therapy. J Biol Chem 277:49870–49876
Malisan F, Franchi L, Tomassini B et al (2002) Acetylation suppresses the proapoptotic activity of GD3 ganglioside. J Exp Med 196:1535–1541
Chapman PB, Wu D, Ragupath G et al (2004) Sequential immunization of melanoma patients with GD3 ganglioside vaccine and anti-idiotypic monoclonal antibody that mimics GD3 ganglioside. Clin Cancer Res 10:4717–4723
Mari M, Caballero F, Colell A et al (2006) Mitochondrial free cholesterol loading sensitizes to TNF- and Fas-mediated steatohepatitis. Cell Metab 4:185–198
Segui B, Cuvillier O, Adam-Klagues S et al (2001) Involvement of FAN in TNF-induced apoptosis. J Clin Invest 108:143–151
Malagarie-Cazenave S, Segui B, Leveque S et al (2004) Role of FAN in tumor necrosis factor-alpha and lipopolysaccharide-induced interleukin-6 secretion and lethality in D-galactosamine-sensitized mice. J Biol Chem 279:18648–18655
Gupta S, Natarajan R, Payne SG et al (2004) Deoxycholic acid activates c-Jun N-terminal kinase pathway via FAS-receptor activation in primary hepatocytes. Role of acidic sphingomyelinase-mediated ceramide generation in FAS receptor activation. J Biol Chem 279:5821–5828
Corazza N, Jakob S, Schaer C et al (2006) TRAIL receptor-mediated JNK activation and Bim phosphorylation critically regulate Fas-mediated liver damage and lethality. J Clin Invest 116:2493–2499
Malhi H, Bronk SD, Werneburg NW, Gores GJ (2006) Free fatty acids induce JNK-dependent hepatocyte lipoapoptosis. J Biol Chem 281:12093–12101
Osawa Y, Hannun YA, Proia RL, Brenner DA (2005) Roles of AKT and sphingosine kinase in the antiapoptotic effects of bile duct ligation in mouse liver. Hepatology 42:1320–1328
Zheng DM, Kitamura T, Ikejima K et al (2006) Sphingosine 1-phosphate protects rat liver sinusoidal endothelial cells from ethanol-induced apoptosis: role of intracellular calcium and nitric oxide. Hepatology 44:1278–1287
Cutler RG, Kelly JK, Storie K et al (2004) Involvement of oxidative stress-induced abnormalities in ceramide and cholesterol metabolism in brain aging and Alzheimer's disease. Proc Natl Acad Sci USA 101:2070–2075
Lee JT, Xu J, Lee JM et al (2004) Amyloid-β peptide induces oligodendrocyte death by activating the neutral sphingomyelinase-ceramide pathway. J Cell Biol 164:123–131
Jana A, Pahan K (2004) Fibrillar amyloid-b peptides kill human primary neurons via NADPH oxidase-mediated activation of neutral sphingomyelinase. J Biol Chem 279:51451–51459
Grimm MO, Grimm HS, Patzold AJ et al (2005) Regulation of cholesterol and sphingomyelin metabolism by amyloid-β and presenilin. Nat Cell Biol 7:1118–1123
Yu ZF, Nikolova-Karakashian M, Zhou D, Cheng G, Schuchman EH, Mattson MP (2000) Pivotal role for acidic sphingomyelinase in cerebral ischemia-induced ceramide and cytokine production, and neuronal apoptosis. J Mol Neurosci 15:85–97
Simon BM, Malisan F, Testi R, Nicotera P, Leist M (2002) Disialoganglioside GD3 in released by microglia and induces oligodendrocyte apoptosis. Cell Death Differ 9:758–767
Selzner M, Bielaswska A, Morse MA et al (2001) Induction of apoptotic cell death and prevention of turmor growth by ceramide analogues in metastatic human colon cancer. Cancer Res 61:1233–1240
Morales A, Paris R, Villanueva A, Llacuna L, Garcia-Ruiz C, Fernandez-Checa JC (2006) Pharmacological inhibition of small interfering RNA targeting acid ceramidase sensitizes hepatoma cells to chemotherapy and reduces tumor growth in vivo. Oncogene in press
Visentin B, Vekich JA, Sibbald BJ et al (2006) Validation of an anti-sphingosine-1-phosphate antibody as a potential therapeutic in reducing growth, invasion, and angiogenesis in multiple tumor lineages. Cancer Cell 9:225–238
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Morales, A., Lee, H., Goñi, F.M. et al. Sphingolipids and cell death. Apoptosis 12, 923–939 (2007). https://doi.org/10.1007/s10495-007-0721-0
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10495-007-0721-0