Abstract
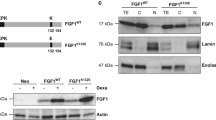
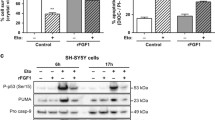
The survival activity of FGF1 and the pro-apoptotic activity of p53 were characterized in vitro and/or in vivo for different types of neurons after different stresses and in different neurodegenerative pathologies. To investigate whether or not FGF1 and p53 pathways interact in neuronal cells, we studied the effect of FGF1 on p53-dependent apoptosis in PC12 cells. We first characterized p53-dependent PC12 cell death induced by etoposide (a DNA damaging agent). We showed that etoposide increased p53 stabilization, phosphorylation (Ser-15), nuclear translocation and transcriptional activity. In particular, p53 promoted mdm2, p21, puma and noxa expression in PC12 cells. The activation of p53 initiated a classical mitochondrial apoptosis process associated with caspases activation and nuclear degradation. We demonstrated that FGF1 protected PC12 cells from p53-dependent apoptosis upstream from mitochondrial and nuclear events. FGF1 inhibited etoposide-induced p53 phosphorylation, stabilization, nuclear translocation and transcriptional activity. This study presents the first evidence that FGF1 and p53 pathways interact in neuronal cells, and that FGF1 protects neuronal cells from p53-dependent apoptosis, suggesting that alterations of FGF1/p53 crosstalk could be involved in a large range of neurons and in neurological disorders.






Similar content being viewed by others
References
Culmsee C, Mattson MP (2005) p53 in neuronal apoptosis. Biochem Biophys Res Commun 331:761–777
Vousden KH, Lu X (2002) Live or let die: the cell’s response to p53. Nat Rev Cancer 2:594–604
Yu J, Zhang L (2005) The transcriptional targets of p53 in apoptosis control. Biochem Biophys Res Commun 331:851–858
Brooks CL, Gu W (2006) p53 ubiquitination: Mdm2 and beyond. Mol Cell 21:307–315
Ho J, Benchimol S (2003) Transcriptional repression mediated by the p53 tumour suppressor. Cell Death Differ 10:404–408
Godefroy N, Bouleau S, Gruel G et al (2004) Transcriptional repression by p53 promotes a Bcl-2-insensitive and mitochondria-independent pathway of apoptosis. Nucleic Acids Res 32:4480–4490
Chipuk JE, Green DR (2006) Dissecting p53-dependent apoptosis. Cell Death Differ 13:994–1002
Bouleau S, Grimal H, Rincheval V et al (2005) FGF1 inhibits p53-dependent apoptosis and cell cycle arrest via an intracrine pathway. Oncogene 24:7839–7849
Powers CJ, McLeskey SW, Wellstein A (2000) Fibroblast growth factors, their receptors and signaling. Endocr Relat Cancer 7:165–197
Szebenyi G, Fallon JF (1999) Fibroblast growth factors as multifunctional signaling factors. Int Rev Cytol 185:45–106
Wiedlocha A, Sorensen V (2004) Signaling, internalization, and intracellular activity of fibroblast growth factor. Curr Top Microbiol Immunol 286:45–79
Imamura T, Engleka K, Zhan X et al (1990) Recovery of mitogenic activity of a growth factor mutant with a nuclear translocation sequence. Science 249:1567–1570
Wesche J, Malecki J, Wiedlocha A et al (2005) Two nuclear localization signals required for transport from the cytosol to the nucleus of externally added FGF-1 translocated into cells. Biochemistry 44:6071–6080
Renaud F, Oliver L, Desset S et al (1994) Up-regulation of aFGF expression in quiescent cells is related to cell survival. J Cell Physiol 158:435–443
Renaud F, Desset S, Oliver L et al (1996) The neurotrophic activity of fibroblast growth factor 1 (FGF1) depends on endogenous FGF1 expression and is independent of the mitogen-activated protein kinase cascade pathway. J Biol Chem 271:2801–2811
Desire L, Courtois Y, Jeanny JC (1998) Suppression of fibroblast growth factors 1 and 2 by antisense oligonucleotides in embryonic chick retinal cells in vitro inhibits neuronal differentiation and survival. Exp Cell Res 241:210–221
Heron-Milhavet L, LeRoith D (2002) Insulin-like growth factor I induces MDM2-dependent degradation of p53 via the p38 MAPK pathway in response to DNA damage. J Biol Chem 277:15600–15606
Anderson CN, Tolkovsky AM (1999) A role for MAPK/ERK in sympathetic neuron survival: protection against a p53-dependent, JNK-independent induction of apoptosis by cytosine arabinoside. J Neurosci 19:664–673
Gottlieb TM, Leal JF, Seger R, Taya Y, Oren M (2002) Cross-talk between Akt, p53 and Mdm2: possible implications for the regulation of apoptosis. Oncogene 21:1299–1303
Mayo LD, Donner DB (2002) The PTEN, Mdm2, p53 tumor suppressor-oncoprotein network. Trends Biochem Sci 27:462–467
Ueba T, Nosaka T, Takahashi JA et al (1994) Transcriptional regulation of basic fibroblast growth factor gene by p53 in human glioblastoma and hepatocellular carcinoma cells. Proc Natl Acad Sci USA 91:9009–9013
Galy B, Creancier L, Zanibellato C, Prats AC, Prats H (2001) Tumour suppressor p53 inhibits human fibroblast growth factor 2 expression by a post-transcriptional mechanism. Oncogene 20:1669–1677
Shaulian E, Resnitzky D, Shifman O et al (1997) Induction of Mdm2 and enhancement of cell survival by bFGF. Oncogene 15:2717–2725
Eckenstein F, Woodward WR, Nishi R (1991) Differential localization and possible functions of aFGF and bFGF in the central and peripheral nervous systems. Ann NY Acad Sci 638:348–360
Bugra K, Oliver L, Jacquemin E, Laurent M, Courtois Y, Hicks D (1993) Acidic fibroblast growth factor is expressed abundantly by photoreceptors within the developing and mature rat retina. Eur J Neurosci 5:1586–1595
Stock A, Kuzis K, Woodward WR, Nishi R, Eckenstein FP (1992) Localization of acidic fibroblast growth factor in specific subcortical neuronal populations. J Neurosci 12:4688–4700
Walicke PA (1988) Basic and acidic fibroblast growth factors have trophic effects on neurons from multiple CNS regions. J Neurosci 8:2618–2627
Raguenez G, Desire L, Lantrua V, Courtois Y (1999) BCL-2 is upregulated in human SH-SY5Y neuroblastoma cells differentiated by overexpression of fibroblast growth factor 1. Biochem Biophys Res Commun 258:745–751
Cuevas P, Carceller F and Gimenez-Gallego G (1995) Acidic fibroblast growth factor prevents death of spinal cord motoneurons in newborn rats after nerve section. Neurol Res 17:396–399
Greene LA, Tischler AS (1976) Establishment of a noradrenergic clonal line of rat adrenal pheochromocytoma cells which respond to nerve growth factor. Proc Natl Acad Sci USA 73:2424–2428
Rydel RE, Greene LA (1987) Acidic and basic fibroblast growth factors promote stable neurite outgrowth and neuronal differentiation in cultures of PC12 cells. J Neurosci 7:3639–3653
Nakajima M, Kashiwagi K, Ohta J et al (1994) Nerve growth factor and epidermal growth factor rescue PC12 cells from programmed cell death induced by etoposide: distinct modes of protection against cell death by growth factors and a protein-synthesis inhibitor. Neurosci Lett 176:161–164
Wu Y, Karas M, Dupont J, Zhao H, Toyoshima Y, Le Roith D (2004) Multiple signaling pathways are involved in the regulation of IGF-I receptor inhibition of PTEN-enhanced apoptosis. Growth Horm IGF Res 14:52–58
Komarov PG, Komarova EA, Kondratov RV et al (1999) A chemical inhibitor of p53 that protects mice from the side effects of cancer therapy. Science 285:1733–1737
Shieh SY, Ikeda M, Taya Y, Prives C (1997) DNA damage-induced phosphorylation of p53 alleviates inhibition by MDM2. Cell 91:325–334
Reimertz C, Kogel D, Rami A, Chittenden T, Prehn JH (2003) Gene expression during ER stress-induced apoptosis in neurons: induction of the BH3-only protein Bbc3/PUMA and activation of the mitochondrial apoptosis pathway. J Cell Biol 162:587–597
Cregan SP, Arbour NA, Maclaurin JG et al (2004) p53 activation domain 1 is essential for PUMA upregulation and p53-mediated neuronal cell death. J Neurosci 24:10003–10012
Wyttenbach A, Tolkovsky AM (2006) The BH3-only protein Puma is both necessary and sufficient for neuronal apoptosis induced by DNA damage in sympathetic neurons. J Neurochem 96:1213–1226
Zhu X, Yu QS, Cutler RG et al (2002) Novel p53 inactivators with neuroprotective action: syntheses and pharmacological evaluation of 2-imino-2,3,4,5,6,7-hexahydrobenzothiazole and 2-imino-2,3,4,5,6,7-hexahydrobenzoxazole derivatives. J Med Chem 45:5090–5097
Khanna KK, Keating KE, Kozlov S et al (1998) ATM associates with and phosphorylates p53:mapping the region of interaction. Nat Genet 20:398–400
Cho YY, He Z, Zhang Y et al (2005) The p53 protein is a novel substrate of ribosomal S6 kinase 2 and a critical intermediary for ribosomal S6 kinase 2 and histone H3 interaction. Cancer Res 65:3596–3603
Chan SL, Culmsee C, Haughey N, Klapper W, Mattson MP (2002) Presenilin-1 mutations sensitize neurons to DNA damage-induced death by a mechanism involving perturbed calcium homeostasis and activation of calpains and caspase-12. Neurobiol Dis 11:2–19
Dumaz N, Meek DW (1999) Serine15 phosphorylation stimulates p53 transactivation but does not directly influence interaction with HDM2. Embo J 18:7002–7010
Biswas SC, Ryu E, Park C, Malagelada C, Greene LA (2005) Puma and p53 play required roles in death evoked in a cellular model of Parkinson disease. Neurochem Res 30:839–845
Acknowledgements
This work was supported in part by grants from the “Association pour la Recherche Contre le Cancer” (#3367) and the “Ligue Nationale Contre le Cancer”. Sylvina Bouleau held successive fellowships from the “Ministère de l’Education Nationale, de l’Enseignement Supérieur et de la Recherche” and the “Association pour la Recherche contre le Cancer”. Ioana Pârvu-Ferecatu is supported by a fellowships from the “Ministère de l’Education Nationale, de l’Enseignement Supérieur et de la Recherche”.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bouleau, S., Pârvu-Ferecatu, I., Rodriguez-Enfedaque, A. et al. Fibroblast Growth Factor 1 inhibits p53-dependent apoptosis in PC12 cells. Apoptosis 12, 1377–1387 (2007). https://doi.org/10.1007/s10495-007-0072-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10495-007-0072-x