Abstract
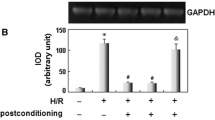
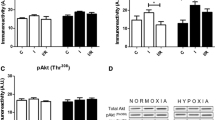
A sequence of intermittent interruptions of oxygen supply (i.e., postconditioning, Postcon) at reoxygenation reduces oxidant-induced cardiomyocyte loss. This study tested the hypothesis that prevention of cardiomyocyte apoptosis by Postcon is mediated by mitogen-activated protein kinases pathways. Primary cultured neonatal rat cardiomyocytes were exposed to 3 h hypoxia followed by 6 h of reoxygenation. Cardiomyocytes were postconditioned by three cycles each of 5 min reoxygenation and 5 min hypoxia after prolonged hypoxia. Relative to hypoxia alone, reoxygenation stimulated expression of JNKs and p38 kinases, corresponding to increased activity of JNKs (phospho-c-Jun) and p38 (phospho-ATF2). The level of TNFα in cell lysates, activity of cytosolic caspases-8, -3, expression of Bax and the number of apoptotic cardiomyocytes were increased while expression of Bcl-2 was decreased with reoxygenation. Consistent with an attenuation in generation of superoxide anions detected by lucigenin-enhanced chemiluminescence at early period of reoxygenation, treatment of cardiomyocytes with Postcon further reduced expression and activity of JNKs and p38 kinases, level of TNFα, the frequency of apoptotic cells and expression of Bax. However, the inhibitory effects of Postcon on these changes were lost when its application was delayed by 5 min after the start of reoxygenation. Addition of a JNK/p38 stimulator, anisomycin into cardiomyocytes at the beginning of reoxygenation eliminated protection by Postcon. These data suggest that 1) hypoxia/reoxygenation elicits cardiomyocyte apoptosis in conjunction with expression and activation of JNK and p38 kinases, release of TNFα, activation of caspases, and an increase in imbalance of pro-/anti-apoptotic proteins; 2) Postcon attenuates cardiomyocyte apoptosis, potentially mediated by inhibiting JNKs/p-38 signaling pathways and reducing TNFα release and caspase expression.
Similar content being viewed by others
References
Zhao Z-Q, Corvera JS, Halkos ME et al (2003) Inhibition of myocardial injury by ischemic postconditioning during reperfusion: comparison with ischemic preconditioning. Am J Physiol (Heart Circ Physiol) 285:579–588
Kin H, Zhao Z-Q, Sun H-Y et al (2004) Postconditioning attenuates myocardial ischemia-reperfusion injury by inhibiting events in the early minutes of reperfusion. Cardiovasc Res 62:74–85
Sun H-Y, Wang N-P, Kerendi F et al (2005) Hypoxic postconditioning reduces cardiomyocyte loss by inhibiting ROS generation and intracellular Ca2+ overload. Am J Phy 288:H1900–H1908
Yang X-M, Proctor JB, Cui L, Krieg T, Downey JM, Cohen MV (2004) Multiple, brief coronary occlusions during early reperfusion protect rabbit hearts by targeting cell signaling pathways. J Am Coll Card 44:1103–1110
Darling CE, Jiang R, Maynard M, Whittaker P, Vinten-Johansen J, Przyklenk K (2005) Postconditioning via stuttering reperfusion limits myocardial infarct size in rabbit hearts: role of ERK1/2. Am J Physiol (Heart Circ Physiol) 289:H1618–H1626
Tsang A, Hausenloy DJ, Mocanu MM, Yellon DM (2004) Postconditioning: a form of “modified reperfusion” protects the myocardium by activating the phosphatidylinositol 3-kinase-Akt pathway. Circul Res 95:230–232
Laskey WK (2005) Brief repetitive balloon occlusions enhance reperfusion during percutaneous coronary intervention for acute myocardial infarction: a pilot study. Catheteriz Cardiovasc Interven 65:361–367
Staat P, Rioufol G, Piot C et al (2005) Postconditioning the human heart. Circulation 112:2143–2148
Mackay K, Mochly-Rosen D (1999) An inhibitor of p38 mitogen-activated protein kinase protects neonatal cardiac myocytes from ischemia. J Biol Chem 274:6272–6279
Meldrum DR, Dinarello CA, Cleveland JC, Jr et al (1998) Hydrogen peroxide induces tumor necrosis factor alpha-mediated cardiac injury by a P38 mitogen-activated protein kinase-dependent mechanmism. Surgery 124:291–297
Klein JB, Wang GW, Zhou Z, Buridi A, Kang YJ (2002) Inhibition of tumor necrosis factor-alpha-dependent cardiomyocyte apoptosis by metallothionein. Cardiovasc Toxicol 2:209–218
Lee MW, Park SC, Yang YG, et al (2002) The involvement of reactive oxygen species (ROS) and p38 mitogen-activated protein (MAP(kinase in TRAIL/Apo2L-induced apoptosis. FEBS Letters 512:313–318
Osone S, Hosoi H, Kuwahara Y, Matsumoto Y, Iehara T, Sugimoto T (2004) Fenretinide induces sustained-activation of JNK/p38 MAPK and apoptosis in a reactive oxygen species-dependent manner in neuroblastoma cells. Int J Cancer 112:219–224
Yoshizumi M, Tsuchiya K, Tamaki T (2001) Signal transduction of reactive oxygen species and mitogen-activated protein kinases in cardiovascular disease. J Invest Med 48:11–24
Marczin N, Bundy RE, Hoare GS, Yacoub M (2003) Redox regulation following cardiac ischemia and reperfusion. Coron Artery Dis 14:123–133
Hahn RA, MacDonald BR, Morgan E et al (1992) Evaluation of LY203647 on cardiovascular leukotriene D4 receptors and myocardial reperfusion injury. Journal of Pharmacol Experim Therap 260:979–989
Gao F, Yue T-L, Shi D-W, et al (2002) p38 MAPK inhibition reduces myocardial reperfusion injury via inhibition of endothelial adhesion molecule expression and blockade of PMN accumulation. Cardiovasc Res 53:414–422
Clanachan AS, Jaswal JS, Gandhi M et al (2003) Effects of inhibition of myocardial extracellular-responsive kinase and p38 mitogen-activated protein kinase on mechanical function of rat hearts after prolonged hypothermic ischemia. Transplant 75:173–180
Cicconi S, Ventura N, Pastore D et al (2003) Characterization of apoptosis signal transduction pathways in HL-5 cardiomyocytes exposed to ischemia/reperfusion oxidative stress model. J Cell Physiol 195:27–37
Fan H, Sun B, Gu Q, Lafond-Walker A, Cao S, Becker LC (2002) Oxygen radicals trigger activation of NF-kB and AP-1 and upregulation of ICAM-1 in reperfused canine heart. Am J Physiol (Heart Circ Physiol) 282:H1778–H1786
Wang T, Zhang X, Li J-J (2002) The role of NF-κB in the regulation of cell stress responses. Intern Immunopharm 2:1509–1520
Meldrum DR (1998) Tumor necrosis factor in the heart. Am J Physiol 274:R577–R595
Zhao Z-Q, Vinten-Johansen J (2002) Myocardial apoptosis and ischemic preconditioning. Cardiovasc Res 55:438–455
Sun H-Y, Wang N-P, Halkos ME et al (2004) Involvement of Na+/H+ exchanger in hypoxia/re-oxygenation-induced neonatal rat cardiomyocyte apoptosis. Euro J Pharmacol 486:121–131
Laderoute KR, Webster KA (1997) Hypoxia/reoxygenation stimulates jun kinase activity through redox signaling in cardiac myocytes. Circul Res 80:336–344
Yang X-M, Philipp S, Downey JM, Cohen MV (2005) Postconditioning's protection is not dependent on circulating blood factors or cells but involves adenosine receptors and requires PI3-kinase and guanylyl cyclase activation. Basic Res Cardiol 100:57–63
Kin H, Zatta AJ, Lofye MT et al (2005) Postconditioning reduces infarct size via adenosine receptor activation by endogenous adenosine. Cardiovasc Res 67:124–133
Nakano A, Cohen MV, Critz SD, Downey JM (2000) SB 203580, an inhibitor of p38 MAPK, abolishes infarct-limiting effect of iscehmic preconditioning in isolated rabbit hearts. Basic Res Cardiol 95:466–471
Weinbrenner C, Liu GS, Cohen MV, Downey JM (1997) Phosphorylation of Tyrosine 182 of p38 mitogen activated protein kinase correlates with the protection of preconditioning in the rabbit heart. J Molec Cell Cardiol 29:2383–2391
Kaiser RA, Bueno OF, Lips DJ et al (2004) Targeted inhibition of p38 mitogen-activated protein kinase antagonizes cardiac injury and cell death following ischemia-reperfusion in vivo. J Biolog Chem 279:15524–15530
Ferrandi C, Ballerio R, Gaillard P et al (2004) Inhibition of c-Jun N-terminal kinase decreases cardiomyocyte apoptosis and infarct size after myocardial ischemia and reperfusion in anaesthetized rats. British J Pharmacol 142:953–960
Ballard-Croft C, White DJ, Maass DL, Hybki DP, Horton JW (2001) Role of p38 mitogen-activated protein kinase in cardiac myocyte seretion of the inflammatory cytokine TNF-alpha. Am J Physiol (Heart Circ Physiol) 280:H1970–H1981
Brown M, McGuinness M, Wright T et al (2005) Cardiac-specific blockade of NF-kappaB in cardiac pathophysiology: differences between acute and chronic stimuli in vivo. Am J Physiol (Heart Circ Physiol) 289:H466–H476
Haunstetter A, Izumo S (1998) Apoptosis. Basic mechanisms and implications for cardiovascular disease. Circulation Research 82:1111–1129
Ma X-L, Kumar S, Gao F, et al (1999) Inhibition of p38 mitogen-activated protein kinase decreases cardiomyocyte apoptosis and improves cardiac function after myocardial ischemia and reperfusion. Circulation 99:1685–1691
Maekawa N, Wada H, Kanda T et al (2002) Improved myocardial ischemia/reperfusion injury in mice lacking tumor necrosis factor-alpha. J Am Coll Cardiol 39:1229–1235
Chandra J, Samali A, Orrenius S (2000) Triggering and modulation of apoptosis by oxidative stress. Free Radic Biol Medi 29:323–333
Esposti MD, Dive C (2003) Mitochondrial membrane permeabilisation by Bax/Bak. Biochem & Biophys Res Comm 304:455–461
Kuwana T, Newmeyer DD (2003) Bcl-2-family proteins and the role of mitochondria in apoptosis. Curr Opin Cell Biol 15:691–699
Halestrap AP, Kerr PM, Javadov S, Woodfield KY (1998) Elucidating the molecular mechanism of the permeability transition pore and its role in reperfusion injury of the heart. Biochim Biophys Acta 1366:79–94
Kim JS, He L, Lemasters JJ (2003) Mitochondrial permeability transition: a common pathway to necrosis and apoptosis. Biochem & Biophys Res Comm 304:463–470
Sun K, Liu ZS, Sun Q (2004) Role of mitochondria in cell apoptosis during hepatic ischemia-reperfusion injury and protective effect of ischemic postconditioning. World J Gastroenterol 10:1934–1938
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sun, HY., Wang, NP., Halkos, M. et al. Postconditioning attenuates cardiomyocyte apoptosis via inhibition of JNK and p38 mitogen-activated protein kinase signaling pathways. Apoptosis 11, 1583–1593 (2006). https://doi.org/10.1007/s10495-006-9037-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10495-006-9037-8