Abstract
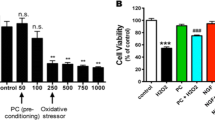
The aim of this study was to investigate the role of JAK-STAT pathway in the cytoprotection afforded by preconditioning with H2O2. It was shown that (1) Preconditioning with 100 μmol/L H2O2 can markedly protect PC12 cells against apoptosis and cytotoxicity induced by 300 μmol/L H2O2; (2) The expression and tyrosine phosphorylation of JAK2, not JAK1 were rapidly increased at 5 min after H2O2 preconditioning; (3) The expression of STAT1 and STAT3 were significantly increased at 15 min after H2O2 preconditioning, and the pTyr-STAT1 and pTyr-STAT3 were markedly increased at 60 min after H2O2 preconditioning; (4) Pretreatment with the JAK inhibitor AG-490 (10 μmol/L) 20 min before H2O2 preconditioning blocked not only the activation of JAK2, STAT1 and STAT3, but also the cytoprotection of H2O2 preconditioning against apoptosis and cytotoxicity induced by oxidative stress. These findings suggested that preconditioning with H2O2 activated the JAK-STAT pathway that played an important role in the cytoprotection induced by H2O2 preconditioning.
Similar content being viewed by others
References
Imada K, Leonard WJ. The Jak-STAT pathway. Mol Immunol 2000; 37: 1-11.
James E, Darnell, Jr. STATs and Gene Regulation. Science 1997; 277: 1630–1635.
Xuan YT, Guo Y, Han H, Zhu YQ, Bolli R. An essential role of the JAK-STAT pathway in ischemic preconditioning. PNAS 2001; 98: 9050–9055.
Bolli R, Dawn B, Xuan YT. Emerging role of the JAK-STAT pathway as a mechanism of protection against ischemia/reperfusion injury. J Mol Cell Cardiol 2001; 33: 1893–1896.
Stephanou A. Role of STAT-1 and STAT-3 in ischaemia/reperfusion injury. J Cell Mol Med 2004; 8: 519–525.
Velazquez L, Fellous M, Stark GR, Pellegrini S. A protein tyrosine kinase in the interferon alpha/beta signaling pathway. Cell 1992; 70: 313–322.
Watling D, Guschin D, Muller M, et al. Complementation by the protein tyrosine kinase JAK2 of a mutant cell line defective in the interferon-gamma signal transduction pathway. Nature 1993; 366: 166–170.
Levy DE, Kessler DS, Pine R, Reich N, Darnell JE, Jr. Interferon-induced nuclear factors that bind a shared promoter element correlate with positive and negative transcriptional control. Genes & Dev 1988; 2: 383–393.
Chin YE, Kitagawa M, Kuida K, Flavell RA, Fu XY. Activation of the STAT signaling pathway can cause _expression of caspase 1 and apoptosis. Mol Cell Biol 1997; 17: 5328–5337.
Chin YE, Kitagawa M, Su WCS, You ZH, Iwamoto Y, Fu XY. Cell Growth Arrest and Induction of Cyclin-Dependent Kinase Inhibitor p21WAF1/CIP1 Mediated by STAT1. Science 1996; 272: 719–722.
Stephanou A, Brar BK, Scarabelli TM, et al. Ischemia-induced STAT-1 _Expression and Activation PlayaCritical Role in Cardiomyocyte Apoptosis. J Biol Chem 2000; 275: 10002–10008.
Negoro S, Kunisada K, Tone E, et al. Activation of JAK/STAT pathway transduces cytoprotective signal in rat acute myocardial infarction. Cardiovasc Res 2000; 47: 797–805.
Smith RM, Suleman N, Lacerda L, et al. Genetic depletion of cardiac myocyte STAT-3 abolishes classical preconditioning. Cardiovasc Res 2004; 63: 611–616.
Yamaura G, Turoczi T, Yamamoto F, Siddqui MAQ, Maulik N, Das DK. STAT signaling in ischemic heart: A role of STAT5A in ischemic preconditioning. Am J Physiol Heart Circ Physiol 2003; 285: 476–482.
Lam M, Dubyak G, Chen L, Nunez G, Miesfeld RL, Distelhorst CW. Evidence that BCL-2 Represses Apoptosis by Regulating Endoplasmic Reticulum-Associated Ca2+ Fluxes. PNAS 1994; 91: 6569–6573.
Stephanou A, Brar BK, Knight RA, Latchman DS. Opposing actions of STAT-1 and STAT-3 on the Bcl-2 and Bcl-x promoters. Cell Death Differ 2000; 7: 329–330.
Simon AR, Rai U, Fanburg BL, Cochran BH. Activation of the JAK-STAT pathway by reactive oxygen species. Am J Physiol Cell Physiol 1998; 275: 1640–1652.
Carballo M, Conde M, Bekay RE, et al. Oxidative Stress Triggers STAT3 Tyrosine Phosphorylation and Nuclear Translocation in Human Lymphocytes. J Biol Chem 1999; 274: 17580–17586.
Lecanu L, Yao W, Teper GL, Yao ZX, Greeson J, Papadopoulos V. Identification of naturally occurring spirostenols preventing _-amyloid-induced neurotoxicity. Steroids 2004; 69: 1–16.
Andoh TS, Chock PB, Chiueh CC. The Roles of Thioredoxin in Protection against Oxidative Stress-induced Apoptosis in SH-SY5Y Cells. J Biol Chem 2002; 277: 9655–9660.
Coligan JE, Kruisbeek AM, Margulies DH, Shevach EM, Strober W. Current Protocols in Immunology (eds). Greene Publishing Associates, New York, 1993; p. 8.0.1.
Hattori R, Maulik N, Otani H, et al. Role of STAT3 in ischemic preconditioning. J Mol Cell Cardiol 2001; 33: 1929–1936.
Jacobson MD. Reactive oxygen species and programmed cell death. Trends Biochem Sci 1996; 21: 83–86.
Choi ME, Ballermann BJ. Inhibition of Capillary Morphogenesis and Associated Apoptosis by Dominant Negative Mutant Transforming Growth Factor-β Receptors. J Biol Chem 1995; 270: 21144–21150.
Stadtman ER, Berlett BS. Reactive oxygen-mediated protein oxidation in aging and disease. Chem Res Toxicol 1997; 10: 485–494.
Youdim MB, Fridkin M, Zheng H. Novel bifunctional drugs targeting monoamine oxidase inhibition and iron chelation as an approach to neuroprotection in Parkinson’s disease and other neurodegenerative diseases. J Neural Transm 2004; 111: 1455–1471.
Zhang W, Qin L, Wang T, et al. 3-hydroxymorphinan is neurotrophic to dopaminergic neurons and is also neuroprotective against LPS-induced neurotoxicity. FASEB J 2005; 19: 395–397.
Markesbery WR, Lovell MA. Four-hydroxynonenal, a product of lipid peroxidation, is increased in the brain in Alzheimer’s disease. Neurobiol Aging 1998; 19: 33–36.
Butterfield DA, Lauderback CM. Lipid peroxidation and protein oxidation in Alzheimer’s disease brain: potential causes and consequences involving amyloid beta-peptide-associated free radical oxidative stress. Free Radic Biol Med 2002; 32: 1050–1060.
Butterfield DA, Drake J, Pocernich C, Castegna A. Evidence of oxidative damage in Alzheimer’s disease brain: Central role for amyloid beta-peptide. Trends Mol Med 2001; 7: 548–554.
Manoury B, Nenan S, Leclerc O, et al. The absent of reactive oxygen species production protects mice against bleomycin-induced pulmonary fibrosis. Respir Res 2005; 6: 11.
Madamanchi NR, Li SZ, Patterson C, Runge MS. Reactive Oxygen Species Regulate Heat-Shock Protein 70 via the JAK/STAT Pathway. Arterioscler. Thromb. Vasc Biol 2001; 21: 321–326.
Han H, Wang HZ, Long H, Nattel S, Wang ZG. Oxidative Preconditioning and Apoptosis in L-cells. Roles of protein kinase B and mitogen-activated protein kinase. J Biol Chem 2001; 276: 26357–26364.
Lee HT, Xu H, Ota-Setlik A, Emala CW. Oxidant preconditioning protects human proximal tubular cells against lethal oxidant injury via p38 MAPK and heme oxygenase-1. Am J Nephrol 2003; 23: 324–333.
Decker T, Kovarik P, Meinke A. GAS elements: a few nucleotides with a major impact on cytokine-induced gene _expression. J Interferon Cytokine Res 1997; 17: 121–134.
Seidel HM, Milocco LH, Lamb P, Darnell JE, Jr, Stein RB, Rosen J. Spacing of Palindromic Half Sites as a Determinant of Selective STAT (Signal Transducers and Activators of Transcription) DNA Binding and Transcriptional Activity. PNAS 1995; 92: 3041–3045.
Horvath CM, Wen Z, Darnell JE, Jr. A STAT protein domain that determines DNA sequence recognition suggests a novel DNA-binding domain. Genes & Dev1995; 9: 984–994.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yu, H.M., Zhi, J.L., Cui, Y. et al. Role of the JAK-STAT pathway in protection of hydrogen peroxide preconditioning against apoptosis induced by oxidative stress in PC12 cells. Apoptosis 11, 931–941 (2006). https://doi.org/10.1007/s10495-006-6578-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10495-006-6578-9