Abstract
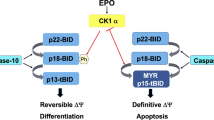
Myelodysplastic syndromes (MDS) constitute a preneoplastic condition in which potentially malignant cancer stem cells continuously die during differentiation. This MDS-associated cell death often involves caspase-3 activation, yet can also occur without caspase activation, for instance in differentiating megakaryocytes (MK). We investigated, the mechanisms through which MK from MDS patients undergo premature cell death. While polyploid, mature MK from healthy subjects or MDS patients manifested caspase-3 activation during terminal differentiation, freshly isolated, immature MK from MDS died without caspase-3 activation. Similarly, purified bone marrow CD34+ cells from MDS patients that were driven into MK differentiation in vitro died without caspase-3 activation at an immature stage, before polyploidization. The premature death of MDS MK was accompanied by the mitochondrial release of cytochrome c, Smac/DIABLO and endonuclease G, a caspase-independent death effector, as well loss of the mitochondrial membrane potential and plasma membrane phosphatidylserine exposure before definitive loss of viability. Thus, a stereotyped pattern of mitochondrial alterations accompanies differentiation-associated MK death in MDS.




Similar content being viewed by others
Abbreviations
- AIF:
-
apoptosis inducing factor
- AML:
-
acute myeloid leukemia
- BMMNC:
-
bone marrow mononuclear cells
- Casp-3a:
-
activated caspase-3
- Cyt c :
-
cytochrome c
- DiOC6(3):
-
3,3′ dihexyloxacarbocyanine iodide
- EndoG:
-
endonuclease G
- Hsp60:
-
heat shock protein 60
- IPSS:
-
international prognostic scoring system
- MDS:
-
myelodysplastic syndrome
- MK:
-
megakaryocytes
- MMP:
-
mitochondrial membrane permeabilization
- PI:
-
propidium iodide
- VWF:
-
Willebrand factor (Factor VIII)
- z-VAD.fmk:
-
N-benzyloxycarbonyl-Val-Ala-Asp- fluoromethylketone
References
Fenaux P (2004) Myelodysplastic syndromes: from pathogenesis and prognosis to treatment. Semin Hematol 41:6–12
Fenaux P (2005) Inhibitors of DNA methylation: beyond myelodysplastic syndromes. Nat Clin Pract Oncol 2 (Suppl 1):S36–44
Mihara K, Chowdhury M, Nakaju N et al (2006) Bmi-1 is useful as a novel molecular marker for predicting progression of myelodysplastic syndrome and patient prognosis. Blood 107:305–308
Kroemer G, Martin SJ (2005) Caspase-independent cell death. Nat Med 11:725–730
Boudard D, Vasselon C, Bertheas MF et al (2002) Expression and prognostic significance of Bcl-2 family proteins in myelodysplastic syndromes. Am J Hematol 70:115–125
Tehranchi R, Fadeel B, Forsblom AM et al (2003) Granulocyte colony-stimulating factor inhibits spontaneous cytochrome c release and mitochondria-dependent apoptosis of myelodysplastic syndrome hematopoietic progenitors. Blood 101:1080–1086
Merchant SH, Gonchoroff NJ, Hutchison RE (2001) Apoptotic index by Annexin V flow cytometry: adjunct to morphologic and cytogenetic diagnosis of myelodysplastic syndromes. Cytometry 46:28–32
Sloand EM, Kim S, Fuhrer M et al (2002) Fas-mediated apoptosis is important in regulating cell replication and death in trisomy 8 hematopoietic cells but not in cells with other cytogenetic abnormalities. Blood 100:4427–4432
Claessens YE, Bouscary D, Dupont JM et al (2002) In vitro proliferation and differentiation of erythroid progenitors from patients with myelodysplastic syndromes: evidence for Fas-dependent apoptosis. Blood 99:1594–1601
Claessens YE, Park S, Dubart-Kupperschmitt A et al (2005) Rescue of early-stage myelodysplastic syndrome-deriving erythroid precursors by the ectopic expression of a dominant-negative form of FADD. Blood 105:4035–4042
Stasi R, Amadori S (2002) Infliximab chimaeric anti-tumour necrosis factor alpha monoclonal antibody treatment for patients with myelodysplastic syndromes. Br J Haematol 116:334–337
Kerbauy DM, Lesnikov V, Abbasi N, Seal S, Scott B, Deeg HJ (2005) NF-kappaB and FLIP in arsenic trioxide (ATO)-induced apoptosis in myelodysplastic syndromes (MDSs). Blood 106:3917–3925
Zang DY, Goodwin RG, Loken MR, Bryant E, Deeg HJ (2001) Expression of tumor necrosis factor-related apoptosis-inducing ligand, Apo2L, and its receptors in myelodysplastic syndrome: effects on in vitro hemopoiesis. Blood 98:3058–3065
Briggs RC, Shults KE, Flye LA et al (2006) Dysregulated human myeloid nuclear differentiation antigen expression in myelodysplastic syndromes: evidence for a role in apoptosis. Cancer Res 66:4645–4651
Braun T, Carvalho G, Fabre C, Grosjean J, Fenaux P, Kroemer G (2006) Targeting NF-kappaB in hematologic malignancies. Cell Death Differ 13:748–758
Clarke MC, Savill J, Jones DB, Noble BS, Brown SB (2003) Compartmentalized megakaryocyte death generates functional platelets committed to caspase-independent death. J Cell Biol 160:577–587
De Botton S, Sabri S, Daugas E et al (2002) Platelet formation is the consequence of caspase activation within megakaryocytes. Blood 100:1310–1317
Houwerzijl EJ, Blom NR, Van Der Want JJ et al (2005) Increased peripheral platelet destruction and caspase-3-independent programmed cell death of bone marrow megakaryocytes in myelodysplastic patients. Blood 105:3472–3479
Houwerzijl EJ, Blom NR, Van Der Want JJ et al (2004) Ultrastructural study shows morphologic features of apoptosis and para-apoptosis in megakaryocytes from patients with idiopathic thrombocytopenic purpura. Blood 103:500–506
Vardiman JW, Harris NL, Brunning RD (2002) The World Health Organization (WHO) classification of the myeloid neoplasms. Blood 100:2292–2302
Greenberg P, Cox C, LeBeau MM et al (1997) International scoring system for evaluating prognosis in myelodysplastic syndromes. Blood 89:2079–2088
Castedo M, Hirsch T, Susin SA et al (1996) Sequential acquisition of mitochondrial and plasma membrane alterations during early lymphocyte apoptosis. J Immunol 157:512–521
Braun T, Carvalho G, Coquelle A et al (2006) NF-kappaB constitutes a potential therapeutic target in high-risk myelodysplastic syndrome. Blood 107:1156–1165
Obeid MTA, Ghiringhelli F et al (2006) Calreticulin exposure dictates the immunogenicity of cancer cell death. Nat Med. In Press
Brada SJ, van de Loosdrecht AA, Koudstaal J, de Wolf JT, Vellenga E (2004) Limited numbers of apoptotic cells in fresh paraffin embedded bone marrow samples of patients with myelodysplastic syndrome. Leuk Res 28:921–925
Kitanaka C, Kuchino Y (1999) Caspase-independent programmed cell death with necrotic morphology. Cell Death Differ 6:508–515
Holler N, Zaru R, Micheau O et al (2000) Fas triggers an alternative, caspase-8-independent cell death pathway using the kinase RIP as effector molecule. Nat Immunol 1:489–495
Jaattela M, Tschopp J (2003) Caspase-independent cell death in T lymphocytes. Nat Immunol 4:416–423
Zong WX, Thompson CB (2006) Necrotic death as a cell fate. Genes Dev 20:1–15
Obeid M, Tesniere A, Ghringhelli F et al (2007) Calreticulin exposure dictates the immunogenicity of cancer cell death. Nat Med 13:54–61
Acknowledgments
This work was supported by Cancéropôle Ile-de-France, Association pour la recherche sur le cancer, Fondation de France, Association Laurette Fugain, European Community (Active p53, TransDeath) (G.K.) and MDS Foundation (T.B.). T.B., J.G., C.F. and S.B. are supported by fellowships from the Etablissement Français du Sang, the Institut National du Cancer, the Fondation pour la Recherche Médicale and Deutsche Forschungsgemeinschaft, respectively.
Author information
Authors and Affiliations
Corresponding author
Additional information
T. Braun and G. Carvalho contributed equally to this paper.
Rights and permissions
About this article
Cite this article
Braun, T., Carvalho, G., Grosjean, J. et al. Differentiating megakaryocytes in myelodysplastic syndromes succumb to mitochondrial derangement without caspase activation. Apoptosis 12, 1101–1108 (2007). https://doi.org/10.1007/s10495-006-0030-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10495-006-0030-z