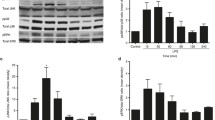
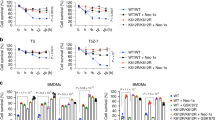
The PI3-kinase/Akt pathway promotes cell survival in many different cell types including intestinal epithelial cells. Increased AKT activation in polyamine depleted intestinal epithelial cells correlated well with the decrease in TNF-α-induced apoptosis. Increased Akt activation and GSK3β (Ser 9) phosphorylation without significant effect on Bad (Ser136) phosphorylation indicate that Akt-mediated protection is independent of Bad phosphorylation but may depend on GSK3β. Pretreatment of polyamine-depleted cells with LY294002 increased caspase-9 and caspase-3 activation and decreased basal levels of GSK-3β phosphorylation. Inhibition of GSK3β activity using AR-A014418 or lithium chloride or siRNA-mediated downregulation of its expression had no effect on apoptosis. Inhibition of PI3-kinase and over-expression of dominant negative Akt (DN-AKT), significantly increased apoptosis in polyamine depleted cells. DN-Akt expression reversed the protective effect of polyamine depletion on apoptosis. DN-Akt, as well as the PI3-kinase inhibitors, prevented Akt activation and subsequent translocation of NF-κB to the nucleus. Constitutively active Akt (CA-AKT) expression increased resistance to TNF-α-induced apoptosis. Constitutively active-Akt expression increased nuclear staining of NF-κB. Moreover, polyamine depletion of DN-Akt cells prevented basal and TNF-α-induced IκBα phosphorylation. Prevention of NF-κB activation in DN-IκBα-transfected cells increased apoptosis in control cells and restored it in polyamine-depleted cells to control levels. These data indicate that Akt regulates the mitochondrial pathway, preventing activation of caspase-9 and thereby caspase-3 via NF-κB and these effects are independent of GSK-3β activity.
Similar content being viewed by others
References
Hengartner MO. The biochemistry of apoptosis. Nature 2000; 407: 770–776.
Meier P, Finch A, Evan G. Apoptosis in development. Nature 2000; 407: 796–801.
Potten CS. The significance of spontaneous and induced apoptosis in the gastrointestinal tract of mice. Cancer Metastasis Rev 1992; 11: 179–195.
Pritchard DM, Watson AJ. Apoptosis and gastrointestinal pharmacology. Pharmacol Ther 1996; 72: 149–169.
Ray RM, Viar MJ, Yuan Q, Johnson LR. Polyamine depletion delays apoptosis of rat intestinal epithelial cells. Am J Physiol Cell Physiol 2000; 278: C480–C489.
Tobias KE, Kahana C. Exposure to ornithine results in excessive accumulation of putrescine and apoptotic cell death in ornithine decarboxylase overproducing mouse myeloma cells. Cell Growth Differ 1995; 10: 1279–1285.
Bhattacharya S, Ray M, Viar MJ, Johnson LR. Polyamines are required for activation of c-Jun NH2-terminal kinase and apoptosis in response to TNF-alpha in IEC-6 cells. Am J Physiol Gastrointest Liver Physiol. 2003; 285: G980–991.
Bhattacharya S, Ray RM, Johnson LR. Prevention of TNF-alpha-induced apoptosis in polyamine-depleted IEC-6 cells is mediated through the activation of ERK1/2. Am J Physiol Gastrointest Liver Physiol 2004; 286: G479–G490.
Deng W, Wang De-An, Gosmanova E, Johnson LR, Tigyi G. LPA protects intestinal epithelial cells from apoptosis by inhibiting the mitochondrial pathway. Am J Physiol Gastrointest Liver Physiol 2003; 284: G821–G829.
Zhang HM, Rao JN, Guo X, et al.. Akt kinase activation blocks apoptosis in intestinal epithelial cells by inhibiting caspase-3 after polyamine depletion. J Biol Chem 2004; 279: 22539–47.
Bellacosa A, Testa J, Staal S, Tsichlis P. A retroviral oncogene, akt, encoding a serine-threonine kinase containing an SH2-like region. Science 1991; 254: 274–277.
Cantley LC. The phosphoinositide 3-kinase pathway. Science 2002; 296: 1655–1657.
Alessi DR, James SR, Downes CP, et al. Characterization of a 3-phosphoinositide-dependent protein kinase which phosphorylates and activates protein kinase B alpha. Curr Biol 1997; 7: 261–269.
Vanhaesebroeck B, Alessi DR. The PI3K-PKD connection: more than just a road tp PKB. Biochem J 2000; 346: 561–576.
Scheid MP, Marignani PA, Wooggett Jr. Multiple phosphoinositide 3-kinase-dependent steps in activation of protein kinase B. Mol Cell Biol 2002; 22: 6247–6260.
Dudek H, Datta SR, Franke TF, et al. Regulation of neuronal survival by the serine-threonine protein kinase Akt. Science 1997; 275: 661–665.
Goswami R, Kilkus J, Dawson SA, Dawson G. Overexpression of Akt (protein kinase B) confers protection against apoptosis and prevents formation of ceramide in response to pro-apoptotic stimuli. J Neurosci Res 1999; 57: 884–893.
Kennedy SG, Wagner AJ, Conzen SD, et al. The PI 3-kinase/Akt signaling pathway delivers an anti-apoptotic signal. Genes Dev 1997; 11: 701–713.
Kulik G, Klippel A, Weber MJ. Antiapoptotic signalling by the insulin-like growth factor I receptor, phosphatidylinositol 3-kinase, and Akt. Mol Cell Biol 1997; 17: 1595–1606.
Lee JW, Juliano RL. alpha5 beta1 integrin protects intestinal epithelial cells from apoptosis through a phosphatidylinositol 3-kinase and protein kinase B-dependent pathway. Mol Biol Cell 2000; 11: 1973–1987.
Fujio Y, Walsh K. Akt mediates cytoprotection of endothelial cells by vascular endothelial growth factor in an anchorage-dependent manner. J Biol Chem 1999; 274: 16349–16354.
Hermann C, Assmus B, Urbich C, Zeiher AM, Dimmeler S. Insulin-mediated stimulation of protein kinase Akt: A potent survival signaling cascade for endothelial cells. Arterioscler Thromb Vasc Biol 2000; 20: 402–409.
Brunet A, Bonni A, Zigmond MJ, et al. Akt promotes cell survival by phosphorylating and inhibiting a Forkhead transcription factor. Cell 1999; 96: 857–868.
Datta SR, Dudek H, Tao X, et al. Akt phosphorylation of BAD couples survival signals to the cell-intrinsic death machinery. Cell 1997; 91: 231–241.
del Peso L, Gonzalez-Garcia M, Page C, Herrera R, Nunez G. Interleukin-3-induced phosphorylation of BAD through the protein kinase Akt. Science 1997; 278: 687–689.
Cardone MH, Roy N, Stennick HR, et al. Regulation of cell death protease caspase-9 by phosphorylation. Science 1998; 282: 1318–1321.
Masuyama N, Oishi K, Mori Y, Ueno T, Takahama Y, Gotoh Y. Akt inhibits the orphan nuclear receptor Nur77 and T-cell apoptosis. J Biol Chem 2001; 276: 32799–32805.
Ozes ON, Mayo LD, Gustin JA, Pfeffer SR, Pfeffer LM, Donner DB. NF-kappaB activation by tumour necrosis factor requires the Akt serine-threonine kinase. Nature 1999; 401: 82–85.
Pap M, Cooper GM. Role of glycogen synthase kinase-3 in the phosphatidylinositol 3-Kinase/Akt cell survival pathway. J Biol Chem 1998; 273: 19929–19932.
Romashkova JA, Makarov SS. NF-kappaB is a target of AKT in anti-apoptotic PDGF signalling. Nature 1999; 401: 86–90.
Kennedy SG, Kandel ES, Cross TK, Hay N. Akt/Protein kinase B inhibits cell death by preventing the release of cytochrome c from mitochondria. Mol Cell Biol 1999; 19: 5800–5810.
Gottlob K, Majewski N, Kennedy S, Kandel E, Robey RB, Hay N. Inhibition of early apoptotic events by Akt/PKB is dependent on the first committed step of glycolysis and mitochondrial hexokinase. Genes Dev 2001; 15: 1406–1418.
Cross DA, Alessi DR, Cohen P, Andjelkovich M, Hemmings, BA. Inhibition of glycogen synthase kinase-3 by insulin mediated by protein kinase B. Nature 1995; 378: 785–789.
Baldwin AS Jr. The NF-kappa B and I kappa B proteins: New discoveries and insights. Annu Rev Immunol 1996; 14: 649–683.
Karin M, Ben-Neriah Y. Phosphorylation meets ubiquitination: The control of NF-[kappa]B activity. Annu Rev Immunol 2000; 16: 621–63.
Bours V, Bentires-Alj M, Hellin AC, et al. Nuclear factor-kappa B, cancer, and apoptosis. Biochem Pharmacol 2000; 60: 1085–1089.
Madrid LV, Wang CY, Guttridge DC, Schottelius AJ, Baldwin AS Jr, Mayo MW. Akt suppresses apoptosis by stimulating the transactivation potential of the RelA/p65 subunit of NF-kappaB. Mol Cell Biol 2000; 20: 1626–1638.
Pfeffer LM, Yang CH, Murti A, et al.. Polyamine depletion induces rapid NF-kappa B activation in IEC-6 cells. J Biol Chem 2001; 276: 45909–45913.
Green DR, Reed JC. Mitochondria and apoptosis. Science 1998; 281: 1309–1312.
Kroemer G, Reed JC. Mitochondrial control of cell death. Nat Med 2000; 6: 513–519.
Wang CY, Guttridge DC, Mayo MW, Baldwin AS, Jr. NF-kappaB induces expression of the Bcl-2 homologue A1/Bfl-1 to preferentially suppress chemotherapy-induced apoptosis. Mol Cell Biol 1999; 19: 5923–5929.
Yu C, Minemoto Y, Zhang J, et al. JNK suppresses apoptosis via phosphorylation of the proapoptotic Bcl-2 family protein BAD. Mol Cell 2004; 13: 329–40.
Zha J, Harada H, Yang E, Jockel J, Korsmeyer SJ. Serine phosphorylation of death agonist BAD in response to survival factor results in binding to 14-3-3 not BCL-X(L). Cell 1996; 87: 619–628.
Zong WX, Edelstein LC, Chen C, Bash J, Gelinas C. The prosurvival Bcl-2 homolog Bfl-1/A1 is a direct transcriptional target of NF-kappaB that blocks TNFalpha-induced apoptosis. Genes Dev 1999; 13: 382–387.
Yuan Q, Ray RM, Johnson LR. Polyamine depletion prevents camptothecin-induced apoptosis by inhibiting the release of cytochrome c. Am J Physiol Cell Physiol 2002; 282: C1290–1297.
Quaroni A, Wands J, Trelstad RL, Isselbacher KJ. Epithelioid cell cultures from rat small intestine. Characterization by morphologic and immunologic criteria. J Cell Biol 1979; 80: 248–265.
Ray RM, Patel A, Viar MJ, et al. RhoA inactivation inhibits cell migration but does not mediate the effects of polyamine depletion. Gastroenterology 2002; 123: 196–205.
Bhat R, Xue Y, Berg S, et al. Structural insights and biological effects of glycogen synthase kinase 3-specific inhibitor AR-A014418. J Biol Chem 2003; 278: 45937–45945.
Juhaszova M, Zoroz DB, Kim SH, et al. Glycogen synthase kinase-3b mediates convergence of protection signaling to inhibit the mitochondrial permeability transition pore. J Clin Invest 2004; 113: 1535–1549.
Zou T, Rao JN, Guo X, et al. NF-kappaB-mediated IAP expression induces resistance of intestinal epithelial cells to apoptosis after polyamine depletion. Am J Physiol Cell Physiol 2004; 286: C1009–18.
Datta SR, Brunet A, Greenberg ME. Cellular survival: A play in three Akts Genes Dev 1999; 13: 2905–2927.
Cardone MH, Roy N, Stennicke HR, et al. Regulation of cell death protease caspase-9 by phosphorylation. Science 1998; 282: 1318–1321.
Kops GJ, Ruiter ND, De Vries-Smits AM, Powell DR, Bos JL, Burgering BM. Direct control of the Forkhead transcription factor AFX by protein kinase B. Nature 1999; 398: 630–634.
Du K, Montminy M. CREB is a regulatory target for the protein kinase Akt/PKB. J Biol Chem 1998; 273: 32377–32379.
Kim AH, Khursigara G, Sun X, Franke TF, Chao MV. Akt phosphorylates and negatively regulates apoptosis signal-regulating kinase 1. Mol Cell Biol 2001; 21: 893–901.
Madrid LV, Mayo MW, Reuther JY, Baldwin AS Jr. Akt stimulates the transactivation potential of the RelA/p65 Subunit of NF-kappa B through utilization of the Ikappa B kinase and activation of the mitogen-activated protein kinase p38. J Biol Chem 2001; 276: 18934–18940.
Yang E, Zha J, Jockel J, Boise LM, Thompson, CB, Korsmeyer, SJ. Bad, a heterodimeric partner for Bcl-XL and Bcl-2, displaces Bax and promotes cell death. Cell 1995; 80: 285–291.
Yuan ZQ, Feldman RI, Sun M, et al. Inhibition of JNK by cellular stress- and tumor necrosis factor alpha-induced AKT2 through activation of the NF kappa B pathway in human epithelial Cells. J Biol Chem 2002; 277: 29973–29982.
Vanhaesebroeck B, Waterfield M. Signaling by distinct classes of phosphoinositide 3-kinases. Exp Cell Res 1999; 253: 239–254.
Schmidt M, Hovelmann S, Beckers TL. A novel form of constitutively active farnesylated Akt1 prevents mammary epithelial cells from anoikis and suppresses chemotherapy-induced apoptosis. British Journal of Cancer 2002; 87: 924–932.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bhattacharya, S., Ray, R.M. & Johnson, L.R. Decreased apoptosis in polyamine depleted IEC-6 cells depends on Akt-mediated NF-κB activation but not GSK3β activity. Apoptosis 10, 759–776 (2005). https://doi.org/10.1007/s10495-005-2943-3
Issue Date:
DOI: https://doi.org/10.1007/s10495-005-2943-3