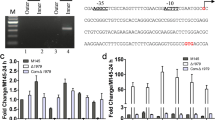
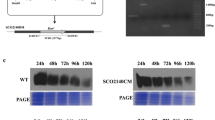
Abstract
The Streptomyces coelicolor genome contains 17 TerD domain-encoding genes (tdd genes) of unknown function. The proteins encoded by these genes have been presumed to be involved in tellurite resistance on the basis of their homology with the protein TerD of Serratia marcescens. To elucidate the role of a Tdd protein (Tdd8), both a deletion mutant for the corresponding gene tdd8 (SCO2368) and a recombinant strain over-expressing tdd8 were produced in S. coelicolor M145. The deletion mutant (Δtdd8), like the wild strain, was not resistant to potassium tellurite. The deletion was not lethal but had a marked effect on differentiation. The deletion strain showed more rapid growth in liquid medium and produced long chains of short spores with a dense and non-spherical spore wall on agar plates. The strain over-expressing tdd8 had a growth delay in liquid medium and produced very few spores of irregular shapes and sizes on solid medium. The results of this study demonstrated that Tdd proteins might have a function other than tellurite resistance and this function seems to be of crucial importance for the proper development of the actinomycete S. coelicolor.





Similar content being viewed by others
References
Benson DA, Boguski MS, Lipman DJ, Ostell J, Ouellette BFF (1998) GenBank. Nucleic Acids Res 26:1–7
Bentley SD, Chater KF, Cerdeno-Tarraga AM et al (2002) Complete genome sequence of the model actinomycete Streptomyces coelicolor A3(2). Nature 417:141–147
Chasteen TG, Fuentes DE, Tantalean JC, Vasquez CC (2009) Tellurite: history, oxidative stress, and molecular mechanisms of resistance. FEMS Microbiol Rev 33:820–832
Claessen D, de Jong W, Dijkhuizen L, Wosten HA (2006) Regulation of Streptomyces development: reach for the sky! Trends Microbiol 14:313–319
Clinical and Laboratory Standards Institute (2006) Methods for dilution antimicrobial susceptibility tests for bacteria that grow aerobicall; approved standard, 7th edn. Clinical and Laboratory Standards Institute, Wayne
Davidsen T, Beck E, Ganapathy A, Montgomery R, Zafar N, Yang Q, Madupu R, Goetz P, Galinsky K, White O, Sutton G (2010) The comprehensive microbial resource. Nucleic Acids Res 38:D340–D345
De Boer E, Heuvelink AE (2000) Methods for the detection and isolation of Shiga toxin-producing Escherichia coli. J Appl Microbiol 88:133S–143S
De Mot R, Schoofs G, Nagy I (2007) Proteome analysis of Streptomyces coelicolor mutants affected in the proteasome system reveals changes in stress-responsive proteins. Arch Microbiol 188:257–271
Dedrick RM, Wildschutte H, McCormick JR (2009) Genetic interactions of smc, ftsK, and parB genes in Streptomyces coelicolor and their developmental genome segregation phenotypes. J Bacteriol 191:320–332
Denis F, Brzezinski R (1992) A versatile shuttle cosmid vector for use in Escherichia coli and actinomycetes. Gene 111:115–118
Fahey RC, Brody S, Mikolajczyk SD (1975) Changes in the glutathione thiol-disulfide status of Neurospora crassa conidia during germination and aging. J Bacteriol 121:144–151
Finn RD, Tate J, Mistry J, Coggill PC, Sammut SJ, Hotz H-R, Ceric G, Forslund K, Eddy SR, Sonnhammer ELL, Bateman A (2008) The comprehensive microbial resource. Nucleic Acids Res 36:D281–D288
Gonzalez-Ceron G, Licona P, Servin-Gonzalez L (2001) Modified xylE and xylTE reporter genes for use in Streptomyces: analysis of the effect of xylT. FEMS Microbiol Lett 196:229–234
Gordon ND, Ottaviano GL, Connell SE, Tobkin GV, Son CH, Shterental S, Gehring AM (2008) Secreted-protein response to σU activity in Streptomyces coelicolor. J Bacteriol 190:894–904
Gust B, Challis GL, Fowler K, Kieser T, Chater KF (2003) PCR-targeted Streptomyces gene replacement identifies a protein domain needed for biosynthesis of the sesquiterpene soil odor geosmin. Proc Natl Acad Sci USA 100:1541–1546
Kanehisa M, Goto S, Hattori M, Aoki-Kinoshita KF, Itoh M, Kawashima S, Katayama T, Araki M, Hirakawa M (2006) From genomics to chemical genomics: new developments in KEGG. Nucleic Acids Res 34:D354–D357
Kay CA, Noce T, Tsang AS (1987) Translocation of an unusual cAMP receptor to the nucleus during development of Dictyostelium discoideum. Proc Natl Acad Sci USA 84:2322–2326
Kieser T, Bibb MJ, Buttner MJ, Chater KF, Hopwood DA (2000) Practical streptomyces genetics. John Innes Foundation, Norwich
Kim DW, Chater K, Lee KJ, Hesketh A (2005) Changes in the extracellular proteome caused by the absence of the bldA gene product, a developmentally significant tRNA, reveal a new target for the pleiotropic regulator AdpA in Streptomyces coelicolor. J Bacteriol 187:2957–2966
Kormutakova R, Klucar L, Turna J (2000) DNA sequence analysis of the tellurite-resistance determinant from clinical strain of Escherichia coli and identification of essential genes. Biometals 13:135–139
Langlois P, Bourassa S, Poirier GG, Beaulieu C (2003) Identification of Streptomyces coelicolor proteins that are differentially expressed in the presence of plant material. Appl Environ Microbiol 69:1884–1889
Lu Y, Wang W, Shu D, Zhang W, Chen L, Qin Z, Yang S, Jiang W (2007) Characterization of a novel two-component regulatory system involved in the regulation of both actinorhodin and a type I polyketide in Streptomyces coelicolor. Appl Microbiol Biotechnol 77:625–635
Manteca A, Mader U, Connolly BA, Sanchez J (2006) A proteomic analysis of Streptomyces coelicolor programmed cell death. Proteomics 6:6008–6022
Manteca A, Claessen D, Lopez-Iglesias C, Sanchez J (2007) Aerial hyphae in surface cultures of Streptomyces lividans and Streptomyces coelicolor originate from viable segments surviving an early programmed cell death event. FEMS Microbiol Lett 274:118–125
Newton GL, Fahey RC (2008) Regulation of mycothiol metabolism by σR and the thiol redox sensor anti-sigma factor RsrA. Mol Microbiol 68:805–809
Newton GL, Arnold K, Price MS, Sherrill C, Delcardayre SB, Aharonowitz Y, Cohen G, Davies J, Fahey RC, Davis C (1996) Distribution of thiols in microorganisms: mycothiol is a major thiol in most actinomycetes. J Bacteriol 178:1990–1995
Novotna J, Vohradsky J, Berndt P, Gramajo H, Langen H, Li XM, Minas W, Orsaria L, Roeder D, Thompson CJ (2003) Proteomic studies of diauxic lag in the differentiating prokaryote Streptomyces coelicolor reveal a regulatory network of stress-induced proteins and central metabolic enzymes. Mol Microbiol 48:1289–1303
Paget MS, Kang JG, Roe JH, Buttner MJ (1998) σR, an RNA polymerase sigma factor that modulates expression of the thioredoxin system in response to oxidative stress in Streptomyces coelicolor A3(2). EMBO J 17:5776–5782
Park JH, Roe JH (2008) Mycothiol regulates and is regulated by a thiol-specific antisigma factor RsrA and σR in Streptomyces coelicolor. Mol Microbiol 68:861–870
Pfaffl MW (2001) A new mathematical model for relative quantification in real-time RT-PCR. Nucleic Acids Res 29:2002–2007
Piette A, Derouaux A, Gerkens P, Noens EE, Mazzucchelli G, Vion S, Koerten HK, Titgemeyer F, De Pauw E, Leprince P, van Wezel GP, Galleni M, Rigali S (2005) From dormant to germinating spores of Streptomyces coelicolor A3(2): new perspectives from the crp null mutant. J Proteome Res 4:1699–1708
Redenbach M, Kieser HM, Denapaite D, Eichner A, Cullum J, Kinashi H, Hopwood DA (1996) A set of ordered cosmids and a detailed genetic and physical map for the 8 Mb Streptomyces coelicolor A3(2) chromosome. Mol Microbiol 21:77–96
Sambrook J, Maniatis T, Fritsch EF (1989) Molecular cloning: a laboratory manual. Cold Spring Harbor Laboratory, Cold Spring Harbor
Setlow B, Setlow P (1977) Levels of acetyl coenzyme A, reduced and oxidized coenzyme A, and coenzyme A in disulfide linkage to protein in dormant and germinated spores and growing and sporulating cells of Bacillus megaterium. J Bacteriol 132:444–452
Taylor DE (1999) Bacterial tellurite resistance. Trends Microbiol 7:111–115
Taylor DE, Rooker M, Keelan M, Ng LK, Martin I, Perna NT, Burland NT, Blattner FR (2002) Genomic variability of O islands encoding tellurite resistance in enterohemorrhagic Escherichia coli O157:H7 isolates. J Bacteriol 184:4690–4698
Touzain F, Schbath S, Debled-Rennesson I, Aigle B, Kucherov G, Leblond P (2008) SIGffRid: a tool to search for sigma factor binding sites in bacterial genomes using comparative approach and biologically driven statistics. BMC Bioinformatics 9:73
Tsang AS, Tasaka M (1986) Identification of multiple cyclic AMP-binding proteins in developing Dictyostelium discoideum cells. J Biol Chem 261:10753–10759
Vohradsky J, Thompson CJ (2006) Systems level analysis of protein synthesis patterns associated with bacterial growth and metabolic transitions. Proteomics 6:785–793
Walter EG, Taylor DE (1992) Plasmid-mediated resistance to tellurite: expressed and cryptic. Plasmid 27:52–64
Whelan KF, Colleran E, Taylor DE (1995) Phage inhibition, colicin resistance, and tellurite resistance are encoded by a single cluster of genes on the IncHI2 plasmid R478. J Bacteriol 177:5016–5027
Wu G, Culley DE, Zhang W (2005) Predicted highly expressed genes in the genomes of Streptomyces coelicolor and Streptomyces avermitilis and the implications for their metabolism. Microbiology 151:2175–2187
Yang YH, Song E, Kim EJ, Lee K, Kim WS, Park SS, Hahn JS, Kim BG (2009) NdgR, an IclR-like regulator involved in amino-acid-dependent growth, quorum sensing, and antibiotic production in Streptomyces coelicolor. Appl Microbiol Biotechnol 82:501–511
Acknowledgments
This work was supported by a grant from the Natural Sciences and Engineering Research Council of Canada (NSERC) to CB. ÉS gratefully acknowledges the receipt of scholarships from NSERC and the Fonds Québécois de la Recherche sur la Nature et les Technologies (FQRNT). The authors thank Yinhua Lu for the gift of plasmid pSET152 m, Karine Robert, Isabelle Madore, Irène Kelsey and Anne-Marie Simao-Beaunoir for technical assistance and Ryszard Brzezinski for critical reading of the manuscript.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Sanssouci, É., Lerat, S., Grondin, G. et al. tdd8: a TerD domain-encoding gene involved in Streptomyces coelicolor differentiation. Antonie van Leeuwenhoek 100, 385–398 (2011). https://doi.org/10.1007/s10482-011-9593-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10482-011-9593-y