Abstract
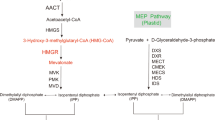
In this study, the gene hmgR encoding the 3-hydroxy-3-methylglutaryl-coenzyme A reductase (HMG-CoA reductase) was cloned and characterized in the zygomycete fungus Rhizomucor miehei. The hmgR gene comprises a total of 3,585 bp including the coding sequence of a 1,058 amino acids length putative protein and five introns (137, 83, 59, 60 and 69 bp in length) dispersed in the whole coding region. Southern hybridization analysis revealed that the gene is present only in one copy in the R. miehei genome. The isolated Rhizomucor gene was expressed in the related fungus, Mucor circinelloides. Transformants harbouring the Rhizomucor hmgR gene in an autoreplicative plasmid proved to be more tolerant to statins (e.g. lovastatin, simvastatin, and fluvastatin), the competitive inhibitors of the HMG-CoA reductase, than the original M. circinelloides strain. At the same time, heterologous expression of the Rhizomucor hmgR did not affect the carotenoid production of M. circinelloides.




Similar content being viewed by others
References
Agarwal B, Hendwal SB, Halmos B, Moss SF, Ramey WG, Holt PR (1999) Lovastatin augments apoptosis induced by chemotherapeutic agents in colon cancer cells. Clin Cancer Res 5:2223–2229
Altschul SF, Madden TL, Schaffer AA, Zhang J, Zhang Z, Miller W, Lipman DJ (1997) Gapped BLAST and PSI-BLAST: a new generation of protein database search programs. Nucleic Acids Res 25:3389–3402
Bejarano ER, Cerda-Olmedo E (1992) Independence of the carotene and sterol pathways of Phycomyces. FEBS J 306:209–212. doi:10.1016/0014-5793(92)81002-4
Benito EP, Díaz-Mínguez JM, Itturiaga EA, Campuzano EA, Eslava AP (1992) Cloning and sequence analysis of the Mucor circinelloides pyrG gene encoding orotidine-5′-monophosphate decarboxylase: use of pyrG for homologous transformation. Gene 116:59–67. doi:10.1016/0378-1119(92)90629-4
Burmester A, Czempinski K (1994) Sequence comparison of a segment of the gene for 3-hydroxy-methylglutaryl-coenzyme A reductase in zygomycetes. Eur J Biochem 220:403–408. doi:10.1111/j.1432-1033.1994.tb18637.x
Chamilos G, Lewis RE, Kontoyiannis DP (2006) Lovastatin has significant activity against zygomycetes and interacts synergistically with voriconazole. Antimicrob Agents Chemother 50:96–103. doi:10.1128/AAC.50.1.96-103.2006
Corrochano LM, Avalos J (1992) Cloning a segment of the gene encoding 3-hydroxy-3-methylglutaryl coenzyme A reductase in Phycomyces blakesleeanus and Gibberella fujikuroi by the polymerase chain reaction. Exp Mycol 16:167–171. doi:10.1016/0147-5975(92)90024-L
Do CB, Mahabhashyam MS, Brudno M, Batzoglou S (2005) PROBCONS: probabilistic consistency-based multiple sequence alignment. Genome Res 15:330–340. doi:10.1101/gr.2821705
Espenshade PJ, Hughes AL (2007) Regulation of sterol synthesis in Eukaryotes. Annu Rev Genet 41:401–427. doi:10.1146/annurev.genet.41.110306.130315
Fu YH, Marzluf GA (1990) Nit-2, the major positive-acting nitrogen regulatory gene of Neurospora crassa, encodes a sequence-specific DNA-binding protein. Proc Natl Acad Sci USA 87:5331–5335. doi:10.1073/pnas.87.14.5331
Galgóczy L, Papp T, Leiter É, Pócsi I, Vágvölgyi Cs (2007) Interactions between statins and Penicillium chrysogenum antifungal protein (PAF) to inhibit the germination of sporangiospores of different sensitive zygomycetes. FEMS Microbiol Lett 270:109–115. doi:10.1111/j.1574-6968.2007.00661.x
Guindon S, Gascuel O (2003) A simple, fast, and accurate algorithm to estimate large phylogenies by maximum likelihood. Syst Biol 52:696–704. doi:10.1080/10635150390235520
Guindon S, Lethiec F, Duroux P, Gascuel O (2005) PHYML online—a web server for fast maximum likelihood-based phylogenetic inference. Nucleic Acids Res 33:W557–W559. doi:10.1093/nar/gki352
Gurr SJ, Unkles SE, Kinghorn JR (1987) The structure and organization of nuclear genes of filamentous fungi. In: Kinghorn JR (ed) Gene structure in eukaryotic microbes. IRL, Oxford, pp 93–139
Hofmann K, Stoffel W (1993) TMbase—a database of membrane spanning proteins segments. Biol Chem Hoppe Seyler 374:166–172
Istvan ES, Palnitkar M, Buchanan SK, Deisenhofer J (2000) Crystal structure of the catalytic portion of human HMG-CoA reductase: insights into regulation of activity and catalysis. EMBO J 19:819–830. doi:10.1093/emboj/19.5.819
Jones DT, Taylor WR, Thornton JM (1992) The rapid generation of mutation data matrices from protein sequences. CABIOS 8:275–282
Katano H, Pesnicak L, Cohen JI (2004) Simvastatin induces apoptosis of eppstein-barr virus (EBV)-transformed lymphoblastoid cell lines and delays development of EBV lymphomas. Proc Natl Acad Sci USA 101:4960–4965. doi:10.1073/pnas.0305149101
Kim SW, Kim MM, Choi HJ, Yoon SS, Lee MH, Park K, Park CH, Kang WK (2001) Phase II study of high dose lovastatin in patients with advanced gastric adenocarcinoma. Invest New Drugs 19:81–83. doi:10.1023/A:1006481423298
Kolar M, Punt PJ, Van den Hondel CA, Schwab H (1988) Transformation of Penicillium chrysogenum using dominant selection markers and expression of an Escherichia coli lacZ fusion gene. Gene 62:127–134. doi:10.1016/0378-1119(88)90586-0
Kudla B, Caddick MX, Langdon T, Martinez-Rossi NM, Bennett CF, Sibley S, Davies RW, Arst HN (1990) The regulatory gene are a mediating nitrogen metabolite repression in Aspergillus nidulans. Mutations affecting specificity of gene activation alter a loop residue of a putative zinc finger. EMBO J 9:1355–1364
Kuwabara PE, Labouesse M (2002) The sterol-sensing domain: multiple families, a unique role? Trends Genet 18:193–201. doi:10.1016/S0168-9525(02)02640-9
Kuzina V, Domenech C, Cerdá-Olmedo E (2006) Relationships among the biosynthesis of ubiquinone, carotene, sterols, and triacylglycerols in zygomycetes. Arch Microbiol 186:485–493. doi:10.1007/s00203-006-0166-9
Lukács Gy, Papp T, Nyilasi I, Nagy E, Vágvölgyi Cs (2004) Differentiation of Rhizomucor species on the basis of their different sensitivities to lovastatin. J Clin Microbiol 42:5400–5402. doi:10.1128/JCM.42.11.5400-5402.2004
Macreadie IG, Johnson G, Schlosser T, Macreadie PI (2006) Growth inhibition of Candida species and Aspergillus fumigatus by statins. FEMS Microbiol Lett 262:9–13. doi:10.1111/j.1574-6968.2006.00370.x
Nagy Á, Vágvölgyi Cs, Balla É, Ferenczy L (1994) Electrophoretic karyotype of Mucor circinelloides. Curr Genet 26:45–48. doi:10.1007/BF00326303
Outtrup H, Boyce COL (1990) Microbial proteinases and biotechnology. In: Fogarty WM, Kelly CT (eds) Microbial enzymes and biotechnology. Elsevier, London, pp 227–254
Pagni M, Ioannidis V, Cerutti L, Zahn-Zabal M, Jongeneel CV, Hau J, Martin O, Kuznetsov D, Falquet L (2007) MyHits: improvements to an interactive resource for analyzing protein sequences. Nucleic Acids Res 35(Web Server issue):W433–W437
Papp T, Velayos A, Bartók T, Eslava AP, Cs Vágvölgyi, Iturriaga EA (2006) Heterologous expression of astaxanthin biosynthesis genes in Mucor circinelloides. Appl Microbiol Biotechnol 69:526–531. doi:10.1007/s00253-005-0026-6
Qiu MS, Pitts AF, Winters TR, Green SH (1991) Ras isoprenilation is required for ras-induced but not for NGF-induced neuronal differentiation of PC12 cells. J Cell Biol 115:795–808. doi:10.1083/jcb.115.3.795
Ribes JA, Vanover-Sams CL, Baker DJ (2000) Zygomycetes in human disease. Clin Microbiol Rev 13:236–301. doi:10.1128/CMR.13.2.236-301.2000
Roze LV, Linz JE (1998) Lovastatin triggers an apoptosis-like cell death process in the fungus Mucor racemosus. Fungal Genet Biol 25:119–133. doi:10.1006/fgbi.1998.1093
Ruiz-Albert J, Cerdá-Olmedo E, Corrochano LM (2002) Genes for mevalonate biosynthesis in Phycomyces. Mol Genet Genomics 266:768–777. doi:10.1007/s004380100565
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning: a laboratory manual, 2nd edn. Cold Spring Harbor Laboratory, Cold Spring Harbor
Tusnády GE, Simon I (2001) The HMMTOP transmembrane topology prediction server. Bioinformatics 17:849–850. doi:10.1093/bioinformatics/17.9.849
van Heeswijck R, Roncero MIG (1984) High frequency transformation of Mucor with recombinant plasmid DNA. Carlsberg Res Commun 49:691–702. doi:10.1007/BF02907500
Vastag M, Kasza Z, Ács K, Papp T, Schwab H, Cs Vágvölgyi (2004) Cloning and sequence analysis of the glyceraldehyde-3-phosphate dehydrogenase gene from the zygomycetes fungus Rhizomucor miehei. Antonie Van Leeuwenhoek 86:111–119. doi:10.1023/B:ANTO.0000036118.41484.e7
Woitek S, Unkles SE, Kinghorn JR, Tudzynski B (1997) 3-Hydroxy-3-methylglutaryl-CoA reductase gene of Gibberella fujikuroi: isolation and characterization. Curr Genet 31:38–47. doi:10.1007/s002940050174
Acknowledgments
The research was supported by ETT grants 214/2006 and 261/2006. Tamás Papp is a grantee of the J. Bolyai Research Scholarship of the Hungarian Academy of Sciences.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lukács, G., Papp, T., Somogyvári, F. et al. Cloning of the Rhizomucor miehei 3-hydroxy-3-methylglutaryl-coenzyme A reductase gene and its heterologous expression in Mucor circinelloides . Antonie van Leeuwenhoek 95, 55–64 (2009). https://doi.org/10.1007/s10482-008-9287-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10482-008-9287-2