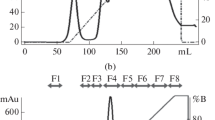
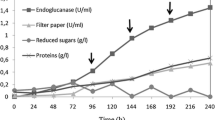
Abstract
A cellulose-degrading fungal strain has been isolated from a rotten rag. Morphological characterization and ITS1, 5.8S and ITS2 rDNA sequencing showed that the strain is a new isolate of Stachybotrys atra. The strain secreted high cellulase activity in media supplemented with rice straw. However, cellulases were not produced in glucose-supplemented media. The crude cellulase showed the highest activity on amorphous celluloses such as carboxymethyl cellulose, while activity on crystalline celluloses such as Avicel was lower. The optimal temperature and pH for CMCase activity were 70°C and pH 5 respectively, although a second peak of activity was found at pH 8. Activity was strongly inhibited by Cu2+, Mn2+ and Hg2+. Analysis by SDS-PAGE, isoelectric focusing and zymography showed that the strain secretes a complex cellulase system comprising several enzymes. Most of these enzymes are alkali-resistant CMCases that remained stable at pH 9 and 65°C for at least 1 h. Cellulose binding assays showed notable differences among the CMCases. While some CMCase bands did not bind Avicel, other bands bound to this polymer and were eluted either with NaCl or by boiling with SDS. Analysis by two-dimensional electrophoresis showed that the band eluted by SDS boiling contained at least 4 different polypeptides. The complex set of cellulases produced by the strain, and their activity and stability at alkaline pH and a high temperature indicate that both the isolated strain and the cellulases identified are good candidates for biotechnological applications involving cellulose modification.






Similar content being viewed by others
References
Abdelmallek AY (1994) Isolation of cellulose-decomposing fungi from damaged manuscripts and documents. Microbiol Res 149:163–165
Altschul S, Madden TL, Schaffer AA, Zhang J, Zhang Z, Miller W, Lipman DJ (1997) Gapped-BLAST and PSI-BLAST: a new generation of protein database search programs. Nucleic Acids Res 25:3389–3402
Andersson MA, Nikulin M, Köljalg U, Andersson MC, Rainey F, Reijula K, Hintikka EL, Salkinoja-Salonen M (1997) Bacteria, molds and toxins in water-damaged building materials. Appl Environ Microbiol 63:387–393
Anish R, Rahman MS, Rao M (2007) Application of cellulases from an alkalothermohilic Thermomonospora sp. in bioplolishing of denims. Biotechnol Bioeng 96:48–56
Archambault C, Coloccia G, Kermasha S, Jabaji-Hare S (1998) Characterization of an endo-1, 3-β-D-glucanase produced during the interaction between the mycoparasite Stachybotrys elegans and its host Rhizoctonia solani. Can J Microbiol 44:989–997
Aro N, Pakula T, Penttilä M (2005) Transcriptional regulation of plant cell wall degradation by filamentous fungi. FEMS Microbiol Rev 29:719–739
Bajpai P, Bajpai PK (1998) Deinking with enzymes: a review. TAPPI J 81(3):111
Béguin P (1990) Molecular biology of cellulose degradation. Annu Rev Microbiol 44:219–248
Bhat MK (2000) Cellulases and related enzymes in biotechnology. Biotechnol Adv 18:355–383
Biely P, Vršanskà M (1988) Xylanase of Cryptococcus albidus. Methods Enzymol 160:638–648
Brasel TL, Douglas DR, Wilson SC, Straus DC (2005) Detection of airborne Stachybotrys chartarum macrocyclic trichothecene mycotoxins on particulates smaller than conidia. Appl Environ Microbiol 71:114–122
Coutinho PM, Henrissat B (1999a) Carbohydrate-active enzymes: an integrated database approach. In: Gilbert HJ, Davies GS, Henrissat B, Svensson B (eds) Recent advances in carbohydrate bioengineering. The Royal Society of Chemistry, Cambridge, pp 3–12
Coutinho PM, Henrissat B (1999b) Carbohydrate active enzymes server, available at http://www.cazy.org/CAZY/fam/acc_GH.html
De Vries RP, Visser J (2001) Aspergillus enzymes involved in degradation of plant cell wall polysaccharides. Microbiol Mol Biol Rev 65:497–522
Foreman PK, Brown D, Dankmeyer L, Dean R, Diener S, Dunn-Coleman NS, Goedegebuur F, Houfek TD, England GJ, Kelley AS, Meerman HJ, Mitchell T, Mitchinson C, Olivares HA, Teunissen PJM, Yao J, Ward M (2003) Transcriptional regulation of biomass-degrading enzymes in the filamentous fungus Trichoderma reesei. J Biol Chem 278:31988–31997
García O, Torres AL, Colom JF, Pastor FIJ, Diaz P, Vidal T (2002) Effect of cellulase-assisted refining on the properties of dried and never-dried eucalyptus pulp. Cellulose 9:115–125
Gilkes NR, Henrissat B, Kilburn DG, Miller RC, Warren RAJ (1991) Domains in microbial β-1, 4-glycanases: sequence conservation, function, and enzyme families. Microbiol Rev 55:303–315
Haugland RA, Heckman JL (1998) Identification of putative sequence specific PCR primers for detection of the toxigenic fungal species Stachybotrys chartarum. Mol Cell Probes 12:387–396
Ilmén M, Saloheimo A, Onnela ML, Penttilä M (1997) Regulation of cellulase gene expression in the filamentous fungus Trichoderma reesei. Appl Environ Microbiol 63:1298–1306
Jarvis BB, Sorenson WG, Hintikka EL, Nikulin M, Zhou Y, Jiang J, Wang S, Hinkley S, Etzel RA, Dearborn D (1998) Study of toxin production by isolates of Stachybotrys chartarum and Memnoniella echinata isolated during a study of pulmonary hemosiderosis in infants. Appl Environ Microbiol 64:3620–3625
Koster B, Scott J, Wong B, Malloch D, Straus N (2003) A geographically diverse set of isolates indicates two phylogenetic lineages within Stachybotrys chartarum. Can J Bot 81:633–643
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685
Lynd LR, Weimer PJ, van Zyl WH, Pretorius IS (2002) Microbial cellulose utilization: fundamentals and biotechnology. Microbiol Mol Biol Rev 66:506–577
Maheshwari R, Bharadwaj G, Bhat MK (2000) Thermophilic fungi: their physiology and enzymes. Microbiol Mol Biol Rev 64:461–488
Mandels M, Weber J (1969) Production of cellulases. Adv Chem Ser 95:391–412
Mander GJ, Wang H, Bodie E, Wagner J, Vienken K, Vinuesa C, Foster C, Leeder AC, Allen G, Hamill V, Janssen GG, Dunn-Coleman N, Karos M, Lemaire HG, Subkowski T, Bollschweiler C, Turner G, Nüsslein B, Fischer F (2006) Use of laccase as a novel, versatile reporter system in filamentous fungi. Appl Environ Microbiol 72:5020–5026
Möller EM, Peltola J (2001) Isolation of high-molecular-weight DNA from mycellium and the recalcitrant and heavily pigmented spores of Stachybotrys chartarum. Anal Biochem 297:99–101
Nikulin M, Pasanen AL, Berg S, Hintikka EL (1994) Stachybotrys atra growth and toxin production in some building materials and fodder under different relative humidities. Appl Environ Microbiol 60:3421–3424
Pastor FIJ, Pujol X, Blanco A, Vidal T, Torres AL, Diaz P (2001) Molecular cloning and characterization of a multidomain endoglucanase from Paenibacillus sp. BP-23: evaluation o fits performance in pulp refining. Appl Microbiol Biotechnol 55:61–68
Quentin M, Ebbelaar M, Derksen J, Mariani C, van der Valk H (2002) Description of a cellulose-binding domain and a linker-sequence from Aspergillus fungi. Appl Microbiol Biotechnol 58:658–662
Saitou N, Nei M (1987) The neighbour-joining method: a new method for reconstructing phylogenetic trees. Mol Bio Evol 4:406–425
Sánchez MM, Irwin D, Pastor FIJ, Wilson DB, Diaz P (2004) Synergistic activity of Paenibacillus sp. BP-23 cellobiohydrolase Cel48C in association with the contiguous endoglucanase Cel9B and with endo- or exo-acting glucanases from Thermobifida fusca. Biotechnol Bioeng 87:161–169
Simonaka A, Koga J, Baba Y, Nishimura T, Murashima K, Kubota H, Kono T (2006) Specific characteristics of family 45 endoglucanases from Mucorales in the use of textiles and laundry. Biosci Biotechnol Biochem 70:1013–1016
Spiro RG (1966) The Nelson-Somogyi copper reduction method. Analysis of sugars found in glycoprotein. Methods Enzymol 8:3–26
Stork G, Pereira H, Wood TM, Düsterhöft EM, Toft A, Puls J (1995) Upgrading recycled pulps using enzymatic treatment. TAPPI J 78(2):79–88
Sunna A, Moracci M, Rossi M, Antranikian G (1997) Glycosyl hydrolases from hyperthermophiles. Extremophiles 1:2–13
Suto M, Tomita F (2001) Induction and catabolite repression mechanisms of cellulase in fungi. J Biosci Bioeng 92:305–311
Thompson JD, Higgins DG, Gibson TJ (1994) CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, positions specific gap penalties and weight matrix choice. Nucleic Acids Res 22:4673–4680
Tomme P, Warren RAJ, Gilkes NR (1995) Cellulose hydrolysis by bacteria and fungi. Adv Microb Physiol 37:1–81
Vieille C, Zeikus GJ (2001) Hyperthermophilic enzymes: sources, uses, and molecular mechanisms for thermostability. Microbiol Mol Biol Rev 65:1–43
Wang T, Liu X, Yu Q, Zhang X, Qu Y, Gao P, Wang T (2005) Directed evolution for engineering pH profile of endoglucanase III from Trichoderma reesei. Biomol Eng 22:89–94
Wood TM (1988) Preparation of crystalline, amorphous, and dyed cellulase substrates. Methods Enzymol 160:19–25
Acknowledgements
This work was partially supported by the Spanish Ministry of Education and Science (CICYT), projects ref. CTQ2004-07560-C02-02 and CTQ2007-68003-CO2-02/PPQ; and the Centre de Referència en Biotecnologia (CeRBa). Pere Picart held an FPI grant ref. FP-2001-1668 from the Spanish Ministry of Science and Technology.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Picart, P., Diaz, P. & Pastor, F.I.J. Stachybotrys atra BP-A produces alkali-resistant and thermostable cellulases. Antonie van Leeuwenhoek 94, 307–316 (2008). https://doi.org/10.1007/s10482-008-9248-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10482-008-9248-9