Abstract
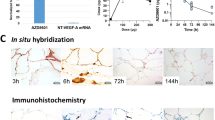
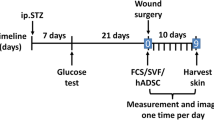
Wounds notoriously accumulate lactate as a consequence of both anaerobic and aerobic glycolysis following microcirculation disruption, immune activation, and increased cell proliferation. Several pieces of evidence suggest that lactate actively participates in the healing process through the activation of several molecular pathways that collectively promote angiogenesis. Lactate indeed stimulates endothelial cell migration and tube formation in vitro, as well as the recruitment of circulating vascular progenitor cells and vascular morphogenesis in vivo. In this study, we examined whether the pro-angiogenic potential of lactate may be exploited therapeutically to accelerate wound healing. We show that lactate delivered from a Matrigel matrix improves reperfusion and opposes muscular atrophy in ischemic hindlimb wounds in mice. Both responses involve lactate-induced reparative angiogenesis. Using microdialysis and enzymatic measurements, we found that, contrary to poly-L-lactide (PLA), a subcutaneous implant of poly-D,L-lactide-co-glycolide (PLGA) allows sustained local and systemic lactate release. PLGA promoted angiogenesis and accelerated the closure of excisional skin wounds in different mouse strains. This polymer is FDA-approved for other applications, emphasizing the possibility of exploiting PLGA therapeutically to improve wound healing.






Similar content being viewed by others
References
Trabold O, Wagner S et al (2003) Lactate and oxygen constitute a fundamental regulatory mechanism in wound healing. Wound Repair Regen 11:504–509
Halestrap AP, Price NT (1999) The proton-linked monocarboxylate transporter (MCT) family: structure, function and regulation. Biochem J 343(Pt 2):281–299
Halestrap AP, Meredith D (2004) The SLC16 gene family-from monocarboxylate transporters (MCTs) to aromatic amino acid transporters and beyond. Pflugers Arch 447:619–628
Schreml S, Szeimies RM et al (2010) Oxygen in acute and chronic wound healing. Br J Dermatol 163:257–268
Vander Heiden MG, Cantley LC, Thompson CB (2009) Understanding the Warburg effect: the metabolic requirements of cell proliferation. Science 324:1029–1033
Hunt TK, Conolly WB, Aronson SB, Goldstein P (1978) Anaerobic metabolism and wound healing: an hypothesis for the initiation and cessation of collagen synthesis in wounds. Am J Surg 135:328–332
Ghani QP, Wagner S, Hussain MZ (2003) Role of ADP-ribosylation in wound repair. The contributions of Thomas K. Hunt, MD. Wound Repair Regen 11:439–444
Gladden LB (2004) Lactate metabolism: a new paradigm for the third millennium. J Physiol 558:5–30
Green H, Goldberg B (1964) Collagen and cell protein synthesis by an established mammalian fibroblast line. Nature 204:347–349
Hussain MZ, Ghani QP, Hunt TK (1989) Inhibition of prolyl hydroxylase by poly(ADP-ribose) and phosphoribosyl-AMP. Possible role of ADP-ribosylation in intracellular prolyl hydroxylase regulation. J Biol Chem 264:7850–7855
Constant JS, Feng JJ et al (2000) Lactate elicits vascular endothelial growth factor from macrophages: a possible alternative to hypoxia. Wound Repair Regen 8:353–360
Xiong M, Elson G, Legarda D, Leibovich SJ (1998) Production of vascular endothelial growth factor by murine macrophages: regulation by hypoxia, lactate, and the inducible nitric oxide synthase pathway. Am J Pathol 153:587–598
Kumar VB, Viji RI, Kiran MS, Sudhakaran PR (2007) Endothelial cell response to lactate: implication of PAR modification of VEGF. J Cell Physiol 211:477–485
Vegran F, Boidot R, Michiels C, Sonveaux P, Feron O (2011) Lactate influx through the endothelial cell monocarboxylate transporter MCT1 supports an NF-kappaB/IL-8 pathway that drives tumor angiogenesis. Cancer Res 71:2550–2560
Sonveaux P, Copetti T et al (2012) Targeting the lactate transporter MCT1 in endothelial cells inhibits lactate-induced HIF-1 activation and tumor angiogenesis. PLoS ONE 7:e33418
Lu H, Forbes RA, Verma A (2002) Hypoxia-inducible factor 1 activation by aerobic glycolysis implicates the Warburg effect in carcinogenesis. J Biol Chem 277:23111–23115
Lu H, Dalgard CL et al (2005) Reversible inactivation of HIF-1 prolyl hydroxylases allows cell metabolism to control basal HIF-1. J Biol Chem 280:41928–41939
Li F, Sonveaux P et al (2007) Regulation of HIF-1alpha stability through S-nitrosylation. Mol Cell 26:63–74
Milovanova TN, Bhopale VM et al (2008) Lactate stimulates vasculogenic stem cells via the thioredoxin system and engages an autocrine activation loop involving hypoxia-inducible factor 1. Mol Cell Biol 28:6248–6261
Murray B, Wilson DJ (2001) A study of metabolites as intermediate effectors in angiogenesis. Angiogenesis 4:71–77
Beckert S, Farrahi F et al (2006) Lactate stimulates endothelial cell migration. Wound Repair Regen 14:321–324
Burns PA, Wilson DJ (2003) Angiogenesis mediated by metabolites is dependent on vascular endothelial growth factor (VEGF). Angiogenesis 6:73–77
Hunt TK, Aslam RS et al (2007) Aerobically derived lactate stimulates revascularization and tissue repair via redox mechanisms. Antioxid Redox Signal 9:1115–1124
Couffinhal T, Silver M et al (1998) Mouse model of angiogenesis. Am J Pathol 152:1667–1679
Sonveaux P, Martinive P et al (2004) Caveolin-1 expression is critical for vascular endothelial growth factor-induced ischemic hindlimb collateralization and nitric oxide-mediated angiogenesis. Circ Res 95:154–161
Del Prete E, Lutz TA, Scharrer E (2004) Inhibition of glucose oxidation by alpha-cyano-4-hydroxycinnamic acid stimulates feeding in rats. Physiol Behav 80:489–498
Sonveaux P, Vegran F et al (2008) Targeting lactate-fueled respiration selectively kills hypoxic tumor cells in mice. J Clin Invest 118:3930–3942
Hishiya A, Iemura S et al (2006) A novel ubiquitin-binding protein ZNF216 functioning in muscle atrophy. EMBO J 25:554–564
Dehoux M, Van Beneden R et al (2004) Role of the insulin-like growth factor I decline in the induction of atrogin-1/MAFbx during fasting and diabetes. Endocrinology 145:4806–4812
Cao Y, Sonveaux P et al (2007) Systemic overexpression of angiopoietin-2 promotes tumor microvessel regression and inhibits angiogenesis and tumor growth. Cancer Res 67:3835–3844
Peters T, Sindrilaru A et al (2005) Wound-healing defect of CD18(-/-) mice due to a decrease in TGF-beta1 and myofibroblast differentiation. EMBO J 24:3400–3410
Gutierrez-Fernandez A, Inada M et al (2007) Increased inflammation delays wound healing in mice deficient in collagenase-2 (MMP-8). FASEB J 21:2580–2591
Sindrilaru A, Peters T et al (2009) Wound healing defect of Vav3-/- mice due to impaired {beta}2-integrin-dependent macrophage phagocytosis of apoptotic neutrophils. Blood 113:5266–5276
Milch HS, Schubert SY, Hammond S, Spiegel JH (2010) Enhancement of ischemic wound healing by inducement of local angiogenesis. Laryngoscope 120:1744–1748
Mendel DB, Schreck RE et al (2000) The angiogenesis inhibitor SU5416 has long-lasting effects on vascular endothelial growth factor receptor phosphorylation and function. Clin Cancer Res 6:4848–4858
Manning Fox JE, Meredith D, Halestrap AP (2000) Characterisation of human monocarboxylate transporter 4 substantiates its role in lactic acid efflux from skeletal muscle. J Physiol 529(Pt 2):285–293
Stabile E, Kinnaird T et al (2006) CD8 + T lymphocytes regulate the arteriogenic response to ischemia by infiltrating the site of collateral vessel development and recruiting CD4 + mononuclear cells through the expression of interleukin-16. Circulation 113:118–124
Koponen JK, Kekarainen T et al (2007) Umbilical cord blood-derived progenitor cells enhance muscle regeneration in mouse hindlimb ischemia model. Mol Ther 15:2172–2177
Bonen A (2001) The expression of lactate transporters (MCT1 and MCT4) in heart and muscle. Eur J Appl Physiol 86:6–11
Dedkov EI, Kostrominova TY, Borisov AB, Carlson BM (2002) Resistance vessel remodeling and reparative angiogenesis in the microcirculatory bed of long-term denervated skeletal muscles. Microvasc Res 63:96–114
Kawai K, Larson BJ et al (2011) Calcium-based nanoparticles accelerate skin wound healing. PLoS ONE 6:e27106
Mendel DB, Laird AD et al (2000) Development of SU5416, a selective small molecule inhibitor of VEGF receptor tyrosine kinase activity, as an anti-angiogenesis agent. Anticancer Drug Des 15:29–41
Menke NB, Ward KR, Witten TM, Bonchev DG, Diegelmann RF (2007) Impaired wound healing. Clin Dermatol 25:19–25
Creager MA, Loscalzo J (2008) Diseases of the extremities. In: Fauci AS, Braunwald E, Kasper DL (eds) Harrison’s principles of internal medicine, 17th edn. McGraw Hill, New-York, pp 1568–1570
Dhup S, Dadhich RK, Porporato PE, Sonveaux P (2012) Multiple biological activities of lactic acid in cancer: influences on tumor growth, angiogenesis and metastasis. Curr Pharm Des 18:1319–1330
Dimmer KS, Friedrich B, Lang F, Deitmer JW, Broer S (2000) The low-affinity monocarboxylate transporter MCT4 is adapted to the export of lactate in highly glycolytic cells. Biochem J 350(Pt 1):219–227
Hashimoto T, Hussien R, Oommen S, Gohil K, Brooks GA (2007) Lactate sensitive transcription factor network in L6 cells: activation of MCT1 and mitochondrial biogenesis. FASEB J 21:2602–2612
Porporato PE, Dadhich RK, Dhup S, Copetti T, Sonveaux P (2011) Anticancer targets in the glycolytic metabolism of tumors: a comprehensive review. Front Pharmacol 2:49
Folkman J (1995) Angiogenesis in cancer, vascular, rheumatoid and other disease. Nat Med 1:27–31
Dorrell MI, Aguilar E, Scheppke L, Barnett FH, Friedlander M (2007) Combination angiostatic therapy completely inhibits ocular and tumor angiogenesis. Proc Natl Acad Sci USA 104:967–972
Chang X, Wei C (2011) Glycolysis and rheumatoid arthritis. Int J Rheum Dis 14:217–222
Barba I, Garcia-Ramirez M et al (2010) Metabolic fingerprints of proliferative diabetic retinopathy: an 1H-NMR-based metabonomic approach using vitreous humor. Invest Ophthalmol Vis Sci 51:4416–4421
Shanmugasundaram M, Ram VK, Luft UC, Szerlip M, Alpert JS (2011) Peripheral arterial disease–what do we need to know? Clin Cardiol 34:478–482
Enerson BE, Drewes LR (2003) Molecular features, regulation, and function of monocarboxylate transporters: implications for drug delivery. J Pharm Sci 92:1531–1544
Cori CF, Cori GT (1929) Glycogen formation in the liver with d- and l-lactic acid. J Biol Chem 81:389–403
Patel MS, Jomain-Baum M, Ballard FJ, Hanson RW (1971) Pathway of carbon flow during fatty acid synthesis from lactate and pyruvate in rat adipose tissue. J Lipid Res 12:179–191
Pagliassotti MJ, Donovan CM (1990) Role of cell type in net lactate removal by skeletal muscle. Am J Physiol 258:E635–E642
Fredenberg S, Wahlgren M, Reslow M, Axelsson A (2011) The mechanisms of drug release in poly(lactic-co-glycolic acid)-based drug delivery systems–a review. Int J Pharm 415:34–52
Acknowledgments
This work was supported by grants from the European Research Council (FP7/2007-2013 ERC Independent Researcher Starting Grant No. 243188 TUMETABO to P.S.), an Action de Recherche Concertée from the Communauté Française de Belgique (to P.S. and O.F.), the Fondation Belge contre le Cancer (200-2008 to P.S. and O.F.), the Fonds National de la Recherche Scientifique Médicale (FRSM), the Association Française contre les Myopathies (AFM), and the Fonds National de la Recherche Scientifique (F.R.S.-FNRS). P.S. is a Research Associate and O.F. an honorary Research Director of the F.R.S.-FNRS. C.D.S. is a Télévie Research Fellow. The authors thank Elise Beneteau, Morgane Tardy and Claire Ploquin for excellent technical assistance, and Pharmacists at Pharmacie Digitale (Kraainem) for providing the cetomacrogol cream. Dedicated to the loving memory of Mrs Anne-Sophie Brouckaert, PharmD.
Ethics statement
All the procedures described in this study and pertaining to the use and care of small laboratory animals have been approved by UCL authorities according to Belgian National Animal Care Regulations.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Porporato, P.E., Payen, V.L., De Saedeleer, C.J. et al. Lactate stimulates angiogenesis and accelerates the healing of superficial and ischemic wounds in mice. Angiogenesis 15, 581–592 (2012). https://doi.org/10.1007/s10456-012-9282-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10456-012-9282-0