Abstract
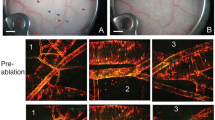
Laser speckle contrast imaging (LSCI) is a high-resolution and high contrast optical imaging technique often used to characterize hemodynamic changes in short-term physiological experiments. In this study, we demonstrate the utility of LSCI for characterizing microvascular remodeling and hemodynamic changes during wound healing angiogenesis in vivo. A 2 mm diameter hole was made in the mouse ear and the periphery of the wound imaged in vivo using LSCI over 12 days. We were able to visualize and quantify the vascular and perfusion changes that accompanied wound healing in the microenvironment proximal to the wound, and validated these changes with histology. We found that consistent with the stages of wound healing, microvessel density increased during the initial inflammatory phase (i.e., day 0–3), stayed elevated through the tissue formation phase (i.e., until day 7) and returned to baseline during the tissue remodeling phase (i.e., by day 12). Concomitant “wide area mapping” of blood flow revealed that tissue perfusion in the wound periphery initially decreased, gradually increased from day 3–7, and subsided as healing completed. Interestingly, some regions exhibited a reestablishment of tissue perfusion approximately 6 days earlier than the ~18 days usually reported for the long term remodeling phase. The results from this study demonstrate that LSCI is an ideal platform for elucidating in vivo changes in microvascular hemodynamics and angiogenesis, and has the potential to offer invaluable insights in a range of disease models involving abnormal hemodynamics, such as diabetes and tumors.





Similar content being viewed by others
References
Singer AJ, Clark RAF (1999) Cutaneous wound healing. New Eng J Med 341:738–746
Kiyama T, Naito M, Shitama H, Shinoda T, Maeyama A (2008) Comparison of skin blood flow between mini- and standard-incision approaches during total hip arthroplasty. J Arthroplast 23:1045–1049
Koskela M, Gaddnas F, Ala-Kokko TI, Laurila JJ, Saarnio J et al (2009) Epidermal wound healing in severe sepsis and septic shock in humans. Crit Care 13:R100
Ngo BT, Hayes KD, DiMiao DJ, Srinivasan SK, Huerter CJ et al (2005) Manifestations of cutaneous diabetic microangiopathy. Am J Clin Dermatol 6:225–237
Chung JH, Eun HC (2007) Angiogenesis in skin aging and photoaging. J Dermatol 34:593–600
Sorg H, Krueger C, Vollmar B (2007) Intravital insights in skin wound healing using the mouse dorsal skin fold chamber. J Anat 211:810–818
Demirdogen B, Elcin AE, Elcin YM (2010) Neovascularization by bFGF releasing hyaluronic acid-gelatin microspheres: in vitro and in vivo studies. Growth Factors 28:426–436
Rendell MS, Johnson ML, Smith D, Finney D, Capp C et al (2002) Skin blood flow response in the rat model of wound healing: expression of vasoactive factors. J Surg Res 107:18–26
Shaterian A, Borboa A, Sawada R, Costantini T, Potenza B et al (2009) Real-time analysis of the kinetics of angiogenesis and vascular permeability in an animal model of wound healing. Burns 35:811–817
Rendell MS, Milliken BK, Finnegan MF, Finney DE, Healy JC et al (1998) The microvascular composition of the healing wound compared at skin sites with nutritive versus arteriovenous perfusion. J Surg Res 80:373–379
North KA, Sanders AG (1958) The development of collateral circulation in the mouse’s ear. Circ Res 6:721–727
Rendell MS, Milliken BK, Finnegan MF, Finney DA, Healy JC (1997) The skin blood flow response in wound healing. Microvasc Res 53:222–234
Murari K, Li N, Rege A, Jia X, All A et al (2007) Contrast-enhanced imaging of cerebral vasculature with laser speckle. Appl Opt 46:5340–5346
Dunn AK, Bolay H, Moskowitz MA, Boas DA (2001) Dynamic imaging of cerebral blood flow using laser speckle. J Cereb Blood Flow Metab 21:195–201
Ayata C, Dunn AK, Gursoy OY, Huang Z, Boas DA et al (2004) Laser speckle flowmetry for the study of cerebrovascular physiology in normal and ischemic mouse cortex. J Cereb Blood Flow Metab 24:744–755
Nakagami G, Sari Y, Nagase T, Iizaka S, Ohta Y, et al. (2010) Evaluation of the usefulness of skin blood flow measurements by laser speckle flowgraphy in pressure-induced ischemic wounds in rats. Ann Plast Surg 64:351–354
Stewart CJ, Gallant-Behm CL, Forrester K, Tulip J, Hart DA et al (2006) Kinetics of blood flow during healing of excisional full-thickness skin wounds in pigs as monitored by laser speckle perfusion imaging. Skin Res Technol 12:247–253
Davidson JM (1998) Animal models for wound repair. Arch Dermatol Res 290(Suppl):S1–S11
Barker JH, Kjolseth D, Kim M, Frank J, Bondar I et al (1994) The hairless mouse ear: an in vivo model for studying wound neovascularization. Wound Repair Regen 2:138–143
Ramirez-San-Juan JC, Ramos-García R, Guizar-Iturbide I, Martínez-Niconoff G, Choi B (2008) Impact of velocity distribution assumption on simplified laser speckle imaging equation. Opt Express 16:3197–3203
Bandyopadhyay R, Gittings A, Suh S, Dixon P, Durian D (2005) Speckle-visibility spectroscopy: a tool to study time-varying dynamics. Rev Sci Instrum 76:0931101–0931111
Duncan DD, Kirkpatrick SJ (2008) Can laser speckle flowmetry be made a quantitative tool? J Opt Soc Am A Opt Image Sci Vis 25:2088–2094
Rege A, Murari K, Li N, Thakor NV (2010) Imaging microvascular flow characteristics using laser speckle contrast imaging. Proc 32nd Ann Intl Conf IEEE Engr Med Biol Soc, Buenos Aires, pp 1978–1981. http://www.ncbi.nlm.nih.gov/pubmed/21096787
Barber DC (1992) Registration of low resolution medical images. Phys Med Biol 37:1485–1498
Miao P, Li N, Rege A, Tong S, Thakor NV (2009) Model based reconstruction for simultaneously imaging cerebral blood flow and deoxyhemoglobin distribution. Proc 31st Ann Intl Conf IEEE Engr Med Biol Soc, Minneapolis, pp 3236–3293. http://www.ncbi.nlm.nih.gov/pubmed/19964288
Patasius M, Marozas V, Lukosevicius A, Jegelevicius D (2007) Model based investigation of retinal vessel tortuosity as a function of blood pressure: preliminary results. Proc 29th Ann Intl Conf IEEE Engr Med Biol Soc, Lyon, pp 6459–6462. http://www.ncbi.nlm.nih.gov/pubmed/18003504
Hart WE, Goldbaum M, Côté B, Kube P, Nelson MR (1999) Measurement and classification of retinal vascular tortuosity. Int J Med Inform 53:239–252
Davis GE, Senger DR (2005) Endothelial extracellular matrix: biosynthesis, remodeling, and functions during vascular morphogenesis and neovessel stabilization. Circ Res 97:1093–1107
Simon-Assmann P, Orend G, Mammadova-Bach E, Spenle C, Lefebvre O (2011) Role of laminins in physiological and pathological angiogenesis. Int J Dev Biol 55:455–465
Pettersson A, Nagy JA, Brown LF, Sundberg C, Morgan E et al (2000) Heterogeneity of the angiogenic response induced in different normal adult tissues by vascular permeability factor/vascular endothelial growth factor. Lab Invest 80:99–115
Bondar I, Uhl E, Barker JH, Galla TJ, Hammersen F et al (1991) A new model for studying microcirculatory changes during dermal wound healing. Res Exp Med (Berl) 191:379–388
Fukumura DAI, Jain RK (2008) Imaging angiogenesis and the microenvironment. APMIS 116:695–715
Hu S, Wang LV (2010) Photoacoustic imaging and characterization of the microvasculature. J Biomed Opt 15:011101
Vakoc BJ, Lanning RM, Tyrrell JA, Padera TP, Bartlett LA et al (2009) Three-dimensional microscopy of the tumor microenvironment in vivo using optical frequency domain imaging. Nat Med 15:1219–1223
Cho CH, Sung HK, Kim KT, Cheon HG, Oh GT et al (2006) COMP-angiopoietin-1 promotes wound healing through enhanced angiogenesis, lymphangiogenesis, and blood flow in a diabetic mouse model. Proc Natl Acad Sci USA 103:4946–4951
Ambrozy E, Waczulikova I, Willfort-Ehringer A, Ehringer H, Koppensteiner R et al (2009) Microcirculation in mixed arterial/venous ulcers and the surrounding skin: clinical study using a laser Doppler perfusion imager and capillary microscopy. Wound Repair Regen 17:19–24
Dunn AK, Devor A, Bolay H, Andermann ML, Moskowitz MA et al (2003) Simultaneous imaging of total cerebral hemoglobin concentration, oxygenation, and blood flow during functional activation. Opt Lett 28:28–30
Werner S, Grose R (2003) Regulation of wound healing by growth factors and cytokines. Physiol Rev 83:835–870
Eming SA, Brachvogel B, Odorisio T, Koch M (2007) Regulation of angiogenesis: wound healing as a model. Prog Histochem Cytochem 42:115–170
Roesken F, Uhl E, Curri SB, Menger MD, Messmer K (2000) Acceleration of wound healing by topical drug delivery via liposomes. Langenbecks Arch Surg 385:42–49
Vollmar B, El-Gibaly AM, Scheuer C, Strik MW, Bruch HP et al (2002) Acceleration of cutaneous wound healing by transient p53 inhibition. Lab Invest 82:1063–1071
Schafer M, Werner S (2008) Cancer as an overhealing wound: an old hypothesis revisited. Natl Rev Mol Cell Biol 9:628–638
Nagy JA, Chang SH, Shih SC, Dvorak AM, Dvorak HF (2010) Heterogeneity of the tumor vasculature. Semin Thromb Hemost 36:321–331
Escamez MJ, Garcia M, Larcher F, Meana A, Munoz E et al (2004) An in vivo model of wound healing in genetically modified skin-humanized mice. J Invest Dermatol 123:1182–1191
Apikoglu-Rabus S, Izzettin FV, Turan P, Ercan F (2009) Effect of topical insulin on cutaneous wound healing in rats with or without acute diabetes. Clin Exp Dermatol 35:180–185
Tian J, Wong KKY, Ho C-M, Lok C-N, Yu W-Y et al (2007) Topical delivery of silver nanoparticles promotes wound healing. Chem Med Chem 2:129–136
Bao P, Kodra A, Tomic-Canic M, Golinko MS, Ehrlich HP et al (2009) The role of vascular endothelial growth factor in wound healing. J Surg Res 153:347–358
Wu Y, Chen L, Scott PG, Tredget EE (2007) Mesenchymal stem cells enhance wound healing through differentiation and angiogenesis. Stem Cells 25:2648–2659
Dvorak HF (1986) Tumors: wounds that do not heal. Similarities between tumor stroma generation and wound healing. N Engl J Med 315:1650–1659
Jain RK (2005) Normalization of tumor vasculature: an emerging concept in antiangiogenic therapy. Science 307:58–62
Dewhirst MW, Richardson R, Cardenas-Navia I, Cao Y (2004) The relationship between the tumor physiologic microenvironment and angiogenesis. Hematol Oncol Clin N Am 18:973–990
Bullitt E, Zeng D, Gerig G, Aylward S, Joshi S et al (2005) Vessel tortuosity and brain tumor malignancy: a blinded study1. Acad Radiol 12:1232–1240
Acknowledgments
This work was supported jointly by National Institute of Aging award number R01AG029681, Department of Health and Human Services award number 1R43CA139983-01, and a JHU Institute of NanoBiotechnology (INBT) Junior Faculty Pilot Award. We gratefully acknowledge Peng Miao for assistance with image registration.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Rege, A., Thakor, N.V., Rhie, K. et al. In vivo laser speckle imaging reveals microvascular remodeling and hemodynamic changes during wound healing angiogenesis. Angiogenesis 15, 87–98 (2012). https://doi.org/10.1007/s10456-011-9245-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10456-011-9245-x