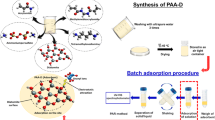
Abstract
Pollution during rapid industrialization process is a serious environmental problem that seriously affects people's lives and livelihoods. Among them, the pollution of excessive heavy metals in wastewater has attracted wide attention. The current study proposes a novel effective and low-cost silica@polyacrylamide composites (SP-C) to capture cadmium ion (Cd2+) from wastewater contaminated by heavy metal. The chemical and physical properties of the surface and interior of silica@polyacrylamide composites were studied by different characterization methods. The adsorption capacity and kinetics of cadmium ions in aqueous solutions using silica@polyacrylamide composites were also studied, and some discussions about the mechanism of adsorption process were made. Silica@polyacrylamide composites showed enhanced cadmium ions adsorption with the maximum adsorption capacity of 211.86 mg/g (pH 6.5, T = 318.15 K), which was fitted by Langmuir model. The adsorption experimental kinetic data of silica@polyacrylamide composites could be excellently verified by pseudo-second-order model. Moreover, a comparison of the cost between the proposed silica@polyacrylamide composites and other reported adsorbents is provided. And the results indicate that the silica@polyacrylamide composites adsorption cost is 0.16 $/g cadmium ion, which means a promising cost-effective adsorbent for cadmium ion removing.








Similar content being viewed by others
References
Alothman, Z.A., Alam, M.M., Naushad, M.: Heavy toxic metal ion exchange kinetics: validation of ion exchange process on composite cation exchanger nylon 6,6 Zr(IV) phosphate. J. Ind. Eng. Chem. 19(3), 956–960 (2013). https://doi.org/10.1016/j.jiec.2012.11.016
Awual, M.R., Khraisheh, M., Alharthi, N.H., Luqman, M., Islam, A., Karim, M.R., Rahman, M.M., Khaleque, M.A.: Efficient detection and adsorption of cadmium(II) ions using innovative nano-composite materials. Chem. Eng. J. 343, 118–127 (2018). https://doi.org/10.1016/j.cej.2018.02.116
Baccar, R., Bouzid, J., Feki, M., Montiel, A.: Preparation of activated carbon from Tunisian olive-waste cakes and its application for adsorption of heavy metal ions. J. Hazard. Mater. 162(2–3), 1522–1529 (2009). https://doi.org/10.1016/j.jhazmat.2008.06.041
Bamgbose, J.T., Adewuyi, S., Bamgbose, O., Adetoye, A.A.: Adsorption kinetics of cadmium and lead by chitosan. Afr. J. Biotech. 9(17), 2560–2565 (2010). https://doi.org/10.1186/1471-2164-11-267
Bessbousse, H., Rhlalou, T., Verchère, J.F., Lebrun, L.: Removal of heavy metal ions from aqueous solutions by filtration with a novel complexing membrane containing poly(ethyleneimine) in a poly(vinyl alcohol) matrix. J. Membr. Sci. 307(2), 249–259 (2008). https://doi.org/10.1016/j.memsci.2007.09.027
Cegłowski, M., Schroeder, G.: Preparation of porous resin with Schiff base chelating groups for removal of heavy metal ions from aqueous solutions. Chem. Eng. J. 263, 402–411 (2015). https://doi.org/10.1016/j.cej.2014.11.047
Chandra, R., Yadav, S., Yadav, S.: Phytoextraction potential of heavy metals by native wetland plants growing on chlorolignin containing sludge of pulp and paper industry. Ecol. Eng. 98, 134–145 (2017). https://doi.org/10.1016/j.ecoleng.2016.10.017
Chen, C.Y., Chiang, C.L., Huang, P.C.: Adsorptions of heavy metal ions by a magnetic chelating resin containing hydroxy and iminodiacetate groups. Sep. Purif. Technol. 50(1), 15–21 (2006). https://doi.org/10.1016/j.seppur.2005.11.002
Chen, J., Wang, Y., Ding, S., Ding, J., Li, M., Zhang, C., Zou, M.: Sub- and super-critical water oxidation of wastewater containing organic and heavy metallic pollutants and recovery of superfine metallic particles. J. Environ. Chem. Eng. 4(3), 2698–2705 (2016). https://doi.org/10.1016/j.jece.2016.05.014
Chen, W., Lu, Z., Xiao, B., Gu, P., Yao, W., Xing, J., Asiri, A.M., Alamry, K.A., Wang, X., Wang, S.: Enhanced removal of lead ions from aqueous solution by iron oxide nanomaterials with cobalt and nickel doping. J. Clean. Prod. 211, 1250–1258 (2019). https://doi.org/10.1016/j.jclepro.2018.11.254
Foo, K.Y., Hameed, B.H.: Insights into the modeling of adsorption isotherm systems. Chem. Eng. J. 156(1), 2–10 (2010). https://doi.org/10.1016/j.cej.2009.09.013
Fu, F., Wang, Q.: Removal of heavy metal ions from wastewaters: a review. J. Environ. Manag. 92(3), 407 (2011). https://doi.org/10.1016/j.jenvman.2010.11.011
Gök, Ö., Özcan, A., Erdem, B., Özcan, A.S.: Prediction of the kinetics, equilibrium and thermodynamic parameters of adsorption of copper(II) ions onto 8-hydroxy quinoline immobilized bentonite. Colloids Surf. A 317(1), 174–185 (2008). https://doi.org/10.1016/j.colsurfa.2007.10.009
Gu, P., Zhang, S., Zhang, C., Wang, X., Khan, A., Wen, T., Hu, B., Alsaedi, A., Hayat, T., Wang, X.: Two-dimensional MAX-derived titanate nanostructures for efficient removal of Pb(II). Dalton Trans. (2019). https://doi.org/10.1039/C8DT04301A
Gurgel, L.V.A., Laurent Frédéric, G.: Adsorption of Cu(II), Cd(II) and Pb(II) from aqueous single metal solutions by succinylated twice-mercerized sugarcane bagasse functionalized with triethylenetetramine. Water Res. 43(18), 4479–4488 (2009). https://doi.org/10.1016/j.watres.2009.07.017
Hizal, J., Apak, R.: Modeling of cadmium(II) adsorption on kaolinite-based clays in the absence and presence of humic acid. Appl. Clay Sci. 32(3–4), 232–244 (2006). https://doi.org/10.1016/j.clay.2006.02.002
Ho, Y.-S.: Citation review of Lagergren kinetic rate equation on adsorption reactions. Scientometrics 59(1), 171–177 (2004). https://doi.org/10.1023/B:SCIE.0000013305.99473.cf
Ho, Y.-S.: Review of second-order models for adsorption systems. J. Hazard. Mater. 136(3), 681–689 (2006). https://doi.org/10.1016/j.jhazmat.2005.12.043
Hu, X.J., Wang, J.S., Liu, Y.G., Li, X., Zeng, G.M., Bao, Z.L., Zeng, X.X., Chen, A.W., Long, F.: Adsorption of chromium (VI) by ethylenediamine-modified cross-linked magnetic chitosan resin: isotherms, kinetics and thermodynamics. J. Hazard. Mater. 185(1), 306–314 (2011). https://doi.org/10.1016/j.jhazmat.2010.09.034
Hu, X., Mangano, E., Friedrich, D., Ahn, H., Brandani, S.: Diffusion mechanism of CO2 in 13X zeolite beads. Adsorption 20(1), 121–135 (2014). https://doi.org/10.1007/s10450-013-9554-z
Hu, X., Liu, L., Luo, X., Xiao, G., Shiko, E., Zhang, R., Fan, X., Zhou, Y., Liu, Y., Zeng, Z., Li, C.E.: A review of N-functionalized solid adsorbents for post-combustion CO2 capture. Appl. Energy 260, 114244 (2020). https://doi.org/10.1016/j.apenergy.2019.114244
Jia, J., Liu, C., Wang, L., Liang, X., Chai, X.: Double functional polymer brush-grafted cotton fiber for the fast visual detection and efficient adsorption of cadmium ions. Chem. Eng. J. 347, 631–639 (2018). https://doi.org/10.1016/j.cej.2018.04.152
Karnitz, O. Jr., Gurgel, L.V.A., Melo, J.C.P.D., Botaro, V.R., Melo, T.M.S., Gil, L.F.: Adsorption of heavy metal ion from aqueous single metal solution by chemically modified sugarcane bagasse. Biores. Technol. 98(6), 1291–1297 (2007). https://doi.org/10.1016/j.biortech.2006.05.013
Li, M., Li, M.Y., Feng, C.G., Zeng, Q.X.: Preparation and characterization of multi-carboxyl-functionalized silica gel for removal of Cu (II), Cd (II), Ni (II) and Zn (II) from aqueous solution. Appl. Surf. Sci. 314(314), 1063–1069 (2014). https://doi.org/10.1016/j.apsusc.2014.06.038
Li, X., Zhou, H., Wu, W., Wei, S., Xu, Y., Kuang, Y.: Studies of heavy metal ion adsorption on chitosan/sulfydryl-functionalized graphene oxide composites. J. Colloid Interface Sci. 448, 389–397 (2015a). https://doi.org/10.1016/j.jcis.2015.02.039
Li, Z., Yan, K., Ge, Y.: Synthesis of porous lignin xanthate resin for Pb2+ removal from aqueous solution. Chem. Eng. J. 270, 229–234 (2015b). https://doi.org/10.1016/j.cej.2015.01.123
Li, G., Wang, B., Sun, Q., Xu, W.Q., Han, Y.: Adsorption of lead ion on amino-functionalized fly-ash-based SBA-15 mesoporous molecular sieves prepared via two-step hydrothermal method. Microporous Mesoporous Mater 252(Supplement C), 105–115 (2017). https://doi.org/10.1016/j.micromeso.2017.06.004
Li, Y., Rui, Z., Shen, C., Sun, B., Wang, C., Xiang, L.: Polydopamine coating assisted synthesis of MnO2 loaded inorganic/organic composite electrospun fiber adsorbent for efficient removal of Pb2+ from water. Chem. Eng. J. 344, 277–289 (2018). https://doi.org/10.1016/j.cej.2018.03.044
Liu, L., Chen, H., Shiko, E., Fan, X., Zhou, Y., Zhang, G., Luo, X., Hu, X.: Low-cost DETA impregnation of acid-activated sepiolite for CO2 capture. Chem. Eng. J. 353, 940–948 (2018). https://doi.org/10.1016/j.cej.2018.07.086
Lv, D., Liu, Y., Zhou, J., Yang, K., Lou, Z., Baig, S.A., Xu, X.: Application of EDTA-functionalized bamboo activated carbon (BAC) for Pb(II) and Cu(II) removal from aqueous solutions. Appl. Surf. Sci. 428, 648–658 (2018). https://doi.org/10.1016/j.apsusc.2017.09.151
Machida, M., Fotoohi, B., Amamo, Y., Ohba, T., Kanoh, H., Mercier, L.: Cadmium(II) adsorption using functional mesoporous silica and activated carbon. J. Hazard. Mater. 221–222(4), 220–227 (2012). https://doi.org/10.1016/j.jhazmat.2012.04.039
Maity, J., Ray, S.K.: Chitosan based nano composite adsorbent—synthesis, characterization and application for adsorption of binary mixtures of Pb(II) and Cd(II) from water. Carbohydr. Polym. 182, 159–171 (2018). https://doi.org/10.1016/j.carbpol.2017.10.086
Mejias Carpio, I.E., Machado-Santelli, G., Kazumi Sakata, S., Ferreira Filho, S.S., Rodrigues, D.F.: Copper removal using a heavy-metal resistant microbial consortium in a fixed-bed reactor. Water Res. 62(7), 156–166 (2014). https://doi.org/10.1016/j.watres.2014.05.043
Mureseanu, M., Reiss, A., Stefanescu, I., David, E., Parvulescu, V., Renard, G., Hulea, V.: Modified SBA-15 mesoporous silica for heavy metal ions remediation. Chemosphere 73(9), 1499–1504 (2008). https://doi.org/10.1016/j.chemosphere.2008.07.039
Niu, Y., Qu, R., Sun, C., Wang, C., Chen, H., Ji, C., Zhang, Y., Shao, X., Bu, F.: Adsorption of Pb(II) from aqueous solution by silica-gel supported hyperbranched polyamidoamine dendrimers. J. Hazard. Mater. 244–245(2), 276–286 (2013). https://doi.org/10.1016/j.jhazmat.2012.11.042
Orozco-Guareño, E., Santiago-Gutiérrez, F., Morán-Quiroz, J.L., Hernandez-Olmos, S.L., Soto, V., Cruz, W.D.L., Manríquez, R., Gomez-Salazar, S.: Removal of Cu(II) ions from aqueous streams using poly(acrylic acid-co-acrylamide) hydrogels. J. Colloid Interface Sci. 349(2), 583–593 (2010). https://doi.org/10.1016/j.jcis.2010.05.048
Özer, A., Pirinççi, H.B.: The adsorption of Cd(II) ions on sulphuric acid-treated wheat bran. J. Hazard. Mater. 137(2), 849–855 (2006). https://doi.org/10.1016/j.jhazmat.2006.03.009
Parida, S.K., Dash, S., Patel, S., Mishra, B.K.: Adsorption of organic molecules on silica surface ☆. Adv. Colloid Interface Sci. 121(1), 77–110 (2006). https://doi.org/10.1016/j.cis.2006.05.028
Pérez, R.M., Cabrera, G., Gómez, J.M., Ábalos, A., Cantero, D.: Combined strategy for the precipitation of heavy metals and biodegradation of petroleum in industrial wastewaters. J. Hazard. Mater. 182(1–3), 896–902 (2010). https://doi.org/10.1016/j.jhazmat.2010.07.003
Pérez-Quintanilla, D., del Hierro, I., Fajardo, M., Sierra, I.: Mesoporous silica functionalized with 2-mercaptopyridine: synthesis, characterization and employment for Hg(II) adsorption. Microporous Mesoporous Mater. 89(1), 58–68 (2006). https://doi.org/10.1016/j.micromeso.2005.10.012
Plazinski, W., Rudzinski, W., Plazinska, A.: Theoretical models of sorption kinetics including a surface reaction mechanism: a review. Adv. Coll. Interface. Sci. 152(1), 2–13 (2009). https://doi.org/10.1016/j.cis.2009.07.009
Qorbani, M., Naseri, N., Moradlou, O., Azimirad, R., Moshfegh, A.Z.: How CdS nanoparticles can influence TiO2 nanotube arrays in solar energy applications? Appl. Catal. B 162, 210–216 (2015). https://doi.org/10.1016/j.apcatb.2014.06.053
Sahraei, R., Ghaemy, M.: Synthesis of modified gum tragacanth/graphene oxide composite hydrogel for heavy metal ions removal and preparation of Silver nanocomposite for antibacterial activity. Carbohydr. Polym. 157, 823–833 (2016). https://doi.org/10.1016/j.carbpol.2016.10.059
Sharma, A., Lee, B.K.: Cd(II) removal and recovery enhancement by using acrylamide–titanium nanocomposite as an adsorbent. Appl. Surf. Sci. 313(9), 624–632 (2014). https://doi.org/10.1016/j.apsusc.2014.06.034
Singh, V., Pandey, S., Singh, S.K., Sanghi, R.: Removal of cadmium from aqueous solutions by adsorption using poly(acrylamide) modified guar gum–silica nanocomposites. Sep. Purif. Technol. 67(3), 251–261 (2009). https://doi.org/10.1016/j.seppur.2009.02.023
Sutirman, Z.A., Sanagi, M.M., Karim, K.J.A., Wan, A.W.I., Jume, B.H.: Equilibrium, kinetic and mechanism studies of Cu(II) and Cd(II) ions adsorption by modified chitosan beads. Int. J. Biol. Macromol. 116, 255–263 (2018). https://doi.org/10.1016/j.ijbiomac.2018.05.031
Tran, T.K., Chiu, K.F., Lin, C.Y., Leu, H.J.: Electrochemical treatment of wastewater: selectivity of the heavy metals removal process. Int. J. Hydrog. Energy 42(45), 27741–27748 (2017). https://doi.org/10.1016/j.ijhydene.2017.05.156
Wang, P., Du, M., Zhu, H., Bao, S., Yang, T., Zou, M.: Structure regulation of silica nanotubes and their adsorption behaviors for heavy metal ions: pH effect, kinetics, isotherms and mechanism. J. Hazard. Mater. 286, 533–544 (2015a). https://doi.org/10.1016/j.jhazmat.2014.12.034
Wang, S., Wang, K., Dai, C., Shi, H., Li, J.: Adsorption of Pb2+ on amino-functionalized core–shell magnetic mesoporous SBA-15 silica composite. Chem. Eng. J. 262, 897–903 (2015b). https://doi.org/10.1016/j.cej.2014.10.035
Weibiao, Q., Bingfan, L., Zhangyang, K.: Differential scanning calorimetry and electrochemical tests for the analysis of delamination of 3PE coatings. Int. J. Electrochem. Sci. (2019). https://doi.org/10.20964/2019.08.05
Xu, R., Zhou, G., Tang, Y., Chu, L., Liu, C., Zeng, Z., Luo, S.: New double network hydrogel adsorbent: Highly efficient removal of Cd(II) and Mn(II) ions in aqueous solution. Chem. Eng. J. 275, 179–188 (2015). https://doi.org/10.1016/j.cej.2015.04.040
Yang, Z., Jia, S., Zhuo, N., Yang, W., Wang, Y.: Flocculation of copper(II) and tetracycline from water using a novel pH- and temperature-responsive flocculants. Chemosphere 141, 112–119 (2015). https://doi.org/10.1016/j.chemosphere.2015.06.050
Zhang, X., Zhang, F., Chan, K.Y.: Synthesis and characterization of amino-functionalized mesostructured cellular foams with large mesopores using microemulsion templating. Scr. Mater. 51(4), 343–347 (2004). https://doi.org/10.1016/j.scriptamat.2004.03.042
Zhang, G., Liu, L., Shiko, E., Cheng, Y., Zhang, R., Zeng, Z., Zhao, T., Zhou, Y., Chen, H., Liu, Y., Hu, X.: Low-price MnO2 loaded sepiolite for Cd2+ capture. Adsorption 25(6), 1271–1283 (2019). https://doi.org/10.1007/s10450-019-00132-3
Zhu, H., Jones, D.J., Zajac, J., Dutartre, R., Rhomari, M., Rozière, J.: Synthesis of periodic large mesoporous organosilicas and functionalization by incorporation of ligands into the framework wall. Chem. Mater. 14(14), 4886–4894 (2002). https://doi.org/10.1021/cm011742+
Acknowledgements
The authors acknowledge the National Natural Science Foundation of China (21506179, 51608464, 21506181), Hunan province science and technology department (2017JJ3291, 2017TP2026, 2018SK2027, 2018RS3088, 2019JJ40281, 2019SK2112, 2019JJ30022), Research Start-up Foundation of Xiangtan University (15KZ08065). Research Centre of Chemical Process Simulation Engineering for National Department of Education, Hunan Key Laboratory of Chemical Process Integration Technology for Friendly Environment, National & Local United Engineering Research Centre of Chemical Process Simulation and Intensification, Optimization and Collaborative Innovation Center of New Chemical Technologies for Environmental Benignity and Efficient Resource Utilization and Xiangtan science and technology project, China Scholarship council (CSC). Hunan Provincal Engineering Research Center of Sepiolite Resource for Efficient Utilization.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Xiayi (Eric) Hu and Xuanping Luo are co-first authors.
Rights and permissions
About this article
Cite this article
Hu, X., Luo, X., Xiao, G. et al. Low-cost novel silica@polyacrylamide composites: fabrication, characterization, and adsorption behavior for cadmium ion in aqueous solution. Adsorption 26, 1051–1062 (2020). https://doi.org/10.1007/s10450-020-00225-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10450-020-00225-4