Abstract
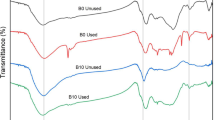
The present study highlights performance of two adsorbents, namely aerocrete and vermiculite, modified with iron oxyhydroxide for removal As(V) from aqueous solution. A face-centered central composite design of response surface methodology (RSM) was employed to optimize efficacy of three experimental variables, adsorbent dosage, pH and initial As(V) concentration, and simultaneously the interactive effects of these variables on As(V) adsorption process were studied. The analysis of variance (ANOVA) of quadratic model shows that the predicted values are in good agreement with experimental data. The results show the leading effect of adsorbent dosage and initial As(V) concentration as well as interactive effect of these two variables on As(V) removal efficiency. Maximum As(V) removal 99.96% and 98.95% are obtained respectively for modified aerocrete and vermiculite at pH of 6, adsorbent dose of 3.1 g/l and initial As(V) concentration of 12 mg/l (ppm). The adsorption data are well fitted with the Langmuir and Freundlich isotherms.





Similar content being viewed by others
References
Bezerra, M.A., Santelli, R.E., Oliveira, E.P., Villar, L.S., Escaleira, L.A.: Response surface methodology (RSM) as a tool for optimization in analytical chemistry. Talanta 76, 965–977 (2008)
Simsek, E.B., Tuna, A.O.A., Beker, U.: A statistical approach for arsenic adsorption onto Turkey clinoptilolite. Environ. Sci. Pollut. Res. 22(5), 3249–3256 (2015)
Bissen, M., Frimmel, F.H.: Arsenic—a review. Part II: Oxidation of arsenic and its removal in water treatment. Acta Hydrochim. Hydrobiol. 31(2), 97–107 (2003)
Cao, J., Yunhai, W., Yanping, J., Palizhati, Y., Wenfu, H.: Response surface methodology approach for optimization of the removal of chromium(VI) by NH2-MCM-41. J. Taiwan Inst. Chem. Eng. 45(3), 860–868 (2014). https://doi.org/10.1016/j.jtice.2013.09.011)
Chaudhry, S.A., Zakiullah, Z., Sharf, I.S.: Isotherm, kinetic and thermodynamics of arsenic adsorption onto iron-zirconium binary oxide-coated sand (IZBOCS): modelling and process optimization. J. Mol. Liq. 229, 230–240 (2017). https://doi.org/10.1016/j.molliq.2016.12.048
Dutta, P.K., Ray, A.K., Sharma, V.K., Millero, F.J.: Adsorption of arsenate and arsenite on titanium dioxide suspensions. J. Colloid Interface Sci. 278(2), 270–275 (2004)
SenGupta, S., Bhattacharyya, K.G.: Kinetics of adsorption of metal ions on inorganic materials: a review. Adv. Colloid Interface Sci. 162(1–2), 39–58 (2011). https://doi.org/10.1016/j.cis.2010.12.004
Han, C., et al.: The optimization of As(V) removal over mesoporous alumina by using response surface methodology and adsorption mechanism. J. Hazard. Mater. 254–255(1), 301–309 (2013). https://doi.org/10.1016/j.jhazmat.2013.04.008
Johnston, R., Heijnen, H.: Safe water technology for arsenic removal. In: Ahmed, M.F., et al. (eds.) Technologies for Arsenic Removal from Drinking Water. Bangladesh University of Engineering and Technology, Dhaka, Bangladesh (2001)
Gallegos-garcia, M., Ramírez-muñiz, K., Song, S.: Arsenic removal from water by adsorption using iron oxide minerals as adsorbents: a review. Miner. Process. Extract. Metall. Rev. 33(5), 301–315 (2012)
Lenoble, V., Bouras, O., Deluchat, V., Serpaud, B., Bollinger, J.C.: Arsenic adsorption onto pillared clays and iron oxides. J. Colloid Interface Sci. 255(1), 52–58 (2002)
Martemianov, D., et al.: Cellular concrete-supported cost-effective adsorbents for aqueous arsenic and heavy metals abatement. J. Environ. Chem. Eng. 5(4), 3930–3941 (2017). https://doi.org/10.1016/j.jece.2017.07.063
Mohan, D., Pittman, C.U.: Arsenic removal from water/wastewater using adsorbents—a critical review. J. Hazard. Mater. 142(1–2), 1–53 (2007)
Namasivayam, C., Senthilkumar, S., 1998. Removal of arsenic(V) from aqueous solution using industrial solid waste: adsorption rates and equilibrium studies. Ind. Eng. Chem. Res. 37(V):4816–22. https://pubs.acs.org/doi/abs/10.1021/ie970774x
Nicomel, N.R., Leus, K., Folens, K., VanDerVoort, P.: Technologies for arsenic removal from water: current status and future perspectives. J. Environ. Res. Public Health. (2015). https://doi.org/10.3390/ijerph13010062
Nieto-delgado, C., Rangel-mendez, J.R.: Anchorage of iron hydro (oxide) nanoparticles onto activated carbon to remove as (V) from water. Water Res. 46(9), 2973–2982 (2012). https://doi.org/10.1016/j.watres.2012.03.026
Panda, H., Tiadi, N., Mohanty, M., Mohanty, C.R.: Studies on adsorption behavior of an industrial waste for removal of chromium from aqueous solution. S. Afr. J. Chem. Eng. 23, 132–138 (2017). https://ac.els-cdn.com/S1026918516300920/1-s2.0-S1026918516300920-main.pdf?_tid=c7e6e68c-47da-11e7-8807-00000aab0f26&acdnat=1496439244_7b20258e1abfe7e518d8125d588b363e.
Pattanayak, J., Mondal, K., Mathew, S., Lalvani, S.B.: Parametric evaluation of the removal of As(V) and As(III) by carbon-based adsorbents. Carbon 38(4), 589–596 (2000)
Rajakovic, L., Todorovic, Z., Rajakovic-Ognjanovic, V., Onjia, A.: Analytical methods for arsenic speciation analysis. J. Serbian Chem. Soc. 78(10), 1461–179 ( ). https://www.scopus.com/inward/record.url?eid=2-s2.0-84887567623&partnerID=tZOtx3y1.
Sasaki, T., Iizuka, A., Watanabe, M., Hongo, T., Yamasaki, A.: Preparation and performance of arsenate (V) adsorbents derived from concrete wastes. Waste Manag. 34(10), 1829–1835 (2014). https://doi.org/10.1016/j.wasman.2014.01.001
Smedley, P.L., Kinniburgh, D.G.: Source and Behaviour of Arsenic in Natural Waters Importance of Arsenic in Drinking Water. British Geological Survey, Oxon (2000)
Smedley, P.L., Kinniburgh, D.G.: A review of the source, behaviour and distribution of arsenic in natural waters. Appl. Geochem. 17(5), 517–568 (2002)
Tian, Y., Wu, M., Lin, X., Huang, P., Huang, Y.: Synthesis of magnetic wheat straw for arsenic adsorption. J. Hazard. Mater. 193, 10–16 (2011). https://doi.org/10.1016/j.jhazmat.2011.04.093
Ungureanu, G., Santos, S., Boaventura, R., Botelho, C.: Arsenic and antimony in water and wastewater: overview of removal techniques with special reference to latest advances in adsorption. J. Environ. Manag. 151, 326–342 (2015)
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Mondal, M., Ray, A.K. Removal of As(V) using low cost adsorbents: aerocrete and vermiculite modified with iron oxy-hydroxide. Adsorption 26, 387–396 (2020). https://doi.org/10.1007/s10450-020-00201-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10450-020-00201-y