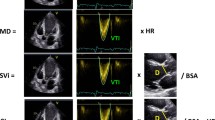
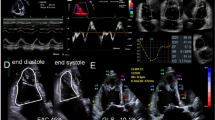
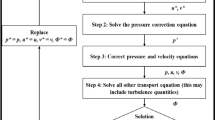
Abstract
Clinical indicators of heart function are often limited in their ability to accurately evaluate the current mechanical state of the myocardium. Biomechanical modeling has been shown to be a promising tool in addition to clinical indicators. By providing a patient-specific measure of myocardial active stress (contractility), biomechanical modeling can enhance the precision of the description of patient’s pathophysiology at any given point in time. In this work we aim to explore the ability of biomechanical modeling to predict the response of ventricular mechanics to the progressively decreasing afterload in repaired tetralogy of Fallot (rTOF) patients undergoing pulmonary valve replacement (PVR) for significant residual right ventricular outflow tract obstruction (RVOTO). We used 19 patient-specific models of patients with rTOF prior to pulmonary valve replacement (PVR), denoted as PSMpre, and patient-specific models of the same patients created post-PVR (PSMpost)—both created in our previous published work. Using the PSMpre and assuming cessation of the pulmonary regurgitation and a progressive decrease of RVOT resistance, we built relationships between the contractility and RVOT resistance post-PVR. The predictive value of such in silico obtained relationships were tested against the PSMpost, i.e. the models created from the actual post-PVR datasets. Our results show a linear 1-dimensional relationship between the in silico predicted contractility post-PVR and the RVOT resistance. The predicted contractility was close to the contractility in the PSMpost model with a mean (± SD) difference of 6.5 (± 3.0)%. The relationships between the contractility predicted by in silico PVR vs. RVOT resistance have a potential to inform clinicians about hypothetical mechanical response of the ventricle based on the degree of pre-operative RVOTO.





Similar content being viewed by others
References
Arts, T., T. Delhaas, P. Bovendeerd, X. Verbeek, and F. W. Prinzen. Adaptation to mechanical load determines shape and properties of heart and circulation: the CircAdapt model. Am. J. Physiol.-Heart Circ. Physiol. 288:H1943–H1954, 2005.
Asner, L., M. Hadjicharalambous, R. Chabiniok, D. Peresutti, E. Sammut, J. Wong, G. Carr-White, P. Chowienczyk, J. Lee, A. King, N. Smith, R. Razavi, and D. Nordsletten. Estimation of passive and active properties in the human heart using 3D tagged MRI. Biomech. Model. Mechanobiol. 15:1121–1139, 2016.
Baumgartner, H., P. Bonhoeffer, N. M. S. De Groot, F. de Haan, J. E. Deanfield, N. Galie, M. A. Gatzoulis, C. Gohlke-Baerwolf, H. Kaemmerer, P. Kilner, F. Meijboom, B. J. M. Mulder, E. Oechslin, J. M. Oliver, A. Serraf, A. Szatmari, E. Thaulow, P. R. Vouhe, E. Walma, and Task Force on the Management of Grown-up Congenital Heart Disease of the European Society of Cardiology (ESC), Association for European Paediatric Cardiology (AEPC), and ESC Committee for Practice Guidelines (CPG). ESC Guidelines for the management of grown-up congenital heart disease (new version 2010). Eur. Heart J. 31:2915–2957, 2010.
Bestel, J., F. Clément, and M. Sorine. A Biomechanical Model of Muscle Contraction. Berlin: Springer, 2001. https://doi.org/10.1007/3-540-45468-3_143.
Caruel, M., R. Chabiniok, P. Moireau, Y. Lecarpentier, and D. Chapelle. Dimensional reductions of a cardiac model for effective validation and calibration. Biomech. Model. Mechanobiol. 13:897–914, 2014.
Castellanos, D. A., K. Škardová, A. Bhattaru, E. Berberoglu, G. Greil, A. Tandon, J. Dillenbeck, B. Burkhardt, T. Hussain, M. Genet, and R. Chabiniok. Left ventricular torsion obtained using equilibrated warping in patients with repaired tetralogy of Fallot. Pediatr. Cardiol. 42:1275–1283, 2021.
Chabiniok, R., P. Moireau, P.-F. Lesault, A. Rahmouni, J.-F. Deux, and D. Chapelle. Estimation of tissue contractility from cardiac cine-MRI using a biomechanical heart model. Biomech. Model. Mechanobiol. 11:609–630, 2012.
Chabiniok, R., V. Y. Wang, M. Hadjicharalambous, L. Asner, J. Lee, M. Sermesant, E. Kuhl, A. A. Young, P. Moireau, M. P. Nash, D. Chapelle, and D. A. Nordsletten. Multiphysics and multiscale modelling, data–model fusion and integration of organ physiology in the clinic: ventricular cardiac mechanics. Interface Focus. 6:20150083, 2016.
Chapelle, D., P. Le. Tallec, P. Moireau, and M. Sorine. An energy-preserving muscle tissue model: formulation and compatible discretizations. Int. J. Multiscale Comput. Eng. 10:189, 2012. https://doi.org/10.1007/978-3-030-21949-9_41
Donati, F., S. Myerson, M. M. Bissell, N. P. Smith, S. Neubauer, M. J. Monaghan, D. A. Nordsletten, and P. Lamata. Beyond Bernoulli: improving the accuracy and precision of noninvasive estimation of peak pressure drops. Circ. Cardiovasc. Imaging. 10:e005207, 2017.
Fučík, R., R. Galabov, P. Pauš, P. Eichler, J. Klinkovský, R. Straka, J. Tintěra, and R. Chabiniok. Investigation of phase-contrast magnetic resonance imaging underestimation of turbulent flow through the aortic valve phantom: experimental and computational study using lattice Boltzmann method. Magn. Reson. Mater. Phys. Biol. Med. 33:649–662, 2020.
Le Gall, A., F. Vallée, K. Pushparajah, T. Hussain, A. Mebazaa, D. Chapelle, É. Gayat, and R. Chabiniok. Monitoring of cardiovascular physiology augmented by a patient-specific biomechanical model during general anesthesia. A proof of concept study. PLoS ONE. 15:e0232830, 2020.
Genet, M., L. C. Lee, B. Baillargeon, J. M. Guccione, and E. Kuhl. Modeling pathologies of diastolic and systolic heart failure. Ann. Biomed. Eng. 44:112–127, 2016.
Geva, T. Repaired tetralogy of Fallot: the roles of cardiovascular magnetic resonance in evaluating pathophysiology and for pulmonary valve replacement decision support. J. Cardiovasc. Magn. Reson. 13:9, 2011.
Göktepe, S., O. J. Abilez, and E. Kuhl. A generic approach towards finite growth with examples of athlete’s heart, cardiac dilation, and cardiac wall thickening. J. Mech. Phys. Solids. 58:1661–1680, 2010.
Gusseva, M., T. Hussain, C. H. Friesen, P. Moireau, A. Tandon, C. Patte, M. Genet, K. Hasbani, G. Greil, D. Chapelle, and R. Chabiniok. Biomechanical modeling to inform pulmonary valve replacement in tetralogy of Fallot patients after complete repair. Can. J. Cardiol. 2021. https://doi.org/10.1016/j.cjca.2021.06.018.
Hadjicharalambous, M., R. Chabiniok, L. Asner, E. Sammut, J. Wong, G. Carr-White, J. Lee, R. Razavi, N. Smith, and D. Nordsletten. Analysis of passive cardiac constitutive laws for parameter estimation using 3D tagged MRI. Biomech. Model. Mechanobiol. 14:807–828, 2015.
Hill, A. V. The heat of shortening and the dynamic constants of muscle. Proc. R. Soc. Lond. Ser. B. 126:136–195, 1938.
Holzapfel, G. A., and R. W. Ogden. Constitutive modelling of passive myocardium: a structurally based framework for material characterization. Philos. Trans. R. Soc. Math. Phys. Eng. Sci. 367:3445–3475, 2009.
Hunter, P. J., and B. H. Smaill. The analysis of cardiac function: a continuum approach. Prog. Biophys. Mol. Biol. 52:101–164, 1988.
Huxley, A. F. Muscle structure and theories of contraction. Prog. Biophys. Biophys. Chem. 7:255–318, 1957.
Kerckhoffs, R. C. P., J. H. Omens, and A. D. McCulloch. A single strain-based growth law predicts concentric and eccentric cardiac growth during pressure and volume overload. Mech. Res. Commun. 42:40–50, 2012.
Kimmig, F., D. Chapelle, and P. Moireau. Thermodynamic properties of muscle contraction models and associated discrete-time principles. Adv. Model. Simul. Eng. Sci. 6:6, 2019.
Kroon, W., T. Delhaas, T. Arts, and P. Bovendeerd. Computational modeling of volumetric soft tissue growth: application to the cardiac left ventricle. Biomech. Model. Mechanobiol. 8:301–309, 2009.
Lee, L. C., M. Genet, G. Acevedo-Bolton, K. Ordovas, J. M. Guccione, and E. Kuhl. A computational model that predicts reverse growth in response to mechanical unloading. Biomech. Model. Mechanobiol. 14:217–229, 2015.
Lumens, J., and T. Delhaas. Cardiovascular modeling in pulmonary arterial hypertension: focus on mechanisms and treatment of right heart failure using the CircAdapt model. Am. J. Cardiol. 110:S39–S48, 2012.
Lumens, J., T. Delhaas, B. Kirn, and T. Arts. Three-wall segment (TriSeg) model describing mechanics and hemodynamics of ventricular interaction. Ann. Biomed. Eng. 37:2234–2255, 2009.
Lumens, J., S. Fan Chun-Po, J. Walmsley, D. Yim, C. Manlhiot, A. Dragulescu, L. Grosse-Wortmann, L. Mertens, W. Prinzen Frits, T. Delhaas, and K. Friedberg Mark. Relative impact of right ventricular electromechanical dyssynchrony versus pulmonary regurgitation on right ventricular dysfunction and exercise intolerance in patients after repair of tetralogy of Fallot. J. Am. Heart Assoc. 8:e010903, 2019.
Lurz, P., J. Nordmeyer, V. Muthurangu, S. Khambadkone, G. Derrick, R. Yates, M. Sury, P. Bonhoeffer, and A. M. Taylor. Comparison of bare metal stenting and percutaneous pulmonary valve implantation for treatment of right ventricular outflow tract obstruction. Circulation. 119:2995–3001, 2009.
McCulloch, A. D., B. H. Smaill, and P. J. Hunter. Left ventricular epicardial deformation in isolated arrested dog heart. Am. J. Physiol. Heart Circ. Physiol. 252:H233–H241, 1987.
Niederer, S. A., G. Plank, P. Chinchapatnam, M. Ginks, P. Lamata, K. S. Rhode, C. A. Rinaldi, R. Razavi, and N. P. Smith. Length-dependent tension in the failing heart and the efficacy of cardiac resynchronization therapy. Cardiovasc. Res. 89:336–343, 2011.
Oosterhof, T., F. J. Meijboom, H. W. Vliegen, M. G. Hazekamp, A. H. Zwinderman, B. J. Bouma, A. P. J. van Dijk, and B. J. M. Mulder. Long-term follow-up of homograft function after pulmonary valve replacement in patients with tetralogy of Fallot. Eur. Heart J. 27:1478–1484, 2006.
Quail, M. A., A. Frigiola, A. Giardini, V. Muthurangu, M. Hughes, P. Lurz, S. Khambadkone, J. E. Deanfield, V. Tsang, and A. M. Taylor. Impact of pulmonary valve replacement in tetralogy of Fallot with pulmonary regurgitation: a comparison of intervention and nonintervention. Ann. Thorac. Surg. 94:1619–1626, 2012.
Regazzoni, F., D. Chapelle, and P. Moireau. Combining data assimilation and machine learning to build data-driven models for unknown long time dynamics—applications in cardiovascular modeling. Int. J. Numer. Methods Biomed. Eng. 37:e3471, 2021.
Ruijsink, B., K. Zugaj, K. Pushparajah, and R. Chabiniok. Model-Based Indices of Early-Stage Cardiovascular Failure and Its Therapeutic Management in Fontan Patients. Cham: Springer, 2019. https://doi.org/10.1007/978-3-030-21949-9_41.
Ruijsink, B., K. Zugaj, J. Wong, K. Pushparajah, T. Hussain, P. Moireau, R. Razavi, D. Chapelle, and R. Chabiniok. Dobutamine stress testing in patients with Fontan circulation augmented by biomechanical modeling. PLOS ONE. 15:e0229015, 2020.
Sainte-Marie, J., D. Chapelle, R. Cimrman, and M. Sorine. Modeling and estimation of the cardiac electromechanical activity. Comput. Struct. 84:1743–1759, 2006.
Sermesant, M., R. Chabiniok, P. Chinchapatnam, T. Mansi, F. Billet, P. Moireau, J. M. Peyrat, K. Wong, J. Relan, K. Rhode, M. Ginks, P. Lambiase, H. Delingette, M. Sorine, C. A. Rinaldi, D. Chapelle, R. Razavi, and N. Ayache. Patient-specific electromechanical models of the heart for the prediction of pacing acute effects in CRT: a preliminary clinical validation. Med. Image Anal. 16:201–215, 2012.
Stergiopulos, N., B. E. Westerhof, and N. Westerhof. Total arterial inertance as the fourth element of the windkessel model. Am. J. Physiol. 276:H81-88, 1999.
Švihlová, H., J. Hron, J. Málek, K. R. Rajagopal, and K. Rajagopal. Determination of pressure data from velocity data with a view toward its application in cardiovascular mechanics. Part 1. Theoretical considerations. Int. J. Eng. Sci. 105:108–127, 2016.
Valente, A. M., K. Gauvreau, G. E. Assenza, S. V. Babu-Narayan, J. Schreier, M. A. Gatzoulis, M. Groenink, R. Inuzuka, P. J. Kilner, Z. Koyak, M. J. Landzberg, B. Mulder, A. J. Powell, R. Wald, and T. Geva. Contemporary predictors of death and sustained ventricular tachycardia in patients with repaired tetralogy of Fallot enrolled in the INDICATOR cohort. Heart. 100:247–253, 2014.
Wang, V. Y., P. M. F. Nielsen, and M. P. Nash. Image-Based Predictive Modeling of Heart Mechanics. Annu. Rev. Biomed. Eng. 17:351–383, 2015.
Acknowledgments
We would like to acknowledge Dr Philippe Moireau, Inria research team MΞDISIM, for the development of the cardiac simulation software CardiacLab used in this work. This work was supported by the Inria-UTSW Associated Team TOFMOD. It was also funded from by the W. B. & Ellen Gordon Stuart Trust, The Communities Foundation of Texas and by the Pogue Family Distinguished Chair (award to Dr F. Gerald Greil in February, 2015). The work was in addition supported by the Ministry of Health of the Czech Republic [NV19-08-00071]. Research reported in this publication was supported by Children’s HealthSM, but the content is solely the responsibility of the authors and does not necessarily represent the official views of Children’s HealthSM.
Funding
Inria, Inria-UTSW Associated Team TOFMOD, Radomír Chabiniok, Ministerstvo Zdravotnictví České Republiky, NV19-08-00071,Radomír Chabiniok, W. B. & Ellen Gordon Stuart Trust, Communities Foundation of Texas, Pogue Family, Pogue Family Distinguished Chair, Gerald Greil.
Author information
Authors and Affiliations
Contributions
Conception of the study: TH, DC, RC. Statistical analysis: MG. Models formulation: DC, RC. Analyze and interpretation of the results: MG, TH, GG, CHF, RC. Design of the figures: MG. Drafting manuscript: MG, RC. Editing and revising manuscript: MG, TH, CHF, GG, DC, RC. Approving final version of manuscript: MG, TH, CHF, GG, DC, RC.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Additional information
Associate Editor Stefan M. Duma oversaw the review of this article.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Gusseva, M., Hussain, T., Hancock Friesen, C. et al. Prediction of Ventricular Mechanics After Pulmonary Valve Replacement in Tetralogy of Fallot by Biomechanical Modeling: A Step Towards Precision Healthcare. Ann Biomed Eng 49, 3339–3348 (2021). https://doi.org/10.1007/s10439-021-02895-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10439-021-02895-9