Abstract
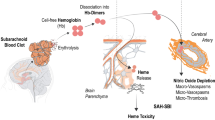
Subarachnoid hemorrhage (SAH) mostly occurs following the rupture of cerebral aneurysm causing blood to leak into the cranial subarachnoid space (SAS). Hemorrhage volume has been linked to the development of secondary vasospasm. Therefore, eliminating blood contaminants from the cerebrospinal fluid (CSF) space after the initial hemorrhage could improve patient outcomes and prevent the development of vasospasm. A number of clinical trials demonstrate that lumbar drainage effectively clears hemorrhagic debris from the cranial compartment. The benefits of optimal lumbar drainage rate and patient orientation are difficult to determine by trial-and-error in live patients, because of the invasive nature, limited subject availability and ethical considerations. Therefore, there is a lack of consensus about clinical guidelines for the use of continuous lumbar drainage following the ictus of SAH. A realistic bench-top model which reproduces the anatomy and CSF dynamics of the human central nervous system (CNS) was built to experimentally study contaminant clearance scenarios under lumbar drainage. To mimic a hemorrhagic event, porcine blood was injected at the basal cistern level of the bench-top model and the efficacy of lumbar drains was assessed experimentally for different drainage rates and patient orientations. In addition, the efficacy of blood clearance was predicted with a computational fluid dynamics (CFD) model. Bench-top experiments and CFD simulations identify body position and drainage rates as key parameters for effective blood clearance. The study findings suggest the importance of treatment in upright position to maximize contaminant diversion from the cranial CSF compartment. The bench-top CNS model together with the validated CFD predictions of lumbar drainage systems can serve to optimize subject-specific treatment options for SAH patients.







Similar content being viewed by others
References
Al-Tamimi, Y. Z., D. Bhargava, R. G. Feltbower, G. Hall, A. J. P. Goddard, A. C. Quinn, and S. A. Ross. Lumbar drainage of cerebrospinal fluid after aneurysmal subarachnoid hemorrhage: a prospective, randomized, controlled trial (LUMAS). Stroke 43:677–682, 2012.
Balédent, O., M. C. Henry-Feugeas, and I. Idy-Peretti. Cerebrospinal fluid dynamics and relation with blood flow: a magnetic resonance study with semiautomated cerebrospinal fluid segmentation. Invest. Radiol. 36:368–377, 2001.
Bednar, M. M., and B. W. Morrison. Cerebrospinal fluid purification system. Patent: WO2011114260 A1, 2011.
Borkar, S., M. Singh, and A. Mahapatra. Spinal cerebrospinal fluid drainage for prevention of vasospasm in aneurysmal subarachnoid hemorrhage: A Prospective Randomized Controlled Study. J. Neurol. Surg. B 73:A379, 2012.
Bottan, S., D. Poulikakos, and V. Kurtcuoglu. Phantom model of physiologic intracranial pressure and cerebrospinal fluid dynamics. IEEE Trans. Biomed. Eng. 59:1532–1538, 2012.
Connolly, E. S., A. A. Rabinstein, J. R. Carhuapoma, C. P. Derdeyn, J. Dion, R. T. Higashida, B. L. Hoh, C. J. Kirkness, A. M. Naidech, C. S. Ogilvy, A. B. Patel, B. G. Thompson, and P. Vespa. Guidelines for the management of aneurysmal subarachnoid hemorrhage a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke 43:1711–1737, 2012.
Cruickshank, A. ACP Best Practice No 166. J. Clin. Pathol. 54:827–830, 2001.
Findlay, J. M., N. F. Kassell, B. K. Weir, E. C. Haley, G. Kongable, T. Germanson, L. Truskowski, W. M. Alves, R. O. Holness, and N. W. Knuckey. A randomized trial of intraoperative, intracisternal tissue plasminogen activator for the prevention of vasospasm. Neurosurgery 37:168–176, 1995; (discussion 177–178).
Fisher, C. M., J. P. Kistler, and J. M. Davis. Relation of cerebral vasospasm to subarachnoid hemorrhage visualized by computerized tomographic scanning. Neurosurgery 6:1–9, 1980.
Ghaffari, M., and C. Y. Hsu. Automatic reconstruction and generation of structured hexahedral mesh for non-planar bifurcations in vascular. Networks 2015. doi:10.13140/RG.2.1.1303.1522.
Gould, I. G., and A. A. Linninger. Hematocrit distribution and tissue oxygenation in large microcirculatory networks. Microcirculation 1994(22):1–18, 2015.
Hänggi, D., J. Liersch, B. Turowski, M. Yong, and H.-J. Steiger. The effect of lumboventricular lavage and simultaneous low-frequency head-motion therapy after severe subarachnoid hemorrhage: results of a single center prospective Phase II trial. J. Neurosurg. 108:1192–1199, 2008.
Hettiarachchi, H. D. M., Y. Hsu, T. J. Harris, Jr, R. Penn, and A. A. Linninger. The effect of pulsatile flow on intrathecal drug delivery in the spinal canal. Ann. Biomed. Eng. 39:2592–2602, 2011.
Hirashima, Y., M. Kurimoto, N. Hayashi, K. Umemura, E. Hori, H. Origasa, and S. Endo. Duration of cerebrospinal fluid drainage in patients with aneurysmal subarachnoid hemorrhage for prevention of symptomatic vasospasm and late hydrocephalus. Neurol. Med. Chir. (Tokyo) 45:177–183, 2005.
Hoekema, D., R. H. Schmidt, and I. Ross. Lumbar drainage for subarachnoid hemorrhage: technical considerations and safety analysis. Neurocrit. Care 7:3–9, 2007.
Hop, J. W., G. J. E. Rinkel, A. Algra, and J. van Gijn. Case-fatality rates and functional outcome after subarachnoid hemorrhage a systematic review. Stroke 28:660–664, 1997.
Hsu, Y. B., H. D. M. Hettiarachchi, D. C. Zhu, and A. A. Linninger. The frequency and magnitude of cerebrospinal fluid pulsations influence intrathecal drug distribution: key factors for interpatient variability. Anesth. Analg. 2012(115):386–394, 2012.
Hsu, C.-Y., B. Schneller, M. Ghaffari, A. Alaraj, and A. Linninger. Medical image processing for fully integrated subject specific whole brain mesh generation. Technologies 3:126–141, 2015.
Inagawa, T., K. Kamiya, and Y. Matsuda. Effect of continuous cisternal drainage on cerebral vasospasm. Acta Neurochir. (Wien) 112:28–36, 1991.
Kassell, N. F., T. Sasaki, A. R. Colohan, and G. Nazar. Cerebral vasospasm following aneurysmal subarachnoid hemorrhage. Stroke 16:562–572, 1985.
Kasuya, H., T. Shimizu, and M. Kagawa. The effect of continuous drainage of cerebrospinal fluid in patients with subarachnoid hemorrhage: a retrospective analysis of 108 patients. Neurosurgery 28:56–59, 1991.
Kawakami, Y., and Y. Shimamura. Cisternal drainage after early operation of ruptured intracranial aneurysm. Neurosurgery 20:8–14, 1987.
Kawamoto, S., K. Tsutsumi, G. Yoshikawa, M. Shinozaki, K. Yako, K. Nagata, and K. Ueki. Effectiveness of the head-shaking method combined with cisternal irrigation with urokinase in preventing cerebral vasospasm after subarachnoid hemorrhage. J. Neurosurg. 100:236–243, 2004.
Kertzscher, U., T. Schneider, L. Goubergrits, K. Affeld, D. Hänggi, and A. Spuler. in vitro study of cerebrospinal fluid dynamics in a shaken basal cistern after experimental subarachnoid hemorrhage. PLoS ONE 7:e41677, 2012.
Kistler, J. P., R. M. Crowell, K. R. Davis, R. Heros, R. G. Ojemann, T. Zervas, and C. M. Fisher. The relation of cerebral vasospasm to the extent and location of subarachnoid blood visualized by CT scan: a prospective study. Neurology 33:424–436, 1983.
Klimo, P., J. R. W. Kestle, J. D. MacDonald, and R. H. Schmidt. Marked reduction of cerebral vasospasm with lumbar drainage of cerebrospinal fluid after subarachnoid hemorrhage. J. Neurosurg. 100:215–224, 2004.
Kodama, N., T. Sasaki, M. Kawakami, M. Sato, and J. Asari. Cisternal irrigation therapy with urokinase and ascorbic acid for prevention of vasospasm after aneurysmal subarachnoid hemorrhage: outcome in 217 patients. Surg. Neurol. 53:110–118, 2000.
Kramer, A. H., and J. J. Fletcher. Locally-administered intrathecal thrombolytics following aneurysmal subarachnoid hemorrhage: a systematic review and meta-analysis. Neurocrit. Care 14:489–499, 2010.
Kwon, O. Y., Y.-J. Kim, Y. J. Kim, C. S. Cho, S. K. Lee, and M. K. Cho. The utility and benefits of external lumbar CSF drainage after endovascular coiling on aneurysmal subarachnoid hemorrhage. J. Korean Neurosurg. Soc. 43:281–287, 2008.
Lad, S., W. C. Mobley, K. Nikolich, and T. Saul. Cerebrospinal fluid purification system. Patent: US8435204 B2, 2013.
Linn, F. H. H., G. J. E. Rinkel, A. Algra, and J. van Gijn. Incidence of subarachnoid hemorrhage: role of region, year, and rate of computed tomography: a meta-analysis. Stroke 27:625–629, 1996.
Linninger, A., C. Tsakiris, D. C. Zhu, M. Xenos, P. Roycewicz, Z. Danziger, and R. Penn. Pulsatile cerebrospinal fluid dynamics in the human brain. IEEE Trans. Biomed. Eng. 52:557–565, 2005.
Linninger, A. A., M. Xenos, B. Sweetman, S. Ponkshe, X. Guo, and R. Penn. A mathematical model of blood, cerebrospinal fluid and brain dynamics. J. Math. Biol. 59:729–759, 2009.
Lueshen, E., I. Venugopal, T. Soni, A. Alaraj, and A. Linninger. Implant-assisted intrathecal magnetic drug targeting to aid in therapeutic nanoparticle localization for potential treatment of central nervous system disorders. J. Biomed. Nanotechnol. 11:253–261, 2015.
Lui, A. C., T. Z. Polis, and N. J. Cicutti. Densities of cerebrospinal fluid and spinal anaesthetic solutions in surgical patients at body temperature. Can. J. Anaesth. J. Can. Anesth. 45:297–303, 1998.
Macdonald, R. L., and B. K. Weir. A review of hemoglobin and the pathogenesis of cerebral vasospasm. Stroke 22:971–982, 1991.
Maeda, Y., S. Shirao, H. Yoneda, H. Ishihara, M. Shinoyama, F. Oka, H. Sadahiro, K. Ueda, Y. Sano, S. Kudomi, Y. Hayashi, T. Shigeeda, K. Nakano, H. Koizumi, S. Nomura, M. Fujii, S. Nomura, and M. Suzuki. Comparison of lumbar drainage and external ventricular drainage for clearance of subarachnoid clots after Guglielmi detachable coil embolization for aneurysmal subarachnoid hemorrhage. Clin. Neurol. Neurosurg. 115:965–970, 2013.
Martin, B. A., R. Labuda, T. J. Royston, J. N. Oshinski, B. Iskandar, and F. Loth. Spinal subarachnoid space pressure measurements in an in vitro spinal stenosis model: implications on syringomyelia theories. J. Biomech. Eng. 132:111007, 2010.
Mcguire, D., T. Saul, and D. F. Gottfried. Combined drug and csf removal therapies and systems. Patent: WO2003015710 A2, 2003
Mizoi, K., T. Yoshimoto, A. Takahashi, S. Fujiwara, K. Koshu, and T. Sugawara. Prospective study on the prevention of cerebral vasospasm by intrathecal fibrinolytic therapy with tissue-type plasminogen activator. J. Neurosurg. 78:430–437, 1993.
Nakagomi, T., K. Takagi, K. Narita, H. Nagashima, and A. Tamura. Cisternal washing therapy for the prevention of cerebral vasospasm following aneurysmal subarachnoid hemorrhage. In: Cerebral Vasospasm, edited by P. D. R. W. Seiler, and P. D. H.-J. Steiger. Vienna: Springer, 2001, pp. 161–165.
Ohira, T., and R. Shiobara. Development of artificial cerebrospinal fluid: basic experiments, and phase II and III clinical trials. J. Neurol. Neurophysiol. 04:173, 2013.
Ormond, D. R., A. Dressler, S. Kim, J. Ronecker, and R. Murali. Lumbar drains may reduce the need for permanent CSF diversion in spontaneous subarachnoid haemorrhage. Br. J. Neurosurg. 27:171–174, 2013.
Osaka, K. Prolonged vasospasm produced by the breakdown products of erythrocytes. J. Neurosurg. 47:403–411, 1977.
Park, S., N. Yang, and E. Seo. The effectiveness of lumbar cerebrospinal fluid drainage to reduce the cerebral vasospasm after surgical clipping for aneurysmal subarachnoid hemorrhage. J. Korean Neurosurg. Soc. 57:167–173, 2015.
Sasaki, T., M. Matsumoto, K. Suzuki, Y. Konno, J. Sakuma, and N. Kodama. Prevention of vasospasm—cisternal irrigation therapy with urokinase and ascorbic acid. Int. Congr. Ser. 1247:611–622, 2002.
Seifert, V., D. Stolke, M. Zimmermann, and A. Feldges. Prevention of delayed ischaemic deficits after aneurysmal subarachnoid haemorrhage by intrathecal bolus injection of tissue plasminogen activator (rTPA). A prospective study. Acta Neurochir. (Wien) 128:137–143, 1994.
Sindhwani, N., O. Ivanchenko, E. Lueshen, K. Prem, and A. A. Linninger. Methods for determining agent concentration profiles in agarose gel during convection-enhanced delivery. IEEE Trans. Biomed. Eng. 58:626–632, 2011.
Solenski, N. J., E. C. Haley, N. F. Kassell, G. Kongable, T. Germanson, L. Truskowski, and J. C. Torner. Medical complications of aneurysmal subarachnoid hemorrhage: a report of the multicenter, cooperative aneurysm study. Participants of the Multicenter Cooperative Aneurysm Study. Crit. Care Med. 23:1007–1017, 1995.
Sonobe, M., and J. Suzuki. Vasospasmogenic substance produced following subarachnoid haemorrhage, and its fate. Acta Neurochir. (Wien) 44:97–106, 1978.
Stockman, H. W. Effect of anatomical fine structure on the flow of cerebrospinal fluid in the spinal subarachnoid space. J. Biomech. Eng. 128:106–114, 2005.
Sweetman, B., and A. A. Linninger. Cerebrospinal fluid flow dynamics in the central nervous system. Ann. Biomed. Eng. 39:484–496, 2011.
Tangen, K. M., Y. Hsu, D. C. Zhu, and A. A. Linninger. CNS wide simulation of flow resistance and drug transport due to spinal microanatomy. J. Biomech. 48:2144–2154, 2015.
Trudnowski, R. J., and R. C. Rico. Specific gravity of blood and plasma at 4 and 37 °C. Clin. Chem. 20:615–616, 1974.
Wahlgren, N. G., and C. Lindquist. Haem derivatives in the cerebrospinal fluid after intracranial haemorrhage. Eur. Neurol. 26:216–221, 1987.
Yushkevich, P. A., J. Piven, H. C. Hazlett, R. G. Smith, S. Ho, J. C. Gee, and G. Gerig. User-guided 3D active contour segmentation of anatomical structures: significantly improved efficiency and reliability. NeuroImage 31:1116–1128, 2006.
Author information
Authors and Affiliations
Corresponding author
Additional information
Associate Editor Kerry Hourigan oversaw the review of this article.
Appendix: Methods
Appendix: Methods
In vitro Model Test with Artificial CSF and Water
The in vitro model is tested with both normal water and artificial CSF to determine any impact the bulk fluid properties will have on blood distribution following subarachnoid hemorrhage.
Artificial CSF is produced using electrolytes, Na, K, Mg, Ca, Cl, HCO3, P, Glucose, and albumin protein; concentrations presented in Appendix Table 3. We made 200 mL of aCSF for each run.
Density as a Function of Temperature for In Vitro Model
The in vitro experiments were performed at the normal laboratory setting of 19oC. We did not create a thermostatic environment due to size limitations. Additionally, the density ratio between CSF and blood was found to be temperature invariant. At 19° laboratory temperature and at body temperature of 37° CSF to blood density ratio is 0.937. The density for water, blood, and CSF at 19° and 37° is compiled in Appendix Table 4.
Womersley numbers (α) were calculated with revision Eq. (4) to characterize the flow conditions due to a pulsatile pressure gradient.38 Here ρ is the density (998.2 kg m−3), T is the period (s), D h is the hydraulic diameter (m), and µ is the viscosity (0.001 kg s−1m−1).
Reynolds number is calculated by:
where ρ is the density (998.2 kg m−3), ν is the peak velocity at the location (m s−1), D h is the hydraulic diameter of that plane (m), and μ is the viscosity (0.001 kg s−1 m−1).
Rights and permissions
About this article
Cite this article
Tangen, K., Narasimhan, N.S., Sierzega, K. et al. Clearance of Subarachnoid Hemorrhage from the Cerebrospinal Fluid in Computational and In Vitro Models. Ann Biomed Eng 44, 3478–3494 (2016). https://doi.org/10.1007/s10439-016-1681-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10439-016-1681-8