Abstract
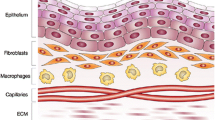
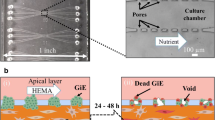
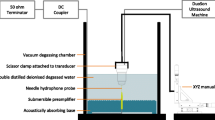
This study uses scanning acoustic microscopy (SAM) ultrasonic profilometry to determine acceptable vs. failed tissue engineered oral mucosa. Specifically, ex vivo-produced oral mucosal equivalents (EVPOMEs) under normal or thermally stressed culture conditions were scanned with the SAM operator blinded to the culture conditions. As seeded cells proliferate, they fill in and smooth out the surface irregularities; they then stratify and produce a keratinized protective upper layer. Some of these transformations could alter backscatter of ultrasonic signals and in the case of the thermally stressed cells, produce backscatter similar to an unseeded device. If non-invasive ultrasonic monitoring could be developed, then tissue cultivation could be adjusted to measure biological variations in the stratified surface. To create an EVPOME device, oral mucosa keratinocytes were seeded onto acellular cadaveric dermis. Two sets of EVPOMEs were cultured: one at physiological temperature 37 °C and the other at 43 °C. The specimens were imaged with SAM consisting of a single-element transducer: 61 MHz center frequency, 32 MHz bandwidth, 1.52 f#. Profilometry for the stressed and unseeded specimens showed higher surface irregularities compared to unstressed specimens. Elevated thermal stress retards cellular differentiation, increasing root mean square values; these results show that SAM can potentially monitor cell/tissue development.







Similar content being viewed by others
References
Bridal, S. L., P. Fornes, P. Bruneval, and G. Berger. Parametric (integrated backscatter and attenuation) images constructed using backscattered radio frequency signals (25–56 MHz) from human aortae in vitro. Ultrasound Med. Biol. 23:215–229, 1997.
Cohen, R. D., J. G. Mottley, J. G. Miller, P. B. Kurnik, and B. E. Sobel. Detection of ischemic myocardium in vivo through the chest wall by quantitative ultrasonic tissue characterization. Am. J. Cardiol. 50:838–843, 1982.
Cohn, N. A., S. Y. Emelianov, M. A. Lubinski, and M. O’Donnell. An elasticity microscope. Part I: methods. IEEE Trans. Ultrason. Ferroelectr. Freq. Control 44:1304–1319, 1997.
Cohn, N. A., S. Y. Emelianov, and M. O’Donnell. An elasticity microscope. Part II: experimental results. IEEE Trans. Ultrason. Ferroelectr. Freq. Control. 44:1320–1331, 1997.
Endo, T., W. J. Finger, M. Kanehira, A. Utterodt, and M. Komatsu. Surface texture and roughness of polished nanofill and nanohybrid resin composites. Dent. Mater. J. 29(2):213–223, 2010.
Holland, M. R., C. S. Hall, S. H. Lewis, S. M. Handley, A. E. Finch-Johnston, A. P. D’Sa, J. E. Perez, and J. G. Miller. Comparison of integrated backscatter values obtained with acoustic densitometry with values derived from spectral analysis of digitized signals from a clinical imaging system. J. Am. Soc. Echocardiogr. 10(5):511–517, 1997.
Hollman, K. W., S. Y. Emelianov, J. H. Neiss, G. Jotyan, G. J. R. Spooner, T. Juhasz, R. M. Kurtz, and M. O’Donnell. Strain imaging of corneal tissue with an ultrasound elasticity microscope. Cornea 21:68–73, 2002.
Insana, M. F., and T. J. Hall. Parametric ultrasound imaging from backscatter coefficient measurements—image-formation and interpretation. Ultrason. Imaging 12:245–267, 1990.
Izumi, K., S. E. Feinberg, A. Iida, and M. Yoshizawa. Intraoral grafting of an ex vivo produced oral mucosa equivalent: a preliminary report. Int. J. Oral Maxillofac. Surg. 32:188–197, 2003.
Izumi, K., J. Song, and S. E. Feinberg. Development of a tissue-engineered human oral mucosa: from the bench to the bed side. Cells Tissues Organs 176:134–152, 2004.
Izumi, K., H. Terashi, C. L. Marcelo, and S. E. Feinberg. Development and characterization of a tissue-engineered human oral mucosa equivalent produced in a serum-free culture system. J. Dent. Res. 79:798–805, 2000.
Kolios, M. C., G. J. Czarnota, M. Lee, J. W. Hunt, and M. D. Sherar. Ultrasonic spectral parameter characterization of apoptosis. Ultrasound Med. Biol. 28:589–597, 2002.
Marchant, R. E., I. Kang, P. S. Sit, Y. Zhou, B. A. Todd, S. J. Eppell, and I. Lee. Molecular views and measurements of hemostatic processes using atomic force microscopy. Curr. Protein Pept. Sci. 3(3):249–274, 2002.
Matsuyama, T., F. G. St. Goar, T. L. Tye, G. Oppenheim, I. Schnittger, and R. L. Popp. Ultrasonic tissue characterization of human hypertrophied hearts in vivo with cardiac cycle-dependent variation in integrated backscatter. Circulation 80:925–934, 1989.
Olerud, J. E., W. D. Obrien, M. A. Riedererhenderson, D. L. Steiger, J. R. Debel, and G. F. Odland. Correlation of tissue constituents with the acoustic properties of skin and wound. Ultrasound Med. Biol. 16:55–64, 1999.
Taggart, L. R., R. E. Baddour, A. Giles, G. J. Czarnota, and M. C. Kolios. Ultrasonic characterization of whole cells and isolated nuclei. Ultrasound Med. Biol. 33:389–401, 2007.
Verstraeten, V. L., and J. Lammerding. Experimental techniques for study of chromatin mechanics in intact nuclei and living cells. Chromosome Res. 16(3):499–510, 2008.
Wickline, S. A., L. J. Thomas, J. G. Miller, B. E. Sobel, and J. E. Perez. The dependence of myocardial ultrasonic integrated backscatter on contractile performance. Circulation 72:183–192, 1985.
Winterroth, F., K. W. Hollman, K. Izumi, S. E. Feinberg, J. B. Fowlkes, and S. J. Hollister. Examination and comparisons of EVPOME/AlloDerm® compositions with human mucosal tissues using scanning acoustic microscopy. In: Society for Biomaterials 2009 Annual Meeting and Exposition, 2009.
Winterroth, F., S. Kuo, K. Izumi, S. E. Feinberg, S. J. Hollister, J. B. Fowlkes, and K. W. Hollman. High-resolution ultrasonic monitoring of cellular differentiation in ex vivo oral mucosal equivalent (EVPOME) tissue culturing. In: Proceedings of the IEEE Bioultrasonics, 2009.
Zuber, M., K. Gerber, and P. Erne. Myocardial tissue characterization in heart failure by real-time integrated backscatter. Eur. J. Ultrasound 9:135–143, 1999.
Acknowledgments
This study was supported through the National Institutes of Health (NIH) Regenerative Sciences Training Grant Number 5T90DK070071 and NIH Grant Numbers R21EY018727, R01 DE13417, and NIH center core (P30) Grant, EY007003. National Institutes of Health, Bethesda, MD. We gratefully acknowledge the NIH Resource Center for Medical Ultrasonic Transducer Technology at the University of Southern California (Los Angeles, CA) for designing and building the high-frequency transducer used in this study.
Author information
Authors and Affiliations
Corresponding author
Additional information
Associate Editor Stefan Jockenhoevel oversaw the review of this article.
Rights and permissions
About this article
Cite this article
Winterroth, F., Lee, J., Kuo, S. et al. Acoustic Microscopy Analyses to Determine Good vs. Failed Tissue Engineered Oral Mucosa Under Normal or Thermally Stressed Culture Conditions. Ann Biomed Eng 39, 44–52 (2011). https://doi.org/10.1007/s10439-010-0176-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10439-010-0176-2