Abstract
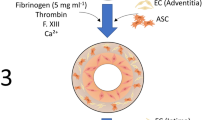
It is generally accepted that endothelialization and subsequent development of a functional endothelium are of paramount importance to the success of any bioartificial artery. In this study, we aimed to assess the ability of smooth muscle cell-remodeled, fibrin-based media-equivalents (MEs) to be endothelialized, examine the morphological changes of endothelial cells (ECs) associated with exposure to physiologically-relevant shear stress in a custom-built bioreactor, and determine if adherent ECs are capable of withstanding average physiological shear stresses. It was found that MEs could be readily endothelialized with surface coverages of 98.8 ± 0.9% after two days, and the ECs expressed von Willebrand factor. Furthermore, EC retention remained high (steady: 96.5 ± 4.4%, pulsatile: 94.3 ± 4.3%) under exposure to physiologically relevant shear stresses for 48 h. The results indicate that these MEs are conducive to generating an EC monolayer, with the ECs possessing adhesion strength sufficient to withstand physiological shear stress and maintain a normal phenotype.









Similar content being viewed by others
REFERENCES
Alberts, B., D. Bray, J. Lewis, M. Raff, K. Roberts, and J. D. Watson. Molecular Biology of the Cell. New York: Garland Publishing, Inc., 1994.
Anderson, J. S., T. M. Price, S. R. Hanson, and L. A. Harker. In vitro endothelialization of small-caliber vascular grafts. Surgery 101:577–586, 1987.
Aper, T., O. E. Teebken, G. Steinhoff, and A. Haverich. Use of a fibrin preparation in the engineering of a vascular graft model. Eur. J. Vasc. Endovasc. Surg. 28:296–302, 2004.
Baguneid, M., D. Murray, H. J. Salacinski, B. Fuller, G. Hamilton, M. Walker, and A. M. Seifalian. Shear-stress preconditioning and tissue-engineering-based paradigms for generating arterial substitutes. Biotechnol. Appl. Biochem. 39:151–157, 2004.
Baker, K. S., S. K. Williams, B. E. Jarrell, E. A. Koolpe, and E. Levine. Endothelialization of human collagen surfaces with human adult endothelial cells. Am. J. Surg. 150:197–200, 1985.
Baumgartner, H. R. Platelet interaction with collagen fibrils in flowing blood. I. Reaction of human platelets with alpha chymotrypsin-digested subendothelium. Thromb. Haemost. 37:1–16, 1977.
Bowlin, G. L., S. E. Rittgers, S. P. Schmidt, T. Alexander, D. B. Sheffer, and A. Milsted. Determination of the prime electrostatic endothelial cell transplantation procedure for e-PTFE vascular prostheses. Cell. Transplant. 9:337–348, 2000.
Buga, G. M., M. E. Gold, J. M. Fukuto, and L. J. Ignarro. Shear stress-induced release of nitric oxide from endothelial cells grown on beads. Hypertension 17:187–193, 1991.
Cines, D. B., E. S. Pollak, C. A. Buck, J. Loscalzo, G. A. Zimmerman, R. P. McEver, J. S. Pober, T. M. Wick, B. A. Konkle, B. S. Schwartz, E. S. Barnathan, K. R. McCrae, B. A. Hug, A. M. Schmidt, and D. M. Stern. Endothelial cells in physiology and in the pathophysiology of vascular disorders. Blood 91:3527–3561, 1998.
Conte, M. S. The ideal small arterial substitute: a search for the Holy Grail? FASEB J. 12:43–45, 1998.
Davies, P. F. Flow-mediated endothelial mechanotransduction. Physiol. Rev. 75:519–560, 1995.
Davies, P. F., A. Remuzzi, E. S. Gordon, C. F. Dewey, and M. A. Gimbrone. Turbulent shear stress induces vascular endothelial turnover in vitro. Proceedings of the National Academy of Science USA. 83:2114–2118, 1986.
Esquivel, C. O., and F. W. Blaisdell. Why small caliber vascular grafts fail: A review of clinical and experimental experience and the significance of the interaction of blood at the interface. J. Vasc. Res. 41:1–15, 1986.
Feugier, P., R. A. Black, J. A. Hunt, and T. V. How. Attachment, morphology and adherence of human endothelial cells to vascular prosthesis materials under the action of shear stress. Biomaterials 26:1457–1466, 2005.
Fields, C., A. Cassano, R. G. Makhoul, C. Allen, R. Sims, J. Bulgrin, A. Meyer, G. L. Bowlin, and S. E. Rittgers. Evaluation of electrostatically endothelial cell seeded expanded polytetrafluoroethylene grafts in a canine femoral artery model. J. Biomater. Appl. 17:135–152, 2002.
Foxall, T. L., K. R. Auger, A. D. Callow, and P. Libby. Adult human endothelial cell coverage of small-caliber Dacron and polytetrafluoroethylene vascular prostheses in vitro. J. Surg. Res. 41:158–172, 1986.
Frangos, J. A., S. G. Eskin, L. V. McIntire, and C. L. Ives. Flow effects on prostacyclin production by cultured human endothelial cells. Science 227:1477–1479, 1985.
Fry, D. L. Acute vascular endothelial changes associated with increased blood velocity gradients. Circ. Res. 22:165–197, 1968.
Galbusera, M., S. Buelli, S. Gastoldi, D. Macconi, S. Angioletti, C. Testa, G. Remuzzi, and M. Morigi. Activation of porcine endothelium in response to xenogeneic serum causes thrombosis independently of platelet activation. Xenotransplantation 12:110–120, 2005.
Galbusera, M., C. Zoja, R. Donadelli, S. Paris, M. Morigi, A. Benigni, M. Figliuzzi, G. Remuzzi, and A. Remuzzi. Fluid shear stress modulates von Willebrand factor release from human vascular endothelium. Blood 90:1558–1564, 1997.
Gosselin, C., D. A. Vorp, V. Warty, D. A. Severyn, E. K. Dick, H. S. Borovetz, and H. P. Greisler. ePTFE coating with fibrin glue, FGF-1, and heparin: effect on retention of seeded endothelial cells. J. Surg. Res. 60:327–332, 1996.
Grabowski, E. F., E. A. Jaffe, and B. B. Weksler. Prostacyclin production by cultured endothelial cell monolayers exposed to step increases in shear stress. J. Lab. Clin. Med. 105:36–43, 1985.
Grassl, E. D., T. R. Oegema, and R. T. Tranquillo. Fibrin as an alternative biopolymer to type-I collagen for the fabrication of a media equivalent. J. Biomed. Mater Res. 60:607–612, 2002.
Grassl, E. D., T. R. Oegema, and R. T. Tranquillo. A fibrin-based arterial media equivalent. J. Biomed. Mater. Res. A 66:550–561, 2003.
Herring, M., A. Gardner, and J. Glover. A single-staged technique for seeding vascular grafts with autogenous endothelium. Surgery 84:498–504, 1978.
Jarrell, B. E., S. K. Williams, J. R. Hoch, and R. A. Carabasi. Perspectives in vascular surgery–biocompatible vascular surfaces: the past and future role of endothelial cells. Bull. N. Y. Acad. Med. 63:156–167, 1987.
Kaehler, J., P. Zilla, R. Fasol, and M. Deutsch. Precoating substrate and surface configuration determine adherence and spreading of seeded endothelial cells on polytetrafluoroethylene grafts. J. Vasc. Surg. 9:535–541, 1989.
Kempczinski, R. F., J. E. Rosenman, W. H. Pearce, L. R. Roedersheimer, Y. Berlatzky, and G. Ramalanjaona. Endothelial cell seeding of a new PTFE vascular prosthesis. J. Vasc. Surg. 2:424–429, 1985.
Kesler, K. A., M. B. Henning, M. P. Arnold, J. L. Glover, H.-M. Park, M. N. Helmus, and P. J. Bendick. Enhanced strength of endothelial attachment on polyester elastomer and polytetrafluoroethylene graft surfaces with fibronectin. J. Vasc. Surg. 3:58–64, 1986.
Klanchar, M., J. M. Tarbell, and D.-M. Wang. In vitro study of radial wall motion on wall shear stress in an elastic tube model of the aorta. Circ. Res. 66:1624–1635, 1990.
Koveker, G. B., L. M. Graham, W. E. Burkel, R. Sell, T. W. Wakefield, K. Dietrich, and J. C. Stanley. Extracellular matrix preparation of expanded polytetrafluoroethylene grafts seeded with endothelial cells: influence on early platelet deposition, cellular growth, and luminal prostacyclin release. Surgery 109:313–319, 1991.
L’Heureux, N., S. Paquet, R. Labbe, L. Germain, and F. A. Auger. A completely biological tissue-engineered human blood vessel. FASEB J. 12:47–56, 1998.
Mazzucotelli, J. P., J. L. Roudiere, F. Bernex, P. Bertrand, J. Leandri, and D. Loisance. A new device for endothelial cell seeding of a small-caliber vascular prosthesis. Artif. Organs. 17:787–790, 1993.
Meinhart, J. G., M. Deutsch, T. Fischlein, N. Howanietz, A. Froschl, and P. Zilla. Clinical autologous in vitro endothelialization of 153 infrainguinal ePTFE grafts. Ann. Thorac. Surg. 71:S327–S331, 2001.
Muller-Glauser, W., P. Zilla, M. Lachat, B. Bisang, F. Rieser, L. von Segesser, and M. Turina. Immediate shear stress resistance of endothelial cell monolayers seeded in vitro on fibrin glue-coated ePTFE prostheses. Eur. J. Vasc. Surg. 7:324–328, 1993.
Nerem, R. M., R. W. Alexander, D. C. Chappell, R. M. Medford, S. E. Varner, and W. R. Taylor. The study of the influence of flow on vascular endothelial biology. Am. J. Med. Sci. 316:169–175, 1998.
Nerem, R. M., and A. E. Ensley. The tissue engineering of blood vessels and the heart. Am. J. Transplant. 4 Suppl 6:36–42, 2004.
Niklason, L. E., J. Gao, W. M. Abbott, K. K. Hirschi, S. Houser, R. Marini, and R. Langer. Functional arteries grown in vitro. Science 284:489–493, 1999.
Ohno, M., J. P. Cooke, V. J. Dzau, and G. H. Gibbons. Fluid shear stress induces endothelial transforming growth factor beta-1 transcription and production. Modulation by potassium channel blockade. J. Clin. Invest. 95:1363–1369, 1995.
Paszkowiak, J. J., and A. Dardik. Arterial wall shear stress: observations from the bench to the bedside. Vasc. Endovasc. Surg. 37:47–57, 2003.
Pawlowski, K. J., S. E. Rittgers, S. P. Schmidt, and G. L. Bowlin. Endothelial cell seeding of polymeric vascular grafts. Front Biosci. 9:1412–1421, 2004.
Prendiville, E. J., J. E. Coleman, A. D. Callow, K. E. Gould, S. Laliberte-Verdon, K. Ramberg, and R. J. Connolly. Increased in vitro incubation time of endothelial cells on fibronectin-treated ePTFE increases cell retention in blood flow. Eur. J. Vasc. Surg. 5:311–319, 1991.
Remy-Zolghadri, M., J. Laganiere, J. F. Oligny, L. Germain, and F. A. Auger. Endothelium properties of a tissue-engineered blood vessel for small-diameter vascular reconstruction. J. Vasc. Surg. 39:613–620, 2004.
Ross, J. J., and R. T. Tranquillo. ECM gene expression correlates with in vitro tissue growth and development in fibrin gel remodeled by neonatal smooth muscle cells. Matrix Biol. 22:477–490, 2003.
Rubanyi, G. M. The role of endothelium in cardiovascular homeostasis and diseases. J. Cardiovasc. Pharmacol. 22 Suppl. 4:S1–S14, 1993.
Salacinski, H. J., S. Goldner, A. Giudiceandrea, G. Hamilton, A. M. Seifalian, A. Edwards, and R. J. Carson. The mechanical behavior of vascular grafts: a review. J. Biomater. Appl. 15:241–278, 2001.
Schmedlen, R. H., W. M. Elbjeirami, A. S. Gobin, and J. L. West. Tissue engineered small-diameter vascular grafts. Clin. Plast. Surg. 30:507–517, 2003.
Schneider, A., M. Chandra, G. Lazarovici, I. Vlodavsky, G. Merin, G. Uretzky, J. B. Borman, and H. Schwalb. Naturally produced extracellular matrix is an excellent substrate for canine endothelial cell proliferation and resistance to shear stress on PTFE vascular grafts. Thromb. Haemost. 78:1392–1398, 1997.
Schneider, P. A., S. R. Hansen, T. M. Price, and L. A. Harker. Preformed confluent endothelial cell monolayers prevent early platelet deposition on vascular prostheses in baboons. J. Vasc. Surg. 8:229–235, 1988.
Seeger, J. M., and N. Klingman. Improved in vivo endothelialization of prosthetic grafts by surface modification with fibronectin. J. Vasc. Surg. 8:476–482, 1988.
Sharefkin, J. B., S. L. Diamond, S. G. Eskin, L. V. McIntire, and C. W. Dieffenbach. Fluid flow decreases preproendothelin mRNA levels and suppresses endothelin-1 peptide release in cultured human endothelial cells. J. Vasc. Surg. 14:1–9, 1991.
Swartz, D. D., J. A. Russell, and S. T. Andreadis. Engineering of fibrin-based functional and implantable small-diameter blood vessels. Am. J. Physiol. Heart. Circ. Physiol. 288:H1451–H1460, 2005.
Teebken, O. E., and A. Haverich. Tissue engineering of small diameter vascular grafts. Eur. J. Vasc. Endovasc. Surg. 23:475–485, 2002.
Tranquillo, R. T. The tissue-engineered small-diameter artery. Ann. N. Y. Acad. Sci. 961:251–254, 2002.
Traub, O., and B. C. Berk. Laminar shear stress: mechanisms by which endothelial cells transduce an antheroprotective force. Arterioscler. Thromb. Vasc. Biol. 18:677–685, 1998.
van Wachem, P. B., J. W. Stronck, R. Koers-Zuideveld, F. Dijk, and C. R. Wildevuur. Vacuum cell seeding: a new method for the fast application of an evenly distributed cell layer on porous vascular grafts. Biomater. 11:602–606, 1990.
Vohra, R. K., G. J. Thompson, H. Sharma, H. M. Carr, and M. G. Walker. Fibronectin coating of expanded polytetrafluoroethylene (ePTFE) grafts and its role in endothelial seeding. Artif. Organs. 14:41–45, 1990.
Vohra, R. K., G. J. Thomson, H. Sharma, H. M. Carr, and M. G. Walker. Effects of shear stress on endothelial cell monolayers on expanded polytetrafluoroethylene (ePTFE) grafts using preclot and fibronectin matrices. Eur. J. Vasc. Surg. 4:33–41, 1990.
Welch, M., D. Durrans, H. M. Carr, R. Vohra, O. B. Rooney, and M. G. Walker. Endothelial cell seeding: a review. Ann. Vasc. Surg. 6:473–484, 1992.
Williams, S. K. Endothelial cell transplantation. Cell Transplant 4:401–410, 1995.
Williams, S. K., D. G. Rose, and B. E. Jarrell. Microvascular endothelial cell sodding of ePTFE vascular grafts: improved patency and stability of the cellular lining. J. Biomed. Mater. Res. 28:203–212, 1994.
Wolf, S., and N. Werthessen. Dynamics of Arterial Flow. New York: Plenum Press, 1979. 472.
Womersley, J. R. Method for the calculation of velocity, rate of flow and viscous drag in arteries when the pressure gradient is known. J. Physiol. 127:553–563, 1955.
Womersley, J. R. Oscillatory motion of a viscous liquid in a thin-walled elastic tube: I. The linear approximation for long waves. Philosophical Magazine 46:199–221, 1955.
Womersley, J. R. Oscillatory flow in arteries: the constrained elastic tube as a model of arterial flow and pulse transmission. Phys. Med. Biol. 2:178–187, 1957.
Xiao, L., and D. Shi. Role of precoating in artificial vessel endothelialization. Chin. J. Traumatol. 7:312–316, 2004.
Ziegler, T., K. Bouzourene, V. J. Harrison, H. R. Brunner, and D. Hayoz. Influence of oscillatory and unidirectional flow environments on the expression of endothelin and nitric oxide synthase in cultured endothelial cells. Arterioscler. Thromb. Vasc. Biol. 18:686–692, 1998.
Zilla, P., R. Fasol, P. Preiss, M. Kadletz, M. Deutsch, H. Schima, S. Tsangaris, and P. Groscurth. Use of fibrin glue as a substrate for in vitro endothelialization of PTFE vascular grafts. Surgery 105:515–522, 1989.
ACKNOWLEDGMENTS
This work was supported by NHLBI HL60495 (R.T.T.). Deborah Cocking-Johnson and Dr. Ginés Escolar performed the Baumgartner assay and the sample analysis, respectively. The technical assistance of Sandra Johnson, Naomi Ferguson, and Diane Tobolt is also gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Isenberg, B.C., Williams, C. & Tranquillo, R.T. Endothelialization and Flow Conditioning of Fibrin-Based Media-Equivalents. Ann Biomed Eng 34, 971–985 (2006). https://doi.org/10.1007/s10439-006-9101-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10439-006-9101-0