Abstract
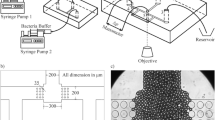
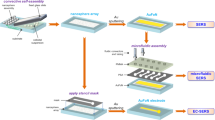
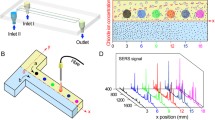
In recent years, researchers have successfully applied diatom biosilica to molecular detection platforms including Surface-Enhanced Raman Scattering (SERS) optofluidic sensors that are currently capable of detecting a variety of biological and chemical molecules at concentrations as low as 10−10 M. This study investigates the feasibility of an SERS device that couples the sensing and pumping capabilities of diatom biosilica thin films by determining flow rate limitations and stability. In this paper, we quantify the ability of porous diatom biosilica thin films to continuously pump deionized (DI) water from a reservoir via wicking flow by utilizing the strong capillary forces of the porous film coupled with evaporation. Our microfluidic device is comprised of a narrow horizontal reservoir fixed to a horizontal capillary whose end contacts a diatom biosilica film. Flow rates were controlled by altering the size and/or temperature of the biosilica porous film, determined by tracking the liquid meniscus displacement in the reservoir, and correlated with a modified laminar boundary-layer model. System stability was observed by tracking flow rates over the course of a given experiment, image analysis of the meniscus contacting the film, and a flow duration study. We found that for untreated DI water bubbles begin to form in the capillary tube at temperatures above 40 °C, but degassed water remains stable at temperatures of 90 °C and below. The pumping capabilities of the films ranged from 0.11 to 10.46 µL/min, matched theoretical predictions, demonstrated stable flow trends, and maintained flow for over 48 h.

















Similar content being viewed by others
References
Bergman TL, Incropera FP (2011) Fundamentals of heat and mass transfer. 7th ed./Theodore 1. Bergman [and others], 7th edn. Wiley, Hoboken
Boyd-Moss M, Baratchi S, Di Venere M, Khoshmanesh K (2016) Self-contained microfluidic systems: a review. Lab Chip 16:3177–3192. https://doi.org/10.1039/C6LC00712K
Chen K-Y, Chen K-E, Wang K (2012) A flexible evaporation micropump with precision flow rate control for micro-fluidic systems. In: IEEE
Choi YH, Chung KH, Lee SS (2009) Microfluidic actuation by dehydration of hydrogel. In: IEEE
Convery N, Gadegaard N (2019) 30 years of microfluidics. Micro Nano Eng 2:76–91. https://doi.org/10.1016/j.mne.2019.01.003
Dollet B, Boulogne F (2017) Natural convection above circular disks of evaporating liquids. Phys Rev Fluids 2:053501. https://doi.org/10.1103/PhysRevFluids.2.053501
Effenhauser C, Harttig H, Krämer P (2002) An evaporation-based disposable micropump concept for continuous monitoring applications. Biomed Microdevice 4:27–32
Ever Aguirre L, Ouyang L, Elfwing A, Hedblom M, Wulff A, Inganäs O (2018) Diatom frustules protect DNA from ultraviolet light. Sci Rep 8:1–6
Goedecke N, Eijkel J, Manz A (2002) Evaporation driven pumping for chromatography application. Lab Chip 2:219–223. https://doi.org/10.1039/B208031C
Gordon R, Losic D, Tiffany MA, Nagy SS, Sterrenburg FAS (2009) The Glass Menagerie: diatoms for novel applications in nanotechnology. Trends Biotechnol 27:116–127. https://doi.org/10.1016/j.tibtech.2008.11.003
Guan Y-X, Xu Z-R, Dai J, Fang Z-L (2006) The use of a micropump based on capillary and evaporation effects in a microfluidic flow injection chemiluminescence system. Talanta 68:1384–1389
Jeffryes C, Campbell J, Li H, Jiao J, Rorrer G (2011) The potential of diatom nanobiotechnology for applications in solar cells, batteries, and electroluminescent devices. Energy Environ Sci 4:3930–3941. https://doi.org/10.1039/C0EE00306A
Jingmin L, Chong L, Zheng X, Kaiping Z, Xue K, Liding W (2012) A microfluidic pump/valve inspired by xylem embolism and transpiration in plants (a bio-inspired microfluidic pump/valve). Plos One 7:e50320. https://doi.org/10.1371/journal.pone.0050320
Juncker D et al (2002) Autonomous microfluidic capillary system. Anal Chem 74:6139–6144
Kearns H, Bedics MA, Shand NC, Faulds K, Detty MR, Graham D (2016) Sensitive SERS nanotags for use with 1550 nm (retina-safe) laser excitation. Analyst 141:5062–5065
Kim H, Kim K, Lee SJ (2016) Compact and thermosensitive nature-inspired micropump. Sci Rep 6:36085
Kirby BJ (2010) Micro- and nanoscale fluid mechanics: transport in microfluidic devices. Cambridge University Press, New York
Kitahama Y, Ozaki Y (2016) Surface-enhanced resonance Raman scattering of hemoproteins and those in complicated biological systems. Analyst 141:5020–5036
Kong X et al (2016) Optofluidic sensing from inkjet-printed droplets: the enormous enhancement by evaporation-induced spontaneous flow on photonic crystal biosilica. Nanoscale 8:17285–17294
Kong X et al (2017) Detecting explosive molecules from nanoliter solution: a new paradigm of SERS sensing on hydrophilic photonic crystal biosilica. Biosens Bioelectron 88:63–70
Kraai JA, Rorrer GL, Wang AX (2019) Highly-porous diatom biosilica stationary phase for thin-layer chromatography. J Chromatogr A 1591:162–170
Laser DJ (2004) Areview of micropumps. J Micromech Microeng 14:R35–R64
Li Y-J, Yang Y-N, Zhang H-J, Xue C-D, Zeng D-P, Cao T, Qin K-R (2019) A microfluidic micropipette aspiration device to study single-cell mechanics inspired by the principle of wheatstone bridge. Micromachines. https://doi.org/10.3390/mi10020131
Lin S, Lin S, Zhu W, Jin Y, Crozier KB (2013) Surface-enhanced Raman scattering with Ag nanoparticles optically trapped by a photonic crystal cavity. Nano Lett 13:559–563
Long DA (2002) The Raman effect: a unified treatment of the theory of Raman scattering by molecules. Chichester Press, Chichester
Lynn NS, Dandy DS (2009) Passive microfluidic pumping using coupled capillary/evaporation effects. Lab Chip 9:3422–3429. https://doi.org/10.1039/B912213C
Marshall KE, Robinson EW, Hengel SM, Pasa-Tolic L, Roesijadi G (2012) FRET imaging of diatoms expressing a biosilica-localized ribose sensor. PLoS One 7(3):e33771
Masoodi R, Pillai KM, Masoodi R, Pillai KM (2012) Wicking in porous materials: traditional and modern modeling approaches. CRC, Boca Raton
Namasivayam V (2003) Transpiration-based micropump for delivering continuous ultra-low flow rates. J Micromech Microeng 13:261–271
Nie C, Frijns AJH, Mandamparambil R, Toonder JMJ (2015) A microfluidic device based on an evaporation-driven micropump. (Report), p 17
Ohno KI, Tachikawa K, Manz A (2008) Microfluidics: applications for analytical purposes in chemistry and biochemistry. Electrophoresis 29:4443–4453
Otto A (1992) Surface-enhanced raman scattering. J Phys Condens Matter 4:1143–1212
Ren F, Campbell J, Hasan D, Wang X, Rorrer GL, Wang AX (2013) Surface-enhanced Raman scattering on diatom biosilica photonic crystals. 8598:85980N-85980N-85988
Sackmann EK, Fulton AL, Beebe DJ (2014) The present and future role of microfluidics in biomedical research. Nature 507:181. https://doi.org/10.1038/nature13118
SE-FIT (2018) https://www.se-fit.com/about/
Sivashanmugan K, Squire K, Kraai JA, Tan A, Zhao Y, Rorrer GL, Wang AX (2019) Biological photonic crystal-enhanced plasmonic mesocapsules: approaching single-molecule optofluidic-SERS sensing. Adv Opt Mater 7:1900415
Squires TM, Quake SR (2005) Microfluidics: fluid physics at the nanoliter scale. Rev Mod Phys 77:977–1026
Taylor J, Huefner A, Li L, Wingfield J, Mahajan S (2016) Nanoparticles and intracellular applications of surface-enhanced Raman spectroscopy. Analyst 141:5037–5055
Temiz Y, Skorucak J, Delamarche E (2014) Capillary-driven microfluidic chips with evaporation-induced flow control and dielectrophoretic microbead trapping vol 8976. In: SPIE
Turner APF (2013) Biosensors: sense and sensibility. Chem Soc Rev 42:3184–3196. https://doi.org/10.1039/C3CS35528D
Wang J, Ahmad H, Ma C, Shi Q, Vermesh O, Vermesh U, Heath J (2010) A self-powered, one-step chip for rapid, quantitative and multiplexed detection of proteins from pinpricks of whole blood. Lab Chip 10:3157–3162. https://doi.org/10.1039/c0lc00132e
White FM (1991) Viscous fluid flow. McGraw-Hill, New York
Woias P (2005) Micropumps—past, progress and future prospects. Sens Actuators B Chem 105:28–38
Xiao-Ming C et al (2019) A capillary-evaporation micropump for real-time sweat rate monitoring with an electrochemical sensor. Micromachines 10:457. https://doi.org/10.3390/mi10070457
Xu Z-R, Zhong C-H, Guan Y-X, Chen X-W, Wang J-H, Fang Z-L (2008) A microfluidic flow injection system for DNA assay with fluids driven by an on-chip integrated pump based on capillary and evaporation effects. Lab Chip 8:1658–1663. https://doi.org/10.1039/B805774E
Xu Y-h, Yan W-p, Qin K-r, Cao T (2019) Three-dimensional flow field simulation of steady flow in the serrated diffusers and nozzles of valveless micro-pumps. J Hydrodyn 31:413–420. https://doi.org/10.1007/s42241-018-0121-7
Zhang A, Zha Y, Zhang J (2014) A surface acoustic wave micropump to pump fluids from a droplet into a closed microchannel using evaporation and capillary effects. AIP Adv 4:127144
Zhou Q, Kim T (2016) Review of microfluidic approaches for surface-enhanced Raman scattering. Sens Actuators B Chem 227:504–514
Zimmermann M, Bentley S, Schmid H, Hunziker P, Delamarche E (2005) Continuous flow in open microfluidics using controlled evaporation. Lab Chip 5:1355–1359
Zimmermann M, Schmid H, Hunziker P, Delamarche E (2007) Capillary pumps for autonomous capillary systems. Lab Chip 7:119–125. https://doi.org/10.1039/B609813D
Acknowledgements
We are grateful to the National Science Foundation (NSF) for funding our research through CBET awards 1701339 and 1701329.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Jarrett, H., Wade, M., Kraai, J. et al. Self-powered microfluidic pump using evaporation from diatom biosilica thin films. Microfluid Nanofluid 24, 36 (2020). https://doi.org/10.1007/s10404-020-02343-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10404-020-02343-5