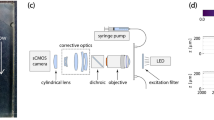
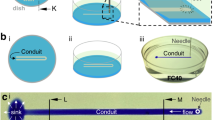
Abstract
Microfluidic systems have been widely used to investigate chemical and biochemical reactions in a small volume. Their geometries are designed to meet the time scale and detection scheme for specific reactions to be analyzed. However, the time scales of the reaction dynamics depend on the type of reactions and concentrations of reactants, and the required time resolution also varies even in a single reaction time course. A system design that is universally applicable to a wide range of time scales should save time and reduce costs. Here, we propose a microfluidic channel design in a fractal-shaped configuration for a kinetic analysis of biochemical reactions encapsulated in microdroplets. We demonstrate that this configuration is effective in resolving the time courses of β-glucuronidase-catalyzed hydrolysis reactions and in vitro translation/transcription reactions, which are different in their reaction time scales, by simply changing the levels of the target nodes in the fractal geometry.




Similar content being viewed by others
References
Anna SL, Bontoux N, Stone HA (2003) Formation of dispersions using “flow focusing” in microchannles. Appl Phys Lett 82:364–366
Clausell-Tormos J, Lieber D, Baret JC, El-Harrak A, Miller OJ, Frenz L, Blouwolff J, Humphry KJ, Koster S, Duan H, Holtze C, Weitz DA, Griffiths AD, Merten CA (2008) Droplet-based microfluidic platforms for the encapsulation and screening of mammalian cells and multicellular organisms. Chem Biol 15:427–437
Courtois F, Olguin LF, Whyte G, Bratton D, Huck WTS, Abell C, Hollfelder F (2008) An integrated device for monitoring time-dependent in vitro expression from single genes in picolitre droplets. Chembiochem 9:439–446
Frenz L, Blank K, Brouzes E, Griffiths AD (2009) Reliable microfluidic on-chip incubation of droplets in delay-lines. Lab Chip 9:1344–1348
Griffiths AD, Tawfik DS (2006) Miniaturising the laboratory in emulsion droplets. Trends Biotech 24:395–402
Haeberle S, Zengerle R (2007) Microfluidic platforms for lab-on-a-chip applications. Lab Chip 7:1094–1110
Holtze C, Rowat AC, Agresti JJ, Hutchison JB, Angile FE, Schmitz CHJ, Koster S, Duan H, Humphry KJ, Scanga RA, Johnson JS, Pisignano D, Weitz DA (2008) Biocompatible surfactants for water-in-fluorocarbon emulsions. Lab Chip 8:1632–1639
Huebner A, Olguin LF, Bratton D, Whyte G, Huck WTS, de Mello AJ, Edel JB, Abell C, Hollfelder F (2008a) Development of quantitative cell-based enzyme assays in microdroplets. Anal Chem 80:3890–3896
Huebner A, Sharma S, Srisa-Art M, Hollfelder F, Edel JB, Demello AJ (2008b) Microdroplets: a sea of applications? Lab Chip 8:1244–1254
Huebner A, Bratton D, Whyte G, Yang M, deMello AJ, Abell C, Hollfelder F (2009) Static microdroplet arrays: a microfluidic device for droplet trapping, incubation and release for enzymatic and cell-based assays. Lab Chip 9:692–698
Kazuta Y, Adachi J, Matsuura T, Ono N, Mori H, Yomo T (2008) Comprehensive analysis of the effects of Escherichia coli ORFs on protein translation reaction. Mol Cell Proteomics 7:1530–1540
Kelly BT, Baret JC, Taly V, Griffiths AD (2007) Miniaturizing chemistry and biology in microdroplets. Chem Commun 1773–1788
Koster S, Angile FE, Duan H, Agresti JJ, Wintner A, Schmitz C, Rowat AC, Merten CA, Pisignano D, Griffiths AD, Weitz DA (2008) Drop-based microfluidic devices for encapsulation of single cells. Lab Chip 8:1110–1115
Matsuura T, Kazuta Y, Aita T, Adachi J, Yomo T (2009) Quantifying epistatic interactions among the components constituting the protein translation system. Mol Syst Biol 5:297
Matsuura T, Hosoda K, Ichihashi N, Kazuta Y, Yomo T (2011) Kinetic analysis of b-galactosidase and b-glucuronidase tetramerization coupled with protein translation. J Biol Chem 286:22028–22034
Mazutis L, Baret JC, Treacy P, Skhiri Y, Araghi AF, Ryckelynck M, Taly V, Griffiths AD (2009) Multi-step microfluidic droplet processing: kinetic analysis of an in vitro translated enzyme. Lab Chip 9:2902–2908
Schaerli Y, Wootton RC, Robinson T, Stein V, Dunsby C, Neil MAA, French PMW, deMello AJ, Abell C, Hollfelder F (2009) Continuous-flow polymerase chain reaction of single-copy DNA in microfluidic microdroplets. Anal Chem 81:302–306
Schmitz CH, Rowat AC, Koster S, Weitz DA (2009) Dropspots: a picoliter array in a microfluidic device. Lab Chip 9:44–49
Shestopalov I, Tice JD, Ismagilov RF (2004) Multi-step synthesis of nanoparticles performed on millisecond time scale in a microfluidic droplet-based system. Lab Chip 4:316–321
Shim JU, Olguin LF, Whyte G, Scott D, Babtie A, Abell C, Huck WT, Hollfelder F (2009) Simultaneous determination of gene expression and enzymatic activity in individual bacterial cells in microdroplet compartments. J Am Chem Soc 131:15251–15256
Shimizu Y, Inoue A, Tomari Y, Suzuki T, Yokogawa T, Nishikawa K, Ueda T (2001) Cell-free translation reconstituted with purified components. Nat Biotechnol 19:751–755
Song H, Ismagilov RF (2003) Millisecond kinetics on a microfluidic chip using nanoliters of reagents. J Am Chem Soc 125:14613–14619
Song H, Tice JD, Ismagilov RF (2003) A microfluidic system for controlling reaction networks in time. Angew Chem Int Ed Engl 42:768–772
Squires TM, Quake SR (2005) Microfluidics: fluid physics at the nanoliter scale. Rev Modern Phys 77:977–1026
Urano Y, Kamiya M, Kanda K, Ueno T, Hirose K, Nagano T (2005) Evolution of fluorescein as a platform for finely tunable fluorescence probes. J Am Chem Soc 127:4888–4894
Zheng B, Roach LS, Ismagilov RF (2003) Screening of protein crystallization conditions on a microfluidic chip using nanoliter-size droplets. J Am Chem Soc 125:11170–11171
Zheng B, Tice JD, Ismagilov RF (2004a) Formation of arrayed droplets by soft lithography and two-phase fluid flow, and application in protein crystallization. Adv Mater 16:1365–1368
Zheng B, Tice JD, Ismagilov RF (2004b) Formation of droplets of alternating composition in microfluidic channels and applications to indexing of concentrations in droplet-based assays. Anal Chem 76:4977–4982
Zheng B, Tice JD, Roach LS, Ismagilov RF (2004c) A droplet-based, composite PDMS/glass capillary microfluidic system for evaluating protein crystallization conditions by microbatch and vapor-diffusion methods with on-chip X-ray diffraction. Angew Chem Int Ed Engl 43:2508–2511
Acknowledgments
We thank Dr. Yasuaki Kazuta, Ms. Hitomi Komai, and Tomomi Sakamoto for producing the PURE system. This research was supported in part by “Special Coordination Funds for Promoting Science and Technology: Yuragi Project”, the Global COE (Centers of Excellence) Program of the Japanese Ministry of Education, Culture, Sports, Science and Technology.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Hirata, K., Ichii, T., Suzuki, H. et al. Fractal-shaped microchannel design for a kinetic analysis of biochemical reaction in a delay line. Microfluid Nanofluid 13, 273–278 (2012). https://doi.org/10.1007/s10404-012-0958-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10404-012-0958-y