Abstract
Introduction
Dual-frequency ultrasound has recently been shown to extensively enhance the acoustic cavitation yield in water.
Methods
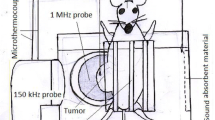
In this study, the in vivo antitumor effect of simultaneous, dual-frequency ultrasound at low-level intensity (I SATA = 2 W/cm2 for 1 MHz and I SATA = 0.2 W/cm2 for 150 kHz) in combination with an intravenous injection of 5 mg/kg hematoporphyrin (Hp) was investigated in a model of breast adenocarcinoma in Balb/c mice. Seventy-one tumor-bearing mice were divided into nine treatment groups: control, sham, Hp injection, and single- and dual-frequency sonication in the presence and absence of Hp. The tumor growth delay was then calculated based on the tumor volume at various times after treatment using the following parameters: relative volume percent, \( T_{5} \) and \( T_{2} \) times (to reach five and two times the initial volume), percent of tumor growth inhibition ratio, and survival period.
Results
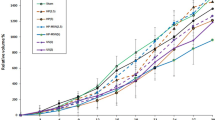
Our results showed no significant difference between the 150 kHz and 1 MHz single-frequency groups when compared with the sham group after 9 days of treatment (p > 0.05). However, treatment with dual-frequency ultrasound significantly delayed tumor growth when compared with the sham group (p < 0.05) after 9 days of treatment. Furthermore, in vivo experiments showed that combined dual-frequency sonication controlled tumor growth more effectively than single-frequency sonication. Evaluation of the therapeutic effects of single- and dual-frequency ultrasound in sonodynamic therapy revealed that treatment with the combination of dual-frequency ultrasound and Hp resulted in a significant reduction in the relative volume percent of tumors after 3 days of treatment (p < 0.05) compared with the controls. Additionally, the T 5 time and the survival period in the group treated with the combination therapy was significantly longer than those in all the other groups (p < 0.05). These findings were further verified histopathologically.
Conclusion
In conclusion, our results reveal that sonodynamic therapy using dual-frequency ultrasound is able to extend the survival time of animals compared with single-frequency sonication.












Similar content being viewed by others
References
Liu Q, Li X, Xiao L, Wang P, Wang X, Tang W. Sonodynamically induced antitumor effect of hematoporphyrin on Hepatoma 22. Ultrason Sonochem. 2008;15:943–8.
Yumita N, Nishigaki R, Umemura S. Sonodynamically induced antitumor effect of Photofrin II on colon 26 carcinoma. J Cancer Res Clin Oncol. 2000;126:601–6.
Yumita N, Umemura S. Sonodynamic therapy with Photofrin II on AH130 solid tumor. Cancer Chemother Pharmacol. 2003;51:174–8.
Tomankova K, Kolarova H, Kolar P, Kejlova K, Jirova D. Study of cytotoxic effect of photodynamically and sonodynamically activated sensitizers in vitro. Toxicol In Vitro. 2009;23:1465–71.
Bailey MR, Khokhlova VA, Sapozhnikov OA, Kargl SG, Crum LA. Physical mechanisms of the therapeutic effect of ultrasound: a review. Acoust Phys. 2003;49:369–88.
Umemura S, Yumita N, Nishigaki R, Umemura K. Mechanism of cell damage by ultrasound in combination with hematoporphyrin. Jpn J Cancer Res. 1990;81:962–6.
Barnett S. Cavitation—its nature, detection and measurement. Ultrasound Med Biol. 1998;24:S11–21.
Neppiras EA, Noltingk BE. Cavitation produced by ultrasonics: theoretical conditions for the onset of cavitation. Proc Phys Soc. 1951;12:1032–8.
Laborde JL, Bouyer C, Caltagirone JP, Gerard A. Acoustic cavitation field prediction at low and high frequency ultrasounds. Ultrasonics. 1998;36:581–7.
Liu HL, Hsieh CM. Single transducer dual frequency ultrasound generation to enhance acoustic cavitation. Ultrason Sonochem. 2009;16:431–8.
Barati AH, Mokhtari-Dizaji M, Mozdarani H, Bathaie SZ, Hassan ZM. Effect of exposure parameters on cavitation induced by low level dual frequency ultrasound. Ultrason Sonochem. 2007;14:783–9.
Brotchie A, Grieser F, Ashokkumar M. Sonochemistry and sonoluminescence under dual frequency ultrasound irradiation in the presence of water soluble solutes. J Phys Chem C. 2008;112:10247–50.
Zheng H, Mukdadi O, Kim H, Hertzberg JR, Shandas R. Advantages in using multifrequency excitation of contrast microbubbles for enhancing echo particle image velocimetry techniques: initial numerical studies using rectangular and triangular waves. Ultrasound Med Biol. 2005;31:99–108.
Servant G, Laborde JL, Hita A, Caltagirone JP, Gerard A. On the interaction between ultrasound waves and bubble clouds in mono and dual frequency sonoreactors. Ultrason Sonochem. 2003;10:347–55.
Iernetti G, Ciuti P, Dezhkunov NV, Reali M, Francescutto A, Johri GK. Enhancement of high frequency acoustic cavitation effects by a low frequency stimulation. Ultrason Sonochem. 1997;4:263–8.
Tachibana K, Feril LB Jr, Ikeda-Dantsuji Y. Sonodynamic therapy. Ultrasonics. 2008;48:253–9.
Rosenthal I, Sostaric JZ, Riesz P. Sonodynamic therapy: a review of the synergistic effects of drugs and ultrasound. Ultrason Sonochem. 2004;11:349–63.
Kessel D, Jeffers R, Fowlkes JB, Cain C. Porphyrin-induced enhancement of ultrasound cytotoxicity. Int J Radiat Biol. 1994;66:221–8.
Warthington AE, Thompson J, Lalonde R, Patterson M, Rauth AM, Hunt JW. Mechanism of ultrasound enhanced porphyrin cytotoxcity: free radical and hematoporphyrin effect. Ultrasound Med Biol. 1997;23:1095–105.
Yumita N, Okuyama N, Sasaki K, Umemura S. Sonodynamic therapy on chemically induced mammary tumor: pharmacokinetics, tissue distribution and sonodynamically induced antitumor effect of porfimer sodium. Cancer Sci. 2004;95:765–9.
Tochner Z, Mitchell JB, Harrington FS, Smith P, Russo DT, Russo A. Treatment of murine intraperitoneal ovarian ascitic tumor with hematoporphyrin derivative and laser light. Cancer Res. 1985;45:2983–7.
Chekulayeva LV, Chekulayev VA, Shevchuk IN. Active oxygen intermediates in the degradation of hematoporphyrin derivative in tumor cells subjected to photodynamic therapy. J Photochem Photobiol B. 2008;93:94–107.
Huang J, Feng R, Zhu C, Chen Z. Low-MHz frequency effect on a sonochemical reaction determined by an electrical method. Ultrason Sonochem. 1995;2:93–7.
Barati AH, Mokhtari-Dizaji M, Mozdarani H, Bathaie SZ, Hassan ZM. Treatment of murine tumors using dual frequency ultrasound in an experimental in vivo model. Ultrasound Med Biol. 2009;35:756–63.
Teicher BA. Tumor model in cancer research. Totawa: Human Press; 2002. p. 407–49.
Barati AH, Mokhtari-Dizaji M. Ultrasound dose fractionation in sonodynamic therapy. Ultrasound Med Biol. 2010;36:880–7.
Austerlitz C, de Souza VLB, Campos DMT, Kurachi C, Bagnato C, Sibata C. Enhanced response of the Fricke solution doped with hematoporphyrin under X-rays irradiation. Braz Arch Biol Technol. 2008;51:271–9.
Bloom-Richardson Grade for Breast Cancer, Cancer Reporting in California: Abstracting and Coding Procedures for Hospitals (California Cancer Reporting System Standards, vol. I). 2007;3–8.
Suslick KS, Flannigan DJ. Inside a collapsing bubble: sonoluminescence and the conditions during cavitation. Annu Rev Phys Chem. 2008;59:659–83.
Wang S, Huang B, Wang Y, Liao L. Comparison of enhancement of pentachlorophenol sonolysis at 20 kHz by dual frequency sonication. Ultrason Sonochem. 2006;13:506–10.
Ciuti P, Dezhkunov NV, Francescutto A, Kulak AL, Iernetti G. Cavitation activity stimulation by low frequency field pulses. Ultrason Sonochem. 2000;7:213–6.
Tang HW, Blankschtein D, Langer R. An investigation of the role of cavitation in low frequency ultrasound mediated transdermal drug transport. Pharm Res. 2002;19:1160–9.
Kanthale PM, Brotchie A, Ashokkumar M, Grieser F. Experimental and theoretical investigations on sonoluminescence under dual frequency conditions. Ultrason Sonochem. 2008;15:629–35.
Hasanzadeh H, Mokhtari-Dizaji M, Bathaie SZ, Hassan ZM. Evaluation of correlation between chemical dosimetry and subharmonic spectrum analysis to examine the acoustic cavitation. Ultrason Sonochem. 2010;17:863–9.
Hasanzadeh H, Mokhtari-Dizaji M, Bathaie SZ, Hassan ZM, Nilchiani V, Goudarzi H. Enhancement and control of acoustic cavitation yield by low level dual frequency sonication: a subharmonic analysis. Ultrason Sonochem. 2011;18:394–400.
Acknowledgments
The authors would like to thank Dr. Shahbazfar from Tabriz University for the histological study. This work was supported in part by the Iran National Science Foundation (INSF).
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Alamolhoda, M., Mokhtari-Dizaji, M., Barati, A.H. et al. Comparing the in vivo sonodynamic effects of dual- and single-frequency ultrasound in breast adenocarcinoma. J Med Ultrasonics 39, 115–125 (2012). https://doi.org/10.1007/s10396-012-0348-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10396-012-0348-9