Abstract
Purpose
In this study, the effects of low-intensity pulsed ultrasound (LIU) as an adjuvant to doxorubicin (DOX) treatment was further investigated in comparison to hyperthermia as another widely used adjuvant. The effects were compared with respect to cell killing and apoptosis induction in U937 cells. Human primary liver cancer (PLC) cells were also used to evaluate the effects of the combinations. The use of an echo contrast agent was investigated for further enhancement of cytotoxicity. Finally, the acoustic mechanisms involved were investigated.
Methods
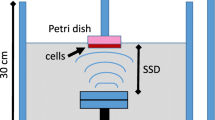
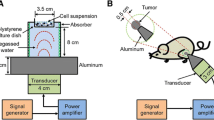
The effects of different treatment regimens on cell viability were determined using the Trypan blue dye-exclusion test. Apoptosis induction was detected by flow cytometry using fluorescein isothiocyanate-annexin V and propidium iodide staining. The mechanistic study involved electron paramagnetic spin trapping for detecting free radical formation as an indicator of the occurrence of inertial cavitation and spectrophotometry for sucrose hydrolysis as an indicator for noncavitational effects.
Results
The combination treatments exerted synergistic effects on cytotoxicity depending on the acoustic conditions used. The use of LIU as an adjuvant to DOX treatment was shown to be superior to the use of hyperthermia as an adjuvant. Moreover, the combination seems to be promising for other cancer types provided that the acoustic conditions are properly selected with respect to drug concentration. The key ultrasound mechanism responsible for the synergism observed was shown to be the production of free radicals by inertial cavitation. Non-cavitational forces were also shown to contribute to the effect.
Conclusion
This study is motivating to engage in in vivo research with various cancer types as a step toward clinical applicability and is emphasizing on the importance of developing therapeutic protocols for setting LIU parameters with respect to other therapeutic conditions.
Similar content being viewed by others
References
Wood RW, Loomis AL. The physical and biological effects of high-frequency sound waves of great intensity. Phil Mag S Series 7 1927;7:417.
Kremkau FW. Cancer therapy with ultrasound: a historical review. J Clin Ultrasound 1979;7:287–300.
Blana A, Murat FJ, Walter B, et al. First analysis of the long-term results with transrectal HIFU in patients with localised prostate cancer. Eur Urol 2008;53:1194–1201.
Chaussy C, Thuroff S, Rebillard X, et al. Technology insight: high-intensity focused ultrasound for urologic cancers. Nat Clin Pract Urol 2005;2:191–198.
Larkin JO, Casey GD, Tangney M, et al. Effective tumor treatment using optimized ultrasound-mediated delivery of bleomycin. Ultrasound Med Biol 2008;34:406–413.
Feril LB Jr, Kondo T, Cui ZG, et al. Apoptosis induced by the sonomechanical effects of low-intensity pulsed ultrasound in a human leukemia cell line. Cancer Lett 2005;221:145–152.
Feng Y, Tian ZM, Wan MX, et al. Low-intensity ultrasoundinduced apoptosis in human gastric carcinoma cells. World J Gastroenterol 2008;14:4873–4879.
Feril LB Jr, Kondo T. Biological effects of low-intensity ultrasound: the mechanism involved, and its implication on therapy and on biosafety of ultrasound. Rev J Radiat Res 2004;45:479–489.
Yoshida T, Kondo T, Ogawa R, et al. Molecular therapy using ultrasound: mechanisms involved in drug activation, apoptosis induction, gene transfer, and alterations of gene expression. Rev Thermal Med 2007;23:113–122.
Harrison GH, Balcer-Kubiczek EK, Eddy HA. Potentiation of chemotherapy by low-level ultrasound. Int J Radiat Biol 1991;59:1453–1466.
Loverock P, ter Haar G, Ormerod MG, et al. The effect of ultrasound on the cytotoxicity of adriamycin. Br J Radiol 1990;63:542–546.
Umemura S, Yumita N, Okano Y, et al. Sonodynamically induced in vitro cell damage enhanced by adriamycin. Cancer Lett 1997;121:195–201.
Yu T, Bai J, Hu K, et al. The effect of free radical scavenger and antioxidant on the increase in intracellular adriamycin accumulation induced by ultrasound. Ultrason Sonochem 2003;10:33–35.
Yu T, Wang Z, Jiang S. Potentiation of cytotoxicity of adriamycin on human ovarian carcinoma cell line 3AO by low-level ultrasound. Ultrasonics 2001;39:307–309.
Yoshida T, Kondo T, Ogawa R, et al. Combination of doxorubicin and low-intensity ultrasound causes a synergistic enhancement in cell killing and an additive enhancement in apoptosis induction in human lymphoma U937 cells. Cancer Chemother Pharmacol 2008;61:559–567.
Honda H, Kondo T, Zhao QL, et al. Role of intracellular calcium ions and reactive oxygen species in apoptosis induced by ultrasound. Ultrasound Med Biol 2004;30:683–692.
Honda H, Zhao QL, Kondo T. Effects of dissolved gases and an echo contrast agent on apoptosis induced by ultrasound and its mechanism via the mitochondria-caspase pathway. Ultrasound Med Biol 2002;28:673–682.
Tabuchi Y, Takasaki I, Zhao QL, et al. Genetic networks responsive to low-intensity pulsed ultrasound in human lymphoma U937 cells. Cancer Lett 2008;270:286–294.
Ando H, Feril LB Jr, Kondo T, et al. An echo-contrast agent, Levovist, lowers the ultrasound intensity required to induce apoptosis of human leukemia cells. Cancer Lett 2006;242:37–45.
Correas JM, Kurtisovski E, Bridal SL, et al. Optimizing an ultrasound contrast agent’s stability using in vitro attenuation measurements. Invest Radiol 2002;37:672–679.
Feril LB Jr, Kondo T. Major factors involved in the inhibition of ultrasound-induced free radical production and cell killing by presonication incubation or by high cell density. Ultrason Sonochem 2005;12:353–357.
Feril LB Jr, Kondo T, Zhao QL, et al. Enhancement of hyperthermia-induced apoptosis by non-thermal effects of ultrasound. Cancer Lett 2002;178:63–70.
Hiraoka W, Honda H, Feril LB Jr, et al. Comparison between sonodynamic effect and photodynamic effect with photosensitizers on free radical formation and cell killing. Ultrason Sonochem 2006;13:535–542.
Woodcock JP. The effect of non-cavitating ultrasound on the hydrolysis of sucrose solutions. Acustica 1969;21:45–50.
Feril LB Jr, Kondo T, Zhao QL, et al. Enhancement of ultrasoundinduced apoptosis and cell lysis by echo-contrast agents. Ultrasound Med Biol 2003;29:331–337.
Ogawa R, Kondo T, Honda H, et al. Effects of dissolved gases and an echo contrast agent on ultrasound-mediated in vitro gene transfection. Ultrason Sonochem 2002;9:197–203.
Watanabe A, Otake R, Nozaki T, et al. Effects of microbubbles on ultrasound-mediated gene transfer in human prostate cancer PC3 cells: comparison among Levovist, YM454, and MRX-815H. Cancer Lett 2008;265:107–112.
Duvshani-Eshet M, Baruch L, Kesselman E, et al. Therapeutic ultrasound-mediated DNA to cell and nucleus: bioeffects revealed by confocal and atomic force microscopy. Gene Ther 2006;13:163–172.
Sauer H, Putz V, Fischer K, et al. Increased doxorubicin uptake and toxicity in multicellular tumour spheroids treated with DC electrical fields. Br J Cancer 1999;80:1204–1213.
Keizer HG, Pinedo HM, Schuurhuis GJ, et al. Doxorubicin (adriamycin): a critical review of free radical-dependent mechanisms of cytotoxicity. Pharmacol Ther 1990;47:219–231.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Kondo, T., Yoshida, T., Ogawa, R. et al. Low-intensity ultrasound adjuvant therapy: enhancement of doxorubicin-induced cytotoxicity and the acoustic mechanisms involved. J Med Ultrasonics 36, 61–68 (2009). https://doi.org/10.1007/s10396-009-0212-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10396-009-0212-8