Abstract
Purpose
Our purpose was to evaluate the anatomical and functional changes in retinae of rabbit eyes following monocular intravitreal injection of sodium iodate (SI).
Methods
Twenty albino rabbits were divided into four groups and underwent monocular intravitreal injection with four different doses of SI (0.1, 0.2, 0.4, and 0.8 mg). Before and for 28 days after injection, the eyes were examined using fundus photography, optical coherence tomography (OCT), and electroretinography (ERG). At postinjection days 2, 7, and 28, the eyes were enucleated and underwent histological examination.
Results
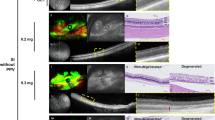
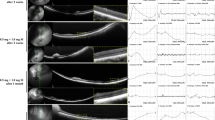
On fundus examination, no distinct retinal changes were seen in any group except the 0.8-mg group, which showed chorioretinal vascular attenuation. In 0.1 and 0.2-mg groups, no significant anatomical changes were found except transient hyperreflective dots over the vitreoretinal interface on OCT. In 0.4 and 0.8-mg groups, disruption of the ellipsoid zone and diffuse retinal swelling were observed in the early period on OCT. In the 0.4-mg group, the outer retina was significantly destroyed at day 28, whereas the inner retina was relatively preserved. In the 0.8-mg group, the entire retina was destroyed irreversibly. The b-wave of ERG was reduced immediately in all groups, which recovered fully (0.1- and 0.2-mg groups), partially (0.4-mg group), or never (0.8-mg group). No structural or functional abnormalities were found in the fellow control eyes.
Conclusions
Retinal degeneration following intravitreal injection of SI appears to be dose dependent; retinal damage is reversible at low doses but irreversible at high doses. At a certain dose, the outer retina may be preferably ablated.







Similar content being viewed by others
References
Chader GJ. Animal models in research on retinal degenerations: past progress and future hope. Vis Res. 2002;42:393–9.
Kondo M, Sakai T, Komeima K, Kurimoto Y, Ueno S, Nishizawa Y, et al. Generation of a transgenic rabbit model of retinal degeneration. Invest Ophthalmol Vis Sci. 2009;50:1371–7.
Li T, Snyder WK, Olsson JE, Dryja TP. Transgenic mice carrying the dominant rhodopsin mutation P347S: evidence for defective vectorial transport of rhodopsin to the outer segments. Proc Natl Acad Sci USA. 1996;93:14176–81.
Nishida K, Kamei M, Kondo M, Sakaguchi H, Suzuki M, Fujikado T, et al. Efficacy of suprachoroidal-transretinal stimulation in a rabbit model of retinal degeneration. Invest Ophthalmol Vis Sci. 2010;51:2263–8.
Yamauchi Y, Agawa T, Tsukahara R, Kimura K, Yamakawa N, Miura M, et al. Correlation between high-resolution optical coherence tomography (OCT) images and histopathology in an iodoacetic acid-induced model of retinal degeneration in rabbits. Br J Ophthalmol. 2011;95:1157–60.
Isago H, Sugano E, Murayama N, Tamai M, Tomita H. Establishment of monocular-limited photoreceptor degeneration models in rabbits. BMC Ophthalmol. 2013;13:19.
Rosch S, Johnen S, Mataruga A, Muller F, Pfarrer C, Walter P. Selective photoreceptor degeneration by intravitreal injection of N-methyl-N-nitrosourea. Invest Ophthalmol Vis Sci. 2014;55:1711–23.
Kiuchi K, Yoshizawa K, Shikata N, Moriguchi K, Tsubura A. Morphologic characteristics of retinal degeneration induced by sodium iodate in mice. Curr Eye Res. 2002;25:373–9.
Sorsby A. Experimental pigmentary degeneration of the retina by sodium iodate. Br J Ophthalmol. 1941;25:58–62.
Yang Y, Ng TK, Ye C, Yip YW, Law K, Chan SO, et al. Assessing sodium iodate-induced outer retinal changes in rats using confocal scanning laser ophthalmoscopy and optical coherence tomography. Invest Ophthalmol Vis Sci. 2014;55:1696–705.
Machalinska A, Lubinski W, Klos P, Kawa M, Baumert B, Penkala K, et al. Sodium iodate selectively injuries the posterior pole of the retina in a dose-dependent manner: morphological and electrophysiological study. Neurochem Res. 2010;35:1819–27.
Kitano S, Hori S, Nagataki S. Transport of fluorescein in the rabbit eye after treatment with sodium iodate. Exp Eye Res. 1988;46:863–70.
Amirpour N, Karamali F, Rabiee F, Rezaei L, Esfandiari E, Razavi S, et al. Differentiation of human embryonic stem cell-derived retinal progenitors into retinal cells by Sonic hedgehog and/or retinal pigmented epithelium and transplantation into the subretinal space of sodium iodate-injected rabbits. Stem Cells Dev. 2012;21:42–53.
Wang K, Li XX, Jiang YR, Dong JQ. Influential factors of thresholds for electrically evoked potentials elicited by intraorbital electrical stimulation of the optic nerve in rabbit eyes. Vis Res. 2007;47:3012–24.
Siu T, Morley J. Implantation of episcleral electrodes via anterior orbitotomy for stimulation of the retina with induced photoreceptor degeneration: an in vivo feasibility study on a conceptual visual prosthesis. Acta Neurochir (Wien). 2008;150:477–85 (discussion 485).
Murray MM. The effects of administration of sodium iodate to man and animals. Bull World Health Organ. 1953;9:211–6.
Siu TL, Morley JW. Influence of callosal transfer on visual cortical evoked response and the implication in the development of a visual prosthesis. Graefes Arch Clin Exp Ophthalmol. 2007;245:1797–803.
Matsuo Y, Sakamoto T, Yamashita T, Tomita M, Shirasawa M, Terasaki H. Comparisons of choroidal thickness of normal eyes obtained by two different spectral-domain OCT instruments and one swept-source OCT instrument. Invest Ophthalmol Vis Sci. 2013;54:7630–6.
Marmor MF, Fulton AB, Holder GE, Miyake Y, Brigell M, Bach M. ISCEV Standard for full-field clinical electroretinography (2008 update). Doc Ophthalmol. 2009;118:69–77.
Dolz-Marco R, Gallego-Pinazo R, Pinazo-Duran MD, Pons-Vazquez S, Domingo-Pedro JC, Diaz-Llopis M. Intravitreal docosahexaenoic acid in a rabbit model: preclinical safety assessment. PLoS One. 2014;9:e96872.
Sorsby A, Reading HW. Experimental degeneration of the retina. XI. The effect of sodium iodate on retinal -SH levels. Vision Res. 1964;4:511–4.
Ringvold A, Olsen EG, Flage T. Transient breakdown of the retinal pigment epithelium diffusion barrier after sodium iodate: a fluorescein angiographic and morphological study in the rabbit. Exp Eye Res. 1981;33:361–9.
Grignolo A, Orzalesi N, Calabria GA. Studies on the fine structure and the rhodopsin cycle of the rabbit retina in experimental degeneration induced by sodium iodate. Exp Eye Res. 1966;5:86–97.
Redfern WS, Storey S, Tse K, Hussain Q, Maung KP, Valentin JP, et al. Evaluation of a convenient method of assessing rodent visual function in safety pharmacology studies: effects of sodium iodate on visual acuity and retinal morphology in albino and pigmented rats and mice. J Pharmacol Toxicol Methods. 2011;63:102–14.
Korte GE, Wanderman MC. Distribution of Na+K(+)-ATPase in regenerating retinal pigment epithelium in the rabbit. A study by electron microscopic cytochemistry. Exp Eye Res. 1993;56:219–29.
Korte GE, Rappa E, Andracchi S. Localization of alkaline phosphatase on basolateral plasma membrane of normal and regenerating retinal pigment epithelium. A cytochemical study in rabbits. Invest Ophthalmol Vis Sci. 1991;32:3187–97.
Obata R, Yanagi Y, Tamaki Y, Hozumi K, Mutoh M, Tanaka Y. Retinal degeneration is delayed by tissue factor pathway inhibitor-2 in RCS rats and a sodium-iodate-induced model in rabbits. Eye (Lond). 2005;19:464–8.
Korte GE, Mrowiec E, Landzberg KS, Youssri A. Reorganization of actin microfilaments and microtubules in regenerating retinal pigment epithelium. Exp Eye Res. 1995;61:189–203.
Korte GE, Reppucci V, Henkind P. RPE destruction causes choriocapillary atrophy. Invest Ophthalmol Vis Sci. 1984;25:1135–45.
Flage T, Ringvold A. The retinal pigment epithelium diffusion barrier in the rabbit eye after sodium iodate injection. A light and electron microscopic study using horseradish peroxidase as a tracer. Exp Eye Res. 1982;34:933–40.
Ogata N, Kanai K, Ohkuma H, Uyama M. Pathologic response of the regenerated retinal pigment epithelium (RPE)—affected by sodium iodate (NaIO3). Nihon Ganka Gakkai Zasshi. 1989;93:466–74.
Mizota A, Adachi-Usami E. Functional recovery of retina after sodium iodate injection in mice. Vis Res. 1997;37:1859–65.
Machalinska A, Kawa MP, Pius-Sadowska E, Roginska D, Klos P, Baumert B, et al. Endogenous regeneration of damaged retinal pigment epithelium following low dose sodium iodate administration: an insight into the role of glial cells in retinal repair. Exp Eye Res. 2013;112:68–78.
Wang J, Iacovelli J, Spencer C, Saint-Geniez M. Direct effect of sodium iodate on neurosensory retina. Invest Ophthalmol Vis Sci. 2014;55:1941–53.
Franco LM, Zulliger R, Wolf-Schnurrbusch UE, Katagiri Y, Kaplan HJ, Wolf S, et al. Decreased visual function after patchy loss of retinal pigment epithelium induced by low-dose sodium iodate. Invest Ophthalmol Vis Sci. 2009;50:4004–10.
Tanaka M, Machida S, Ohtaka K, Tazawa Y, Nitta J. Third-order neuronal responses contribute to shaping the negative electroretinogram in sodium iodate-treated rats. Curr Eye Res. 2005;30:443–53.
Stone JL, Barlow WE, Humayun MS, de Juan E, Milam AH Jr. Morphometric analysis of macular photoreceptors and ganglion cells in retinas with retinitis pigmentosa. Arch Ophthalmol. 1992;110:1634–9.
Enzmann V, Row BW, Yamauchi Y, Kheirandish L, Gozal D, Kaplan HJ, et al. Behavioral and anatomical abnormalities in a sodium iodate-induced model of retinal pigment epithelium degeneration. Exp Eye Res. 2006;82:441–8.
Ashburn FS Jr, Pilkerton AR, Rao NA, Marak GE. The effects of iodate and iodoacetate on the retinal adhesion. Invest Ophthalmol Vis Sci. 1980;19:1427–32.
Konda BR, Pararajasegaram G, Wu GS, Stanforth D, Rao NA. Role of retinal pigment epithelium in the development of experimental autoimmune uveitis. Invest Ophthalmol Vis Sci. 1994;35:40–7.
Baich A, Ziegler M. The effect of sodium iodate and melanin on the formation of glyoxylate. Pigment Cell Res. 1992;5:394–5.
Heike M, Marmor MF. l-cystein protects the pigment epithelium from acute sodium iodate toxicity. Doc Ophthalmol. 1990;75:15–22.
Noell WK. Experimentally induced toxic effects on structure and function of visual cells and pigment epithelium. Am J Ophthalmol. 1953;36:103–16.
Tao Z, Dai J, He J, Li C, Li Y, Yin ZQ. The influence of NaIO(3)-induced retinal degeneration on intra-retinal layer and the changes of expression profile/morphology of DA-ACs and mRGCS. Mol Neurobiol. 2013;47:241–60.
Negi A, Marmor MF. The resorption of subretinal fluid after diffuse damage to the retinal pigment epithelium. Invest Ophthalmol Vis Sci. 1983;24:1475–9.
Machalinska A, Lejkowska R, Duchnik M, Kawa M, Roginska D, Wiszniewska B, et al. Dose-dependent retinal changes following sodium iodate administration: application of spectral-domain optical coherence tomography for monitoring of retinal injury and endogenous regeneration. Curr Eye Res. 2014;39:1033–41.
Acknowledgments
This study was supported by the Public Welfare and Safety Program 2012-0006566 by Ministry of Education and Science Technology of Korea.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
B.-J. Cho, None; J.-M. Seo, None; H. G. Yu, None; H. Chung, None.
About this article
Cite this article
Cho, BJ., Seo, JM., Yu, H.G. et al. Monocular retinal degeneration induced by intravitreal injection of sodium iodate in rabbit eyes. Jpn J Ophthalmol 60, 226–237 (2016). https://doi.org/10.1007/s10384-016-0429-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10384-016-0429-1