Abstract
Purpose
To investigate the incidence of adverse effects on eyelids and eyelashes related to treatment with prostaglandin analogs (PGAs) in patients who apply the medication in only one eye.
Methods
A clinical examination of 39 glaucoma patients treated for over 3 months continuously in one eye with one of four PGAs. Face photographs were used to judge adverse effects of PGA treatment on eyelids and eyelashes of the treated eyes by comparing them with the eyelids and eyelashes of the contralateral eyes. Each of three examiners then calculated scores for the eyelids and eyelashes by counting how many adverse effects were present (out of five possible effects for eyelids and two for eyelashes). The findings for the eyelids or eyelashes were considered “positive” if two or more examiners judged that at least one adverse effect was present.
Results
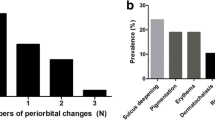
Thirty-five (89.7 %) patients were judged to have positive eyelid findings and 37 (94.9 %) patients were judged to have positive eyelash findings. There was a significant correlation between the period of PGA administration and eyelid score (P = 0.0218).
Conclusion
There was a high incidence of adverse effects caused by PGAs in the eyelids and eyelashes. The frequency of adverse effects of the eyelid seemed to increase as the use of PGAs was prolonged. The cosmetic complications caused by PGA use should be considered, and patients should be informed of them in advance.

Similar content being viewed by others
References
Maruyama K. Medical therapy for normal-tension glaucoma. Atarashii Ganka (in Japanese). 2008;25:795–8.
Kashiwagi K, Tsumura T, Tsukahara S. Long-term effects of latanoprost monotherapy on intraocular pressure in Japanese glaucoma patients. J Glaucoma. 2008;17:662–6.
Yamazaki S, Nanno M, Kimura T, Suzumura H, Yoshikawa K. Effects of switching to SofZia-preserved travoprost in patients who preserved with superficial punctate keratopathy while under treatment with latanoprost. Jpn J Ophthalmol. 2010;54:7–14.
Peplinski LS, Smith KA. Deepening of lid sulcus from topical bimatoprost therapy. Optom Vis Sci. 2004;81:574–7.
Filippopoulos T, Paula JS, Torun N, Hatton MP, Pasquale LR, Grosskreutz CL. Periorbital changes associated with topical bimatoprost. Ophthal Plast Reconstr Surg. 2008;24:302–7.
Ha SJ, Kim HD, Lee HJ, Choi K-S. Deep lid sulcus and blepharoptosis after treatment with topical travoprost and bimatoprost. Am Acad Ophthalmol. 2008;11:8–11.
Tappeiner C, Perren B, Iliev ME, Frueh BE, Goldblum D. Orbital fat atrophy in glaucoma patients treated with topical bimatoprost: can bimatoprost cause enophthalmos? Klin Monbl Augenheilkd. 2008;225:443–5.
Yam JC, Yuen NS, Chan CW. Bilateral deepening of upper lid sulcus from topical bimatoprost therapy. J Ocul Pharmacol Ther. 2009;25:471–2.
Yang HK, Park KH, Kim TW, Kim DM. Deepening of eyelid superior sulcus during topical travoprost treatment. Jpn J Ophthalmol. 2009;53:176–9.
Jayaprakasam A, Ghazi-Nouri S. Periorbital fat atrophy; an unfamiliar side effect of prostaglandin analogues. Orbit. 2010;29:357–9.
Park J, Cho HK, Moon JI. Changes to upper eyelid orbital fat from use of topical bimatoprost, travoprost, and latanoprost. Jpn J Ophthalmol. 2011;55:22–7.
Watanabe I, Maruo H, Watanabe I. Deepening of the sulcus of upper eyelid following instillation of travoprost. Rinsho Ganka (in Japanese). 2011;65:679–82.
Aihara M, Shirato S, Sakata R. Incidence of deepening of the upper eyelid sulcus after switching from latanoprost to bimatoprost. Jpn J Ophthalmol. 2011;55:600–4.
Chiba T, Kashiwagi K, Kogure S, Abe K, Shibuya T, Furuichi M, et al. Iridial pigmentation induced by latanoprost ophthalmic solution in Japanese glaucoma patients. J Glaucoma. 2001;10:406–10.
Inoue K, Wakakura M, Inoue J, Matsuo H, Hara T, Tomita G. Adverse reaction after use of latanoprost in Japanese glaucoma patients. Nippon Ganka Gakkai Zasshi (in Japanese). 2006;110:581–7.
Alm A, Schoenfelder J, McDermott J. A 5-year, multicenter, open-label, safety study of adjunctive latanoprost therapy for glaucoma. Arch Ophthalmol. 2004;122:957–65.
Goldberg I, Li XY, Selaru P, Paggiarino D. A 5-year, randomized, open-label safety study of latanoprost and usual care in patients with open-angle glaucoma or ocular hypertension. Eur J Ophthalmol. 2008;18:408–16.
Hara T. Increased iris pigmentation after use of latanoprost in Japanese brown eyes. Nippon Ganka Gakkai Zasshi (in Japanese). 2001;105:314–21.
Latanoprost-Induced Iris Pigmentation Study Group. Incidence of a latanoprost-induced increase in iris pigmentation in Japanese eyes. Jpn J Ophthalmol. 2006;50:96–9.
Lee AJ, McCluskey P. Clinical utility and differential effects of prostaglandin analogs in the management of raised intraocular pressure and ocular hypertension. Clin Ophthalmol. 2010;4:741–64.
Kitazawa Y, Komemushi S. Single-masked, randomized, parallel-group comparison of bimatoprost ophthalmic solution and latanoprost ophthalmic solution in patients with primary open-angle glaucoma or ocular hypertension. Atarashii Ganka (in Japanese). 2010;27:401–10.
Doshi M, Edward DP, Osmanovic S. Clinical course of bimatoprost-induced periocular skin changes in Caucasians. Ophthalmology. 2006;113:1961–7.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Yoshino, T., Fukuchi, T., Togano, T. et al. Eyelid and eyelash changes due to prostaglandin analog therapy in unilateral treatment cases. Jpn J Ophthalmol 57, 172–178 (2013). https://doi.org/10.1007/s10384-012-0199-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10384-012-0199-3