Abstract
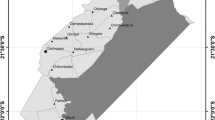
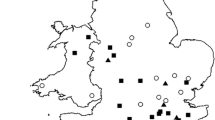
Chlamydophila (C.) abortus is the most common infectious abortigenic agent in small domestic ruminants in Switzerland. In contrast, the knowledge about chlamydiae in wild ruminants is scarce. As interactions between livestock and Alpine ibex (Capra i. ibex) occur on alpine pastures, the question raises if wild ruminants could play a role as carriers of chlamydiae. Thus, we investigated the prevalence of chlamydiae in Alpine ibex in Switzerland. In total, 624 sera, 676 eye swabs, 84 organ samples and 51 faecal samples from 664 ibex were investigated. Serum samples were tested by two commercial ELISA kits specific for C. abortus. Eye swabs, organs and faecal samples were examined by a Chlamydiaceae-specific real-time polymerase chain reaction (PCR). Positive cases were further investigated by the ArrayTube (AT) microarray method for chlamydial species determination. Of 624 serum samples investigated, 612 animals were negative, whereas nine sera (1.5%) reacted positively in one of the two tests and three sera showed an inconclusive result. Eye swabs of seven out of 412 ibex (1.7%) were tested positive for Chlamydiaceae by real-time PCR. By AT microarray, Chlamydophila (C.) pecorum was identified in two animals, Chlamydophila (C.) pneumoniae was detected in one animal and a mixed infection with C. abortus and C. pecorum was found in four animals. Organs and faecal samples were all negative by real-time PCR analysis. In summary, we conclude that C. abortus is not a common infectious agent in the Swiss ibex population. To our knowledge, this is the first description of C. pneumoniae in ibex. Further studies are necessary to elucidate the situation in other species of wild ruminants as chamois (Rupicapra r. rupicapra), red deer (Cervus elaphus) and roe deer (Capreolus c. capreolus) in Switzerland.

Similar content being viewed by others
References
Belloy L, Janovsky M, Vilei EM, Pilo P, Giacometti M, Frey J (2003) Molecular epidemiology of Mycoplasma conjunctivae in Caprinae: transmission across species in natural outbreaks. Appl Environ Microbiol 69:1913–1919
Bodetti TJ, Jacobson E, Wan C, Hafner L, Pospischil A, Rose K, Timms P (2002) Molecular evidence to support the expansion of the host range of Chlamydophila pneumoniae to include reptiles as well as humans, horses, koalas and amphibians. Syst Appl Microbiol 25:146–152
Borel N, Doherr MG, Vretou E, Psarrou E, Thoma R, Pospischil A (2004) Seroprevalences for ovine enzootic abortion in Switzerland. Prev Vet Med 65(3–4):205–216
Borel N, Kempf E, Hotzel H, Schubert E, Torgerson P, Slickers P, Ehricht R, Tasaraf T, Pospischil A, Sachse K (2008) Direct identification of chlamydiae from clinical samples using a DNA microarray assay—a validation study. Mol Cell Probes 22:55–64
Chanton-Greutmann H, Thoma R, Corboz L, Borel N, Pospischil A (2002) Abortion in small ruminants in Switzerland: investigations during two lambing seasons (1996–1998) with special regard to chlamydial abortions. Schweiz Arch Tierheilkd 144(9):483–492
Cubero-Pablo MJ, Plaza M, Pérez L, González M, León-Vizcaíno L (2000) Seroepidemiology of chlamydial infections of wild ruminants in Spain. J Wildl Dis 36(1):35–47
Debbie JG (1967) Chlamydia (Psittacosis) antibody study in White-tailed deer. J Wildl Dis 3:152–155
Donn A, Jones GE, Ruiu A, Ladu M, Machell J, Stancanelli A (1997) Serological diagnosis of chlamydial abortion in sheep and goats: comparison of the complement-fixation test and an enzyme-linked immuno-sorbent assay employing solubilised proteins as antigen. Vet Microbiol 59:27–36
Ehricht R, Slickers P, Goellner S, Hotzel H, Sachse K (2006) Optimized DNA microarray assay allows detection and genotyping of single PCR-amplifiable target copies. Mol Cell Probes 20:60–63
Eidgenössische Jagdstatistik, BAFU (2008) http://www.wild.uzh.ch/jagdst/. Accessed 28 December 2009
Giacometti M, Roganti R, De Tann D, Stahlberger-Saitbekova N, Obexer-Ruff G (2004) Alpine ibex (Capra ibex ibex) × domestic goat (C. aegagrus domestica) hybrids in a restricted area of southern Switzerland. Wildl Biol 10:137–143
Giacometti M, Tolari F, Mannelli A, Lanfranchi P (1995) Seroepidemiologic investigations in the alpine ibex (Capra i. ibex) of piz albris in the canton of grigioni (Switzerland). Schweiz Arch Tierheilkd 137(12):537–542
Giovannini A, Cancellotti FM, Turilli C, Randi E (1988) Serological investigations for some bacterial and viral pathogens in fallow deer (Cervus dama) and wild boar (Sus scrofa) of the San Rossore Preserve, Tuscany, Italy. J Wildl Dis 24(1):127–132
Jones GE, Low JC, Machell J, Armstrong K (1997) Comparison of five tests for the detection of antibodies against chlamydial (enzootic) abortion of ewes. Vet Rec 141:164–168
Kita J, Anusz K (1991) Serologic survey for bovine pathogens in free-ranging European bison from Poland. J Wildl Dis 27(1):16–20
Longbottom D, Coulter LJ (2003) Animal chlamydioses and zoonotic implications. J Comp Pathol 128(4):217–244
Longbottom D, Fairley S, Chapman S, Psarrou E, Vretou E, Livingstone M (2002) Serological diagnosis of ovine enzootic abortion by enzyme-linked immunosorbent assay using a recombinant protein fragment of the polymorphic outer membrane protein POMP90 of Chlamydophila abortus. J Clin Microbiol 40:4235–4243
Markey BK, McNulty MS, Todd D (1993) Comparison of serological tests for the diagnosis of Chlamydia psittaci infection of sheep. Vet Microbiol 36:233–252
Marreros N, Hüssy D, Albini S, Frey C, Abril C, Vogt H-R, Holzwarth N, Wirz-Dittus S, Friess M, Engels M, Borel N, Willisch C, Signer C, Hoelzle L, Ryser-Degiorgis M-P (submitted) Epizootiologic investigations of selected abortive agents in free-ranging Alpine ibex (Capra ibex ibex) in Switzerland. J Wildl Dis
Mayer D, Degiorgis MP, Meier W, Nicolet J, Giacometti M (1997) Lesions associated with infectious keratoconjunctivitis in Alpine ibex. J Wildl Dis 33(3):413–419
Meagher M, Quinn WJ, Stackhouse L (1992) Chlamydial-caused infectious keratoconjunctivitis in bighorn sheep of Yellowstone National Park. J Wildl Dis 28(2):171–176
Myers GS, Mathews SA, Eppinger M, Mitchell C, O’Brien KK, White OR, Benahmed F, Brunham RC, Read TD, Ravel J, Bavoil PM, Timms P (2009) Evidence that human Chlamydia pneumoniae was zoonotically acquired. J Bacteriol 191(23):7225–7233
Pioz M, Loison A, Gauthier D, Gibert P, Jullien J-M, Artois M, Gilot-Fromont E (2008a) Diseases and reproductive success in a wild mammal: example in the alpine chamois. Oecologia 155:691–704
Pioz M, Loison A, Gibert P, Jullien J-M, Artois M, Gilot-Fromont E (2008b) Antibodies against Salmonella is associated with reduced reproductive success in female alpine chamois (Rupicapra rupicapra). Can J Zool 86:1111–1120
Polkinghorne A, Borel N, Becker A, Lu ZH, Zimmermann DR, Brugnera E, Pospischil A, Vaughan L (2009) Molecular evidence for chlamydial infections in the eyes of sheep. Vet Microbiol 135:142–146
Pospischil A, Thoma R, Hilbe M, Grest P, Zimmermann D, Gebbers JO (2002) Abortion in humans caused by Chlamydophila abortus (Chlamydia psittaci serovar 1). Schweiz Arch Tierheilkd 144(9):463–466
Rodolakis A, Salinas J, Papp J (1998) Recent advances on ovine chlamydial abortion. Vet Rec 29:275–288
Ryser-Degiorgis MP, Bischof DF, Marreros N, Willisch C, Signer C, Filli F, Brosi G, Frey J, Vilei EM (2009) Detection of Mycoplasma conjunctivae in the eyes of healthy, free-ranging alpine ibex: possible involvement of alpine ibex as carriers for the main causing agent of infectious keratoconjunctivitis in wild Caprinae. Vet Microbiol 134:368–374
Ryser-Degiorgis MP, Ingold P, Tenhu H, Tebar-Less AM, Ryser A, Giacometti M (2002) Encounters between alpine ibex, alpine chamois and domestic sheep in the Swiss Alps. Hystrix 13:1–11
Saikku P (1992) The epidemiology and significance of Chlamydia pneumoniae. J Infect 25(Suppl 1):27–34
Salinas J, Caro MR, Vicente J, Cuello F, Reyes-Garcia AR, Buendía AJ, Rodolakis A, Gortázar C (2009) High prevalence of antibodies against Chlamydiaceae and Chlamydophila abortus in wild ungulates using two “in house” blocking-ELISA tests. Vet Microbiol 135:46–53
Salti-Montesanto V, Tsoli E, Papavassiliou P, Psarrou E, Markey BK, Jones GE, Vretou E (1997) Diagnosis of ovine enzootic abortion, using a competitive ELISA based on monoclonal antibodies against variable segments 1 and 2 of the major outer membrane protein of Chlamydia psittaci serotype 1. Am J Vet Res 58:228–235
Schneider J (2006) Zucht in Gehegen und Aussetzungen bis 1938. In: Giacometti M (ed) Von Königen und Wilderern – Die Rettung und Wiederansiedlung des Alpensteinbockes. Salm, Wohlen/Bern, pp 109–158
Taylor SK, Vieira VG, Williams ES, Pilkington R, Fedorchak SL, Mills KW, Cavender JL, Boerger-Fields AM, Moore RE (1996) Infectious keratoconjunctivitis in free-ranging mule deer (Odocoileus hemionus) from Zion National Park, Utah. J Wildl Dis 32(2):326–330
Teankum K, Pospischil A, Janett F, Brugnera E, Hoelzle LE, Hoelzle K, Weilenmann R, Zimmermann DR, Gerber A, Polkinghorne A, Borel N (2007) Prevalence of chlamydiae in semen and genital tracts of bulls, rams and bucks. Theriogenology 67:303–310
Tournut J, Lautie R, Geral F, Alcieu JP, Pluye JP (1985) Osservacioni e ricerche sulla cherato-congiuntivite del camoscio (Rupicapra rupicapra) dei pirinei. In: Balbo T, Lanfranchi P, Meneguz PG, Rossi L (eds) Proceeding of the Simposio Internazionale sulla cheratocongiuntivite infecttiva del camoscio. Amministrazione Provinciale di Vercelli, Vercelli, Italy, pp 41–52
Wilson K, Livingstone M, Longbottom D (2009) Comparative evaluation of eight serological assays for diagnosing Chlamydophila abortus infection in sheep. Vet Microbiol 135:38–45
Acknowledgements
This study was kindly supported by the Federal Veterinary Office (FVO), Bern, Switzerland (project 1.08.18). Collection of samples of Groups 1 and 2 was financed by the Federal Office of Environment (FOE). We are very grateful to the hunting authorities, game-wardens and hunters for the excellent collaboration and for providing numerous ibex samples. Samples of Group 3 were collected thank to the professional advice of Dr. med. vet. G. Brosi concerning the coordination of the sampling in canton of Grisons, and to the support of Dr. med. vet. G. Bearth and Dr. med. vet. U. Probst who provided the laboratory equipment during field work in this canton. We thank ID Vet diagnostics for the providing of ELISA kits. We are also grateful to Nadine Regenscheit, Mirjam Pewsner, Christian Willisch, Claudio Signer and numerous students and biologists at the FIWI for their assistance in sample collection. Many thanks to the laboratory technical staff of the Institute of Veterinary Pathology and of the Clinical Laboratory, Vetsuisse Faculty, University of Zurich.
Conflict of interest statement
None of the authors (NH, AP, NM, MPR, FM, JF, RT, NB) has a financial or personal relationship with other people or organisations that could inappropriately influence or bias the paper entitled “Alpine ibex (C. i. ibex) is not a reservoir for chlamydial infections of domestic ruminants and humans”.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by C. Gortázar
Rights and permissions
About this article
Cite this article
Holzwarth, N., Pospischil, A., Marreros, N. et al. Alpine ibex (Capra i. ibex) is not a reservoir for chlamydial infections of domestic ruminants and humans. Eur J Wildl Res 57, 233–240 (2011). https://doi.org/10.1007/s10344-010-0416-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10344-010-0416-7