Abstract
The aim of this study was to find alternatives (even partial) to mineral nitrogen fertilization to reduce its harmful accumulation in the edible parts of fennel. Fennel (Foeniculum vulgare) is a perennial plant of the Apiaceae family grown for its edible shoots, leaves, and seeds. The present work was conducted in two successive seasons, in 2018/2019 and 2019/2020, at the farm of the Agricultural Botany Department, Faculty of Agriculture, Ain Shams University, Cairo, Egypt, to investigate the efficiency of application of two biostimulants—Spirulina platensis algae extract (SP) at 10% and compost tea (CT) at 6.72 ml L−1—with partial doses (75 and 50%) of the recommended mineral nitrogen fertilizer dose on growth, photosynthetic pigments, fruit yield, and essential oil productivity. The results indicated that vegetative parameters, leaf number, branch number, shoot and bulb fresh weight, and shoot dry weight, as well as yield components such as 100-fruit weight and number of umbels per plant were significantly enhanced with application of SP as a foliar spray coupled with 75% of the recommended dose of nitrogen fertilizer (SP + 75% N) as compared to other treatments. Photosynthetic pigments and total soluble carbohydrate were improved significantly when CT was applied as soil drenching coupled with 75% of the recommended N dose (CT + 75% N). In this regard, a clear effect was noticed in terms of oil yield and fruit yield using SP and CT with a 75% N dose. It is concluded that using Spirulina platensis algae extract and compost tea as biostimulants coupled with only 75% of the recommended nitrogen fertilizer dose can be recommended for economical fennel production.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
During recent decades, adding nitrogen fertilizer to boost agricultural production has been a common practice around the world. However, mineral nitrogen produces high levels of greenhouse gas emissions, which exacerbates climate change and global warming (Guo et al. 2022). Nitrogen-based fertilizers in agriculture have contributed substantially to N2O emissions globally, with ~ 2% of fertilizer N returned to the atmosphere as N2O, which has a global warming potential that is 300 times greater than that of carbon dioxide (Park et al. 2012; Coskun et al. 2017; Thompson et al. 2019).
Biostimulants are defined as organic substances or microorganisms that increase nutrient uptake, promote growth, improve crop quality, and enhance plant tolerance to biotic and abiotic stress. They directly affect plant physiological and metabolic processes (El-Serafy 2015, 2019; Caradonia et al. 2018; Rezaei-Chiyaneh et al. 2019).
Fennel, Foeniculum vulgare, is one of the most important medicinal and aromatic plants of the Apiaceae family, grown for its edible bulblike stem base and the blanched shoots, and is also used in flavoring. The seeds and extracted oil are used in the medical, pharmaceutical, chemical, food, and agricultural industries (Basem 2012; Shabbara et al. 2018). The FAO (Food and Agriculture Organization of the United Nations) estimated the production of anises, star anise, fennel, and coriander at almost 2,274,700 billion tones in 2020 (Schurr et al. 2022). Fennel’s ripe and dry green-brown fruits, also known as seeds, are used alone or in preparations to cure a variety of ailments in folk remedies. They are also used to treat digestive and respiratory problems and have stimulant, sedative, lactagogue, and diuretic effects (Lucinewton et al. 2005; Ebeed et al. 2010; Mohamad et al. 2011; Figueredo et al. 2016).
Biostimulants can reduce the dependency on chemical fertilizers in the agricultural sector. They potentially mitigate climate change-induced stress and limit eutrophication (Hunter et al. 2017). Biostimulants can promote plant physiological processes (enhancement of crop nutrition, stress tolerance, yield quantity or quality) without harming the surrounding environment (Barone et al. 2018). One biostimulant is the water extract of compost, termed “compost tea” (CT). CT retains all the beneficial soluble bioactive components of compost, making it a potent source of plan-stimulatory and defensive compounds. CTs contain a significant quantity of total nutrients, with the majority being primary macronutrients. CTs have higher levels of humic substances and mineral nutrients than the composts from which they are formed (Ingham 1999; Eudoxie and Martin 2019). CT is used as a plant stimulant for improving growth, productivity, and phytopathogen control.
Spirulina platensis algae (SP) has also been suggested as an effective alternative to synthetic agricultural fertilizers. The extract of Spirulina platensis is a common commercial biostimulant. It has a high capacity to enhance plant growth and development, which consequently improves crop development and productivity (Dias et al. 2016).
This study aimed to investigate the behavior of fennel plant’s growth and yield as a consequence of reducing mineral nitrogen doses with SP algae and CT extracts.
Materials and Methods
Plant Material and Agricultural Practices
This research was carried out in the 2018/2019 and 2019/2020 seasons, at the experimental farm of the Agricultural Botany Department, Faculty of Agriculture, Ain Shams University, Cairo, Egypt (30° 06′ 42″ N 31° 14′ 46″ E). Seeds of fennel (Foeniculum vulgare) sp. dulce Mill were obtained from SEKEM company, Egypt. Seeds were planted September 15 and 19 in foam trays filled with a mixture of peat moss:vermiculite (1:1 v:v) in both the first and the second seasons. Thirty days after planting, seedlings were transplanted into the open field and planted on hills at a distance of 35 cm between hills and 60 cm within rows (soil and water analysis is shown in Tables 1, 2 and 3). After 5 weeks, seedlings were thinned to one plant per hill. Soil received 21,428 kg ha−1 compost (Table 4) before plantation and the surface layer of all plots was mixed with calcium superphosphate (15.5% P2O5) at the rate of 142.85 kg ha−1 (Wafaa et al. 2017). The experimental plot was divided into plots of 12 m2 (3.0 × 4.0 m) to include four rows each. The experimental design was a simple system in randomized complete blocks with three replicates. The levels of nitrogen were applied in plots at the rate of 385.5 kg ha−1. Ammonia nitrate (33.5% NH4NO3) was used as nitrogen fertilizer, with the full recommended dose of nitrogen fertilizer as a control (Ctr). Treatments were performed for a total period of 7 weeks (three times per season, after 21, 45, and 65 days of transplanting). All treatments received a dose of potassium sulfate (48% K2O) as a source of potassium at the rate of 95.23 K2O kg ha−1 split into two equal doses 36 and 57 days after transplanting and applied beside the plants.
Biostimulant Application
Compost Tea Extract
The compost used in preparing the CT was produced by the Saline Agriculture Unit of the Faculty of Agriculture, Ain Shams University, Egypt, by adopting routine standard methods. The obtained chemical analysis of the used compost is recorded in Table 5. CT was prepared by soaking 12 kg of fresh mature compost in 30 L of water at room temperature (25 ± 2 °C) for 4 days with aeration to ensure plenty of oxygen in the mixture as it composes. Thereafter, 50 ml sugarcane molasses was added according to the method of Ingham (2005), with modifications described by Hegazy et al. (2015). After CT had been filtered through muslin cloth, representative samples were taken before each treatment and analyzed for determination of various chemical characteristics (Table 4). CT was applied as soil drenching at a concentration of 6.7 ml L−1 for each treated plot. Fennel plants received CT application three times per season, after 21, 45, and 65 days of transplanting.
Spirulina Platensis Algae Extract
Spirulina platensis (SP) algae extract was obtained from the Center of Genetic Engineering and Biotechnology (ACGEB), Ain Shams Univ, Egypt, with a concentration of 10%. Before treatment, representative samples of the SP algae extract were taken for chemical characteristic determination (Table 6). Fennel plants were foliar sprayed three times per season after 21, 45, and 65 days from transplanting, with a concentration of 10 ml/L for each treated plot.
Treatments and Experimental Design
The experimental design was arranged in simple design in randomized complete blocks of five treatments with three replicates each. The treatments were arranged in plots as follows:
-
CT + 75% of recommended dose of N (CT + 75% N)
-
CT + 50% of recommended dose of N (CT + 50% N)
-
SP + 75% of recommended dose of N (SP + 75% N)
-
SP + 50% of recommended dose of N (SP + 50% N)
Additionally, a control plot (Ctr) received the full recommended dose of nitrogen fertilizer (100%) without any CT or SP treatment during either season.
Growth Attributes
Two successive samples were taken for both seasons, the first one at vegetative growth 90 days after transplanting, the second sample at the harvest stage (yield) 140 days after transplanting. Growth parameters of plant height (cm) and fresh and dry weight of shoot (g) were recorded at the harvest stage for both seasons.
Fruit Yield Measurements
Yield parameters studied in this experiment were 100-fruit weight (g), umbels number per plant, and fruit yield (kg ha−1).
Essential Oil Determination
Fennel essential oil percentage was extracted from air-dried mature fruit by hydro distillation for 3 h using a modified Clevenger apparatus according to the British Pharmacopoeia (1963)
Photosynthetic Pigments
Samples of fennel leaves (2–6) were randomly collected at vegetative growth of each treatment separately for chlorophyll determination, as described by Moran (1982). Fresh leaves (100 mg) were ground with 10 ml N,N-dimethyl formamide and the extractions were then incubated in a dark fridge overnight before analysis using a UV-VIS spectrophotometer (Model CT200; Randolph, NJ, USA). According to Lichtenthaler and Wellburn (1983), the data were expressed as mg/g fresh weight.
Chlorophyll a, b, and carotenoid concentrations were measured at wavelengths of 664, 647, and 470 nm, respectively, and calculated and expressed in mg g−1 FW using the formula of Shlyk (1971).
Amino Acids Determination
Amino acids in fennel bulbs were measured following the colorimetric method according to Jayaraman (1985) using the ninhydrin reaction. Amino acids in fennel bulbs were extracted by soaking 1 g of sample in 80% ethanol. Thereafter, 1 ml of sample extract was added to 3 ml distilled water and mixed well with 1 ml of ninhydrin reagent. The mixture was heated in a water bath for 15 min. After cooling, 50% ethanol was added to reach a volume of 10 ml and the absorbance was determined at 570 nm. Total amino acid concentration was standardized using glycine.
Total Soluble Carbohydrates Determination
Total soluble carbohydrates concentration in bulbs was assessed as recommended by Hedge and Hofreiter (1962) using the anthrone reagent method. Total soluble carbohydrates were extracted by hydrolysis of 0.1 g of dried sample with 2.5 N HCl in a water bath for 3 h, then the pH was adjusted to 7 and distilled water added to 100 ml. Taking 1 ml of sample extract with 4 mL of frozen anthrone reagent, this was heated in a water bath followed by cooling, and the developed green color was measured at 630 nm.
Statistical Analysis
Data were statistically analyzed using analysis of variance (ANOVA) with the CoStat program. Means were compared using Duncan’s test at the P ≤ 0.05 level. Results were presented as means ± standard deviation (SD).
Results
Plant Growth
In order to reduce the recommended dose of mineral nitrogen fertilizer applied to fennel plants, two biostimulants (Spirulina platensis, SP, and compost tea, CT) were used with different N doses (75 and 50% of the recommended dose). Growth characteristics of fennel plants treated with these biostimulants are presented in Fig. 1 and Tables 7 and 8. Generally, the data showed that plant height (cm) was not significantly affected by reducing the N dose with different treatments. Meanwhile, the number of leaves showed a significant increase in plants that received 75% N and were treated with both SP and CT as compared to control plants (100% N). The branch number in plants treated with SP + 75% N was higher than the number of branches in all other treatments. The same trend was found in both fresh and dry weight of fennel shoots, as treating plants with 75% + SP increased plant weight. Although the bulb fresh weight showed significant increment in plants receiving 75% N + SP when compared with control and other treatments, the bulb dry weight was hardly affected by treatments.
Effect of different nitrogen doses with Spirulina platensis (SP) or compost tea (CT) on some vegetative growth characteristics of fennel plants during the first season. Ctr control. Each column represents the mean of three replicates ± SD. Significant differences (P ≤ 0.05) between treatments are indicated by different letters
Fruit Characteristics and Total Yield
Like plant growth parameters, fruit yield was also clearly changed and affected by the application of biostimulants in comparison to the control. The weight of 100 fruits and fruit yield per hectare were significantly improved with application of 75% N and SP and 75% N and CT compared to control at a rate of 28.12 and 10.41% for 100-fruit weight and 16.95 and 5.25% for fruit yield, respectively. Also, 75% N and SP significantly increased the number of umbels per plant compared with control; this increase was estimated at 21.83% (Table 9; Fig. 2).
Effect of different nitrogen doses with Spirulina platensis (SP) or compost tea (CT) on some yield parameters at the mature stage of fennel plants during the first season. Ctr control. Each column represents the mean of three replicates ± SD. Significant differences (P ≤ 0.05) between treatments are indicated by different letters
Essential Oil
Essential oil percentage and characteristics were not improved by biostimulant application (Fig. 3). Foliar spray of SP + 75% N and soil drenching with CT + 75% N gave a similar total essential oil percentage to control plants.
Photosynthetic Pigments
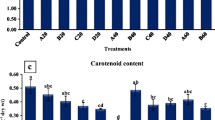
The data presented in Fig. 4 indicate that the concentrations of photosynthetic pigments (chlorophyll a, b, and carotenoids) recorded their highest values when plants received 75% N + CT. However, statistically, control plants have the same photosynthetic pigment concentration values as 75% + CT. Other treatments did not elicit any significant improvements, except plants treated with 50% N + CT, which showed a significant increase in chlorophyll b concentration.
Effect of different nitrogen doses with Spirulina platensis (SP) or compost tea (CT) on chlorophyll a, chlorophyll b, and carotenoids at the vegetative growth stage of fennel plants during the first season. Ctr control. Each column represents the mean of three replicates ± SD. Significant differences (P ≤ 0.05) between treatments are indicated by different letters
Total Soluble Carbohydrate
The results presented in Fig. 5 showed that there was a significant increase in total soluble carbohydrates percentage when plants received 75% N + CT (37.59%), while control plants showed no significant difference to plants with 50% N + CT which followed the control plants (100% N). The lowest soluble carbohydrates percentage was recorded with plants treated with SP + 50% N.
Effect of different nitrogen doses with Spirulina platensis (SP) or compost tea (CT) on concentrations of total soluble carbohydrates at the vegetative growth stage of fennel plants during the first season. Ctr control. Each column represents the mean of three replicates ± SD. Significant differences (P ≤ 0.05) between treatments are indicated by different letters
Amino Acids
Figure 6 also indicates that the highest amino acid percentage was obtained by adding 75% N + SP, while plants receiving 50% N + SP showed no significant difference to control plants and both have a higher respective percentage of amino acids than CT treatments.
Effect of different nitrogen doses with Spirulina platensis (SP) or compost tea (CT) on concentrations of amino acids at the vegetative growth stage of fennel plants during the first season. Ctr control. Each column represents the mean of three replicates ± SD. Significant differences (P ≤ 0.05) between treatments are indicated by different letters
Discussion
Despite the necessity of nitrogen fertilizers, the dependency on mineral N fertilizers (annually, nearly 120 million tons of nitrogen are applied to agricultural fields) has shifted the natural balance, leading to many damaging and long-lasting environmental and ecological consequences (FAOSTAT 2017). Finding alternative natural resources, even for partial substitution of mineral nitrogen fertilizers, aims to preserve soil and atmospheric components and lead to environmental sustainability.
Blue-green algae (Spirulina platensis) plays a key role in improving the growth of many plants when applied as a biostimulant. This evidence was clearly shown in the growth criteria of fennel plants that received low N doses and treatment with biostimulants. CTs contain a significant quantity of total nutrients, with the majority being primary macronutrients. CT shows a similar trend to SP when applied to plants receiving low, partial N doses. Regardless of the purpose of CT use, its positive effects on crop growth and soil fertility, whilst controlling pest and disease, make it a contemporary sustainable tool aligned to organic agriculture (Fig. 7).
Biostimulant applications clearly enhanced the growth and yield of fennel plants under the lower-than-recommended doses of nitrogen fertilizer (75%). The potential of biostimulants as an alternative to chemical fertilizers has been reported by many scholars due to their ability to increase nutrient-use efficiency in plants without causing damage to the environment (Arnau and Richard 2016; Barone et al. 2018; Kopta et al. 2018). Results have indicated that foliar application of SP extract significantly improved fennel growth and fruit yield when applied with 75% of the recommended N dose in comparison to the control. The stimulatory effect of foliar spray of SP could be due to its high content of macro- and microelements as well as the high content of free amino acids, which can stimulate plant metabolism and thus raise the synthesis of proteins and some hormones responsible for plant growth (Mógor et al. 2018) to directly improve plant growth.
The algae extract application promoted fennel plant performance in both seasons, as represented by plant height and fresh and dry weights. SP has many beneficial effects on plants as it contains the growth-promoting hormones indole-3-acetic acid (IAA) and cytokinin, trace elements of Fe, Cu, Zn, Co, Mo, Mn, and Ni, and vitamins (Challen and Hemingway 1966).
Similarly, the number umbels per plant as well as fruit and oil percentage showed a significant increase with SP + 75% N, which might be attributed to the particular molecular structure of SP extract which has an important and vital role in increasing plant production and increasing the capacity of the plant to resist various stress conditions (Aly and Esawy 2008; Ogbonda et al. 2007; Anitha et al. 2016). These results were also confirmed by Wafaa et al. (2017), who found that foliar spray of SP extract on fennel plants had a significant and marked effect on growth parameters, fruit characteristics, and oil percentage as compared to untreated plants. On the other hand, CT has the capacity to stimulate plant growth and productivity via plant nutrition and hormone-like activities. The results of the current study show no significant effect on plant productivity, including number of umbels/plant, 100-fruit weight, fruit yield, and oil percentage in plants receiving CT with 75% of the recommended N dose in comparison to control plants that received the full recommended dose of N. These results may be due to the ability of water-based CT to enhance the uptake and accumulation of nutrient elements in the plant (Rodríguez-Ortíz et al. 2006) as a result of its content of organic extracts which develop the root system of plants and improve nutrient uptake (Abd El-Kader and El-Shaboury 2013; Meshref et al. 2010). Moreover, similar to compost, CT can also improve soil quality by altering the physicochemical properties, water-holding capacity, and biodiversity of soil (Scheuerell and Mahaffee 2002), which leads to improved plant productivity.
The stimulatory effect of blue-green alga extract on biochemical components of fennel could be due to its high content of macro- and microelements. However, micronutrient fertilizer only has the effect of encouraging roots to absorb more nutrients from the soil medium, as mentioned by (Shaaban and Mubarak 2000). Also, SP extract contains a high percentage of amino acids (about 62% free amino acids), essential oils, fiber, protein, and ash content in seeds (Ehsanipour et al. 2012). Blue-green algae extract contains the whole spectrum of natural mixed carotene and xanthophyll phytopigments, which are considered as the richest natural source of vitamin B12.
Eudoxie and Martin (2019) reported that CT enhanced soil quality through increased microbial diversity and nutrient availability to increase crop growth and, importantly, yield. Several mechanisms have been postulated for the altered effects associated with CT, including the increased availability and uptake of nutrients. Secondary mechanisms include increased soil organic matter and nutrient turnover via microbial activity. Stimulatory effects occur in plants through plant growth regulators, humic substances, and other biostimulatory compounds present in CTs. Further benefit is derived through the suppression of plant pathogens, which provides the best opportunity for maximum growth.
Conclusion
It can be concluded that using biostimulants (Spirulina platensis or compost tea) to reduce mineral nitrogen doses may improve or preserve the vegetative characteristics and yield components of fennel plant. Adding SP or CT to fennel plants not only enables reduction of nitrogen dose but also improves plant nutritional status and essential oil content. Reducing mineral N fertilizers decreases the accumulation of nitrogen derivatives in the edible parts of fennel plant and maintains the sustainability of soil and environment. These results lead to the conclusion that addition of SP or CT saved about 25% of the recommended nitrogen fertilizer dose and enhanced fennel yield quantity and quality. Therefore, farmers could reduce the agricultural cost by this means.
References
Abd El-Kader MG, El-Shaboury MG (2013) Evaluation of soaking and foliar methods with compost tea, humic acid and bio-fertilizer on soil fertility and faba bean yield productivity and quality under saline soil condition. Minufiya J Agric Res 6:1663–1675
Aly MS, Esawy MA (2008) Evaluation of Spirulina platensis as bio stimulator for organic farming systems. J Genet Eng Biotechnol 6:1–7
Anitha L, Bramari GS, Kalpana P (2016) Effect of supplementation of Spirulina platensis to enhance the zinc status in plants of Amaranthus gangeticus, Phaseolus aureus and tomato. Adv Biosci Biotechnol 7:289–299
Arnau L, Richard H (2016) Techno-economic feasibility study for the production of microalgae based plant biostimulant. KTH Royal Institute of Technology
Barone V, Baglieri A, Stevanato P, Broccanello C, Bertoldo G, Bertaggia M, Cagnin M, Pizzeghello D, Moliterni VM, Mandolino G, Fornasier F, Squartini A, Nardi S, Concheri G (2018) Root morphological and molecular responses induced by microalgae extracts in sugar beet (Beta vulgaris L.). J Appl Phycol 30:1061–1071
Basem DH (2012) Production of fennel and cumin crops in Asyut Governorate. Assiut J Agric Sci 43:1–15
British Pharmacopoeia B (1963) Determination of volatile oil in drugs
Caradonia F, Battaglia V, Righi L, Pascali G, La Torre A (2018) Plant biostimulant regulatory framework: prospects in Europe and current situation at international level. J Plant Growth Regul 38:438–448
Challen SB, Hemingway JC (1966) Growth of higher plants in response to feeding with seaweed extracts. In: Proceedings of the Fifth International Seaweed Symposium, Halifax, August 1965, vol 17. Academic Press, New York, pp 356–367
Coskun D, Britto DT, Shi W, Kronzucker HJ (2017) How plant root exudates shape the nitrogen cycle. Trends Plant Sci 22:661–673
Dias GA, Rocha RH, Araújo JL, de Lima JF, Guedes WA (2016) Growth, yield, and postharvest quality in eggplant produced under different foliar fertilizer (Spirulina platensis) treatments. SCA 37:3893–3901
Ebeed NM, Abdou HS, Booles HF, Salah SH, Ahmed ES, Fahmy K (2010) Antimutagenic and chemoprevention potentialities of sweet fennel (Foeniculum vulgare Mill.) hot water crude extract. J Am Sci 6:831–842
Ehsanipour A, Razmjoo J, Zeinali H (2012) Effect of nitrogen rates on yield and quality of fennel (Foeniculum vulgare Mill.) accessions. Ind Crops Prod 35:121–125
El-Serafy RS (2015) Effect of silicon and calcium on productivity and flower quality of carnation. Doctoral dissertation, Tanta University
El-Serafy RS (2019) Silica nanoparticles enhance sphysio-biochemical characters and postharvest quality of Rosa hybrid L. cut flowers. J Hortic Res 27:47–54. https://doi.org/10.2478/johr-2019-0006
Eudoxie G, Martin M (2019) Compost tea quality and fertility. In: Organic fertilizers-history, production and applications. IntechOpen, Rijeka, pp 79–104 (chapter 5)
FAOSTAT (2017) World fertilizer trends and outlook to 2020
Figueredo G, Özcan MM, Al Juhaimi F, Chalchat JC, Özcan MM, Chalard P (2016) The effect of heights on chemical composition of essential oil of bitter fennel (Foeniculum vulgare subsp. piperitum) fruits. J Essent Oil Bear Plants 19:1273–1276
Guo C, Liu X, He X (2022) A global meta-analysis of crop yield and agricultural greenhouse gas emissions under nitrogen fertilizer application. Sci Total Environ 831:154982
Hegazy WI, Hussein ET, Ali AS (2015) Improving physico-chemical and microbiological quality of compost tea using different treatments during extraction. Afr J Microbiol Res 9:763–770
Hedge JE, Hofreiter BT (1962) In: Whistler RL, Be Miller JN (eds) Carbohydrate chemistry 17. Academic Press, New York
Hunter MC, Smith RG, Schipanski ME, Atwood LW, Mortensen DA (2017) Agriculture in 2050: recalibrating targets for sustainable intensification. BioScience 67:386–391
Ingham ER (1999) What is compost tea? Part 1. BioCycle 40:74–75
Ingham ER (2005) The compost tea brewing manual, 5th edn. Soil Food Web, Corvallis, p 79
Jayaraman J (1985) Laboratory manual in biochemistry vol 107. Wiley Eastern, New Delhi, pp 75–76
Kopta T, Pavlikova M, Sękara A, Pokluda R, Maršálek B (2018) Effect of bacterial-algal biostimulant on the yield and internal quality of lettuce (Lactuca sativa L.) produced for spring and summer crop. Not Bot Horti Agrobo 46:615–621
Lichtenthaler HK, Wellburn AR (1983) Determinations of total carotenoids and chlorophylls a and b of leaf extracts in different solvents. Biochem Soc Trans 11:591–592
Lucinewton S, Raul N, Carvalho J, Mirian B, Lin C, Angela A (2005) Supercritical fluid extraction from fennel (Foeniculum vulgare), global yield, composition and kinetic data. J Supercrit Fluids 35:212–219
Meshref HA, Rabie MH, El-Ghamry AM, El-Agamy AM (2010) Maximizing utilization of compost addition using foliar compost extract and humic substances in alluvial soil. J Soil Sci Agric Eng 1:957–971
Mógor ÁF, Ördög V, Lima GP, Molnár Z, Mógor G (2018) Biostimulant properties of cyanobacterial hydrolysate related to polyamines. J Appl Phycol 30:453–460
Mohamad RH, El-Bastawesy AM, Abdel-Monem MG, Noor AM, Al-Mehdar HA, Sharawy SM, El-Merzabani MM (2011) Antioxidant and anticarcinogenic effects of methanolic extract and volatile oil of fennel seeds (Foeniculum vulgare). J Med Food 14:986–1001
Moran R (1982) Formulae for determination of chlorophyllous pigments extracted with N, N‑dimethylformamide. Plant Physiol 69:1376–1381
Ogbonda KH, Aminigo RE, Abu GO (2007) Influence of temperature and pH on biomass production and protein biosynthesis in a putative Spirulina sp. Bioresour Technol 98:2207–2211
Park S, Croteau P, Boering K, Etheridge D, Ferretti D, Fraser P, Kim KR, Krummel P, Langenfelds R, Van Ommen T (2012) Trends and seasonal cycles in the isotopic composition of nitrous oxide since 1940. Nat Geosci 5:261–265
Rezaei-Chiyaneh E, Amirnia R, Machiani MA, Javanmard A, Maggi F, Morshedloo MR (2019) Intercropping fennel (Foeniculum vulgare L.) with common bean (Phaseolus vulgaris L.) as affected by PGPR inoculation: a strategy for improving yield, essential oil and fatty acid composition. Sci Hortic 261:108951. https://doi.org/10.1016/j.scienta.2019.108951
Rodríguez-Ortíz JC, Valdez-Cepeda RD, Lara-Mireles JL, Rodríguez-Fuentes H, Vázquez-Alvarado RE, Magallanes-Quintanar R, García-Hernández JL (2006) Soil nitrogen fertilization effects on phytoextraction of cadmium and lead by tobacco (Nicotiana tabacum L.). Bioremediat J 10:105–114
Scheuerell S, Mahaffee W (2002) Compost tea: principles and prospects for plant disease control. Compost Sci Util 10:313–338
Schurr L, Masotti V, Geslin B, Gachet S, Mahé P, Jeannerod L, Affre L (2022) To what extent is fennel crop dependent on insect pollination. Agric Ecosyst Environ 338:108047
Shaaban M, Mubarak ZM (2000) Effect of some green plant materials as soil additives on soil nutrient availability, growth, yield and yield components of faba bean plants. J Plant Prod 25:2005–2016
Shabbara MH, Karima AM, Yousria AE, Eman AA (2018) Comparative analytical economic study of the high yield varieties production of Foeniculum vulgare Mill. (Fennel) between Arabic Republic of Egypt and India. Middle East J Agric Res 7:1514–1520
Shlyk AA (1971) Determination of chlorophylls and carotenoids in extracts of green leaves. In: Pavlina OA (ed) Biokhimicheskie metody v fiziologii rastenii [Biochemical methods in plant physiology]. Nauka, Moscow, RU, pp 154–170
Thompson R, Lassaletta L, Patra P, Wilson C, Wells K, Gressent A, Koffi E, Chipperfield M, Winiwarter W, Davidson E (2019) Acceleration of global N2O emissions seen from two decades of atmospheric inversion. Nat Clim Chang 9:993–998
Wafaa AE, Hendawy SF, Hamed ES, Toaima WI (2017) Effect of planting dates, organic fertilization and foliar spray of algae extract on productivity of Dutch fennel plants under
Funding
Open access funding provided by The Science, Technology & Innovation Funding Authority (STDF) in cooperation with The Egyptian Knowledge Bank (EKB).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
A.A. Shawky, G.S. Khalifa, A. Hegazi, and M. ElSherif declare that they have no competing interests.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Shawky, A.A., Khalifa, G.S., Hegazi, A. et al. Growth, Productivity, and Essential Oil Content of Fennel Plants Treated with Spirulina Platensis Extract and Compost Tea Under Low Nitrogen Doses. Gesunde Pflanzen 75, 2899–2908 (2023). https://doi.org/10.1007/s10343-023-00870-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10343-023-00870-z