Abstract
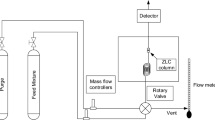
The system constants of the solvation parameter model are used to prepare system maps for the retention of small neutral compounds on an ethyl-bridged, ocatadecylsiloxane-bonded superficially porous silica stationary phase (Kinetex EVO C18) for aqueous mobile phases containing 10–70% (v/v) methanol or acetonitrile. Electrostatic interactions (cation-exchange) are important for the retention of weak bases with acetonitrile–water but not methanol–water mobile phase compositions. Compared with a superficially porous octadecylsiloxane-bonded silica stationary phase (Kinetex C18) with a similar morphology but different topology statistically significant differences in selectivity at the 95% confidence level are observed for neutral compounds that vary by size and hydrogen-bond basicity with other intermolecular interactions roughly similar. These selectivity differences are dampened with acetonitrile–water mobile phases, but are significant for methanol–water mobile phase compositions containing <30% (v/v) methanol. A comparison of a totally porous ethyl-bridged, octadecylsiloxane-bonded silica stationary phase (XBridge C18) with Kinetex EVO C18 indicated that they are effectively selectivity equivalent.





Similar content being viewed by others
References
Fanali S, Haddad PR, Poole CF, Riekkola M-L (eds) (2017) Liquid chromatography: fundamentals and instrumentation, 2nd edn. Elsevier, Amsterdam
Snyder LR, Kirkland JJ, Dolan JW (2009) Introduction to modern liquid chromatography, 3rd edn. Wiley-Blackwell, Hoboken
Kirkland JJ (2004) Development of some stationary phases for reversed-phase high-performance liquid chromatography. J Chromatogr A 1060:9–21
Davies NH, Euerby MR, McCalley DV (2008) Analysis of basic compounds by reversed-phase high-performance liquid chromatography using hybrid inorganic/organic phases at high pH. J Chromatogr A 1178:71–78
McCalley DV (2010) The challenges of the analysis of basic compounds by high performance liquid chromatography: some possible approaches for improved separations. J Chromatogr A 1217:858–880
Wyndham KD, O’Gara JE, Walter TH, Glose KH, Lawrence NL, Alden BA, Izzo GS, Hudalla CJ, Irenata PC (2003) Characterization and evaluation of C18HPLC stationary phase based on ethyl-bridged hybrid organic/inorganic particles. Anal Chem 74:6781–6788
O’Gara JE, Wyndham KD (2006) Porous hybrid organic-inorganic particles in reversed-phase liquid chromatography. J Liq Chromatogr Relat Technol 29:1025–1045
Yu H, Jia CY, Wu HB, Song GJ, Jin Y, Ke HX, Liang XM (2012) Highly stable high performance liquid chromatography stationary phases based on direct chemical modification of organic bridges in hybrid silica. J Chromatogr A 1247:63–70
Imran A, Al-Othman ZA, Nagae N, Gaitonde VD, Dutta KK (2012) Recent trends in ultra-fast HPLC. New generation superficially porous silica columns. J Sep Sci 35:3235–3249
Atapattu SN, Poole CF, Praseuth MB (2016) System maps for retention of small neutral compounds on a superficially porous particle column in reversed-phase liquid chromatography. J Chromatogr A 1468:250–256
Kiridena W, Poole CF, Atapattu SN, Qian J, Koziol WW (2007) Comparison of the separation characteristics of the organic-inorganic hybrid octadecyl stationary phases XTerra MS C18 and XBridge C18 and Shield RP18 in RPLC. Chromatographia 66:453–460
Buntz S, Figus M, Liu Z, Kazakevich YV (2012) Excess adsorption of binary aqueous organic mixtures on various reversed-phase packing materials. J Chromatogr A 1240:104–112
Poole CF (2015) An interphase model for retention in liquid chromatography. J Planar Chromatogr 28:98–105
Poole CF, Lenca N (2017) Applications of the solvation parameter model in reversed-phase liquid chromatography. J Chromatogr A 1486:2–19
Poole CF, Poole SK (2002) Column selectivity from the perspective of the solvation parameter model. J Chromatogr A 965:263–299
Abraham MH, Ibrahim A, Zissmos AM (2004) Determination of sets of solute descriptors from chromatographic measurements. J Chromatogr A 1037:29–47
Poole CF, Atapattu SN, Poole SK, Bell AN (2009) Determination of solute descriptors by chromatographic methods. Anal Chim Acta 652:32–53
Poole CF, Ariyasena TC, Lenca N (2013) Estimation of the environmental properties of compounds from chromatographic measurements and the solvation parameter model. J Chromatogr A 1317:85–104
Poole CF, Poole SK (2009) Foundations of retention in partition chromatography. J Chromatogr A 1216:1530–1550
Poole CF, Ahmed H, Kiridena W, DeKay C, Koziol WW (2005) Contribution of steric repulsion to retention on an octadecylsiloxane-bonded silica stationary phase in reversed-phase liquid chromatography. Chromatographia 62:553–561
Carr PW, Dolan JW, Neue UD, Snyder LR (2011) Contributions to reversed-phase column selectivity. 1. Steric interaction. J Chromatogr A 1218:1724–1742
McDonald PD (2003) Improving our understanding of the reversed-phase separations for the 21st century. Adv Chromatogr 42:323–375
Acknowledgements
Antonia Napoli (Phenomenex) is acknowledged for providing the columns used in this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Authors Atapattu and Poole have no conflict of interest. Author Praseuth is an employee of Phenomenex who manufactured the column used in this study. Authors Atapattu and Poole received no financial support from Phenomenex for this study.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Rights and permissions
About this article
Cite this article
Atapattu, S.N., Poole, C.F. & Praseuth, M.B. System Maps for Retention of Small Neutral Compounds on a Superficially Porous Ethyl-Bridged, Octadecylsiloxane-Bonded Silica Stationary Phase in Reversed-Phase Liquid Chromatography. Chromatographia 80, 1279–1286 (2017). https://doi.org/10.1007/s10337-017-3350-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10337-017-3350-y