Abstract
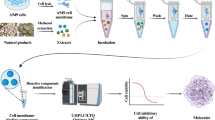
For the first time, a novel biological affinity separating system called lipid raft stationary phase chromatography (LRSC) was developed. It was employed to screen bioactive components from Chinese gallnut, a traditional Chinese medicine (TCM). The LRSC was prepared by the addition of activated silica to Tris–HCl solution, which contains the isolated lipid rafts of U251 cells. This was followed by agitation, washing, centrifugation and then re-suspension of the residue in another Tris–HCl solution. The lipid rafts possess abundant receptor tyrosine kinase, specifically tropomyosin-related kinase A (TrkA), which is a widely researched anti-tumor drug target. Thus, TrkA provided the LRSC model with the ability to select fractions that specifically interact with it. Using a non-TrkA targeted anti-tumor drug (gemcitabine) and TrkA targeted anti-tumor drugs (lestaurtinib and gefitinib) as controls to evaluate the specific affinity of the LRSC column, the different fractions of Chinese gallnut were subjected to LRSC screening for the identification of anti-tumor components. As a result, the ether fraction of Chinese gallnut manifested desirable affinity properties. The methyl thiazolyl tetrazolium assay confirmed the anti-tumor effect of the screened ether fraction, and more importantly, the ether fraction failed woefully to exhibit its anti-proliferative activity in the presence of TrkA inhibitors (K252a and primary antibody). This further proves the selectivity of LRSC on TrkA-targeted drugs. The LRSC model has, therefore, shown to be of high efficiency and selectivity in screening bioactive components from the complex TCM extracts, thus offering an effective approach for the development of anticancer natural products.





Similar content being viewed by others
References
Huang X, Kong L, Li X, Chen X, Guo M, Zou H (2004) Strategy for analysis and screening of bioactive compounds in traditional Chinese medicines. J Chromatogr B 812:71–84
Su X, Hu L, Kong L, Lei X, Zou H (2007) Affinity chromatography with immobilized DNA stationary phase for biological fingerprinting analysis of traditional Chinese medicines. J Chromatogr A 1154:132–137
Noctor TAG, Wainer IW, Hage DS (1992) Allosteric and competitive displacement of drugs from human serum albumin by octanoic acid, as revealed by high-performance liquid affinity chromatography, on a human serum albumin-based stationary phase. J Chromatogr B 577:305–315
Vuignier K, Guillarme D, Veuthey JL, Carrupt PA, Schappler J (2013) High performance affinity chromatography (HPAC) as a high-throughput screening tool in drug discovery to study drug–plasma protein interactions. J Pharm Biomed Anal 74:205–212
Zhang C, Li J, Xu L, Shi ZG (2012) Fast immobilized liposome chromatography based on penetrable silica microspheres for screening and analysis of permeable compounds. J Chromatogr A 1233:78–84
Beigi F, Lundahl P (1999) Immobilized biomembrane chromatography of highly lipophilic drugs. J Chromatogr A 852:313–317
Moaddel R, Musyimi HK, Sanghvi M, Bashore C, Frazier CR, Khadeer M, Bhatia P, Wainer IW (2010) Synthesis and characterization of a cellular membrane affinity chromatography column containing histamine 1 and P2Y1 receptors: a multiple G-protein coupled receptor column. J Pharm Biomed Anal 52:416–419
Habicht KL, Frazier C, Singh N, Shimmo R, Wainer IW, Moaddel R (2013) The synthesis and characterization of a nuclear membrane affinity chromatography column for the study of human breast cancer resistant protein (BCRP) using nuclear membranes obtained from the LN-229 cells. J Pharm Biomed Anal 72:159–162
He L, Wang S, Geng X (2001) Coating and fusing cell membranes onto a silica surface and their chromatographic characteristics. Chromatographia 54:71–76
He L, Yang G, Geng X (1999) Enzymatic activity and chromatographic characteristics of the cell membrane immobilized on silica surface. Chin Sci Bull 44:826–831
Hou X, Zhou M, Jiang Q, Wang S, He L (2009) A vascular smooth muscle/cell membrane chromatography–offline-gas chromatography/mass spectrometry method for recognition, separation and identification of active components from traditional Chinese medicines. J Chromatogr A 1216:7081–7087
Munro S (2003) Lipid rafts: elusive or illusive? Cell 115:377–388
Li YC, Park MJ, Ye SK, Kim CW, Kim YN (2006) Elevated levels of cholesterol-rich lipid rafts in Cancer cells are correlated with apoptosis sensitivity induced by cholesterol-depleting agents. Am J Pathol 168:1107–1118
Patra SK (2008) Dissecting lipid raft facilitated cell signaling pathways in cancer. Biochim Biophys Acta 1785:182–206
Harris TJC, Siu CH (2002) Reciprocal raft-receptor interactions and the assembly of adhesion complexes. BioEssays 24:996–1003
Simons K, Toomre D (2000) Lipid rafts and signal transduction. Nat Rev Mol Cell Biol 1:31–39
Zhuang L, Kim J, Adam RM, Solomon KR, Freeman MR (2005) Cholesterol targeting alters lipid raft composition and cell survival in prostate cancer cells and xenografts. J Clin Invest 115:959–968
Pike LJ (2003) Lipid rafts. J Lipid Res 44:655–667
Mollinedo F, Gajate C (2006) Fas/CD95 death receptor and lipid rafts: new targets for apoptosis-directed cancer therapy. Drug Resist Update 9:51–73
Ren Y, Zhou X, Mei M, Yuan XB, Han L, Wang GX, Jia ZF, Xu P, Pu PY, Kang CS (2010) MicroRNA-21 inhibitor sensitizes human glioblastoma cells U251 (PTEN-mutant) and LN229 (PTEN-wild type) to taxol. BMC Cancer 10:27
Zhang D, Yuan BX, Deng XL, Yang GD, He LC (2008) Comparative Studies on Cell Membrane Chromatography with Rat Aorta T issue, Cultured Rat Aorta SmoothMuscle Cell and A1D SubtypesOver Expressed Cell. Chin Pharm J 43:1710–1713
Sariozlu NY, Kivanc M (2011) In: Victor RP, Ronald Ross W, Vinood RRW, PatelA2-Victor B, Preedy R, Vinood BP (eds.) Nuts and Seeds in Health and Disease Prevention, 60th edn. Academic Press, San Diego
Nishizawa M, Yamagishi T, Nonaka GI, Nishioka I (1982) Tannins and related compounds. Part 5. Isolation and characterization of polygalloylglucoses from Chinese gallotannin. J Chem Soc, Perkin Trans 10:2963–2968
Liangwu B, Zaisong W (1997) Chemicals from Gallnut. Chemical industry. Times 10:001
Deveoglu O, Torgan E, Karadag R (2012) Identification by RP-HPLC-DAD of natural dyestuffs from lake pigments prepared with a mixture of weld and dyer’s oak dye plants. J Liq Chromatogr Relat Technol 35:331–342
Salzer U, Prohaska R (2001) Stomatin, flotillin-1, and flotillin-2 are major integral proteins of erythrocyte lipid rafts. Blood 97:1141–1143
Cross NL (2004) Reorganization of lipid rafts during capacitation of human sperm. Biol Reprod 71:1367–1373
Yu W, Yuan B, Deng X, He L, Youyi Z, Qide H (2005) The preparation of HEK293 α1A or HEK293 α1B cell membrane stationary phase and the chromatographic affinity study of ligands of α1 adrenoceptor. Anal Biochem 339:198–205
Chen X, Cao Y, Lv D, Zhu Z, Zhang J, Chai Y (2012) Comprehensive two-dimensional HepG2/cell membrane chromatography/monolithic column/time-of-flight mass spectrometry system for screening anti-tumor components from herbal medicines. J Chromatogr A 1242:67–74
Mao Q, Yang J, Cui XM, Li JJ, Qi YT, Zhang PH, Wang Q (2012) Target separation of a new anti-tumor saponin and metabolic profiling of leaves of Panax notoginseng by liquid chromatography with eletrospray ionization quadrupole time-of-flight mass spectrometry. J Pharm Biomed Anal 59:67–77
He H, Han S, Zhang T, Zhang J, Wang S, Hou J (2012) Screening active compounds acting on the epidermal growth factor receptor from Radix scutellariae via cell membrane chromatography online coupled with HPLC/MS. J Pharm Biomed Anal 62:196–202
Xu X, Zhu L, Chen L (2004) Separation and screening of compounds of biological origin using molecularly imprinted polymers. J Chromatogr B 804:61–69
Kerbel RS (2008) Tumor angiogenesis. N Engl J Med 358:2039–2049
Krause DS, Van Etten RA (2005) Tyrosine kinases as targets for cancer therapy. N Engl J Med 353:172–187
Moaddel R, Oliveira RV, Kimura T, Hyppolite P, Juhaszova M, Xiao Y, Kellar KJ, Bernier M, Wainer IW (2007) Initial synthesis and characterization of an α7 nicotinic receptor cellular membrane affinity chromatography column: effect of receptor subtype and cell type. Anal Chem 80:48–54
Levis M, Ravandi F, Wang ES, Baer MR, Perl A, Coutre S, Erba H, Stuart RK, Baccarani M, Cripe LD, Tallman MS, Meloni G, Godley LA, Langston AA, Amadori S, Lewis ID, Nagler A, Stone R, Yee K, Advani A, Douer D, Wiktor-Jedrzejczak W, Juliusson G, Litzow MR, Petersdorf S, Sanz M, Kantarjian HM, Sato T, Tremmel L, Bensen-Kennedy DM, Small D, Smith BD (2011) Results from a randomized trial of salvage chemotherapy followed by lestaurtinib for patients with FLT3 mutant AML in first relapse. Blood 117:3294–3301
Lippa B, Morris J, Corbett M, Kwan TA, Noe MC, Snow SL, Gant TG, Mangiaracina M, Coffey HA, Foster B, Knauth EA, Wessel MD (2006) Discovery of novel isothiazole inhibitors of the TrkA kinase: structure-activity relationship, computer modeling, optimization, and identification of highly potent antagonists. Bioorg Med Chem Lett 16:3444–3448
Wood ER, Kuyper L, Petrov KG, Hunter Iii RN, Harris PA, Lackey K (2004) Discovery and in vitro evaluation of potent TrkA kinase inhibitors: oxindole and aza-oxindoles. Bioorg Med Chem Lett 14:953–957
Acknowledgments
This work was supported by National Natural Science Foundation of China (30973677), National Key Technology Support Program(2013BAD16B07-1), Doctoral Fund of Ministry of Education of China (20113227110012), Industry-university-research institution cooperation (2011-YY012, GY2010023, GY2011028) in Jiangsu Province and Zhenjiang City.
Author information
Authors and Affiliations
Corresponding authors
Additional information
S. Tong, M. Fu and X. Cao contributed equally to this work.
Rights and permissions
About this article
Cite this article
Tong, S., Fu, M., Cao, X. et al. Lipid Raft Stationary Phase Chromatography for Screening Anti-tumor Components from Galla chinensis . Chromatographia 77, 419–429 (2014). https://doi.org/10.1007/s10337-014-2623-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10337-014-2623-y