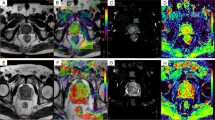
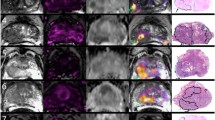
Abstract
Objective
To evaluate the utility of amide proton transfer (APT) imaging in estimating the Gleason score (GS) of prostate cancer (Pca).
Materials and methods
Sixty-six biopsy-proven cancers were categorized into four groups according to the GS: GS-6 (3 + 3); GS-7 (3 + 4/4 + 3); GS-8 (4 + 4) and GS-9 (4 + 5/5 + 4). APT signal intensities (APT SIs) and apparent diffusion coefficient (ADC) values of each GS group were compared by one-way analysis of variance with Tukey’s HSD post hoc test.
Results
The mean and standard deviation of the APT SIs (%) and ADC values (×10−3 mm2/s) were as follows: GS-6, 2.48 ± 0.59 and 1.16 ± 0.26; GS-7, 5.17 ± 0.66 and 0.92 ± 0.18; GS-8, 2.56 ± 0.85 and 0.86 ± 0.17; GS-9, 1.96 ± 0.75 and 0.85 ± 0.18, respectively. The APT SI of the GS-7 group was highest, and there were significant differences between the GS-6 and GS-7 groups and the GS-7 and GS-9 groups (p < 0.05). The ADC value of the GS-6 group was significantly higher than each value of the GS-7, GS-8, and GS-9 groups (p < 0.05), but no significant differences were obtained among the GS-7, GS-8, and GS-9 groups.
Conclusion
The mean APT SI in Pca with a GS of 7 was higher than that for the other GS groups.





Similar content being viewed by others
References
Damber JE, Aus G (2008) Prostate cancer. Lancet 371:1710–1721
Siegel R, Naishadham D, Jemal A (2012) Cancer statistics, 2012. CA Cancer J Clin 62:10–29
Mohler JL, Kantoff PW, Armstrong AJ et al (2014) Prostate cancer, version 2.2014. J Natl Compr Canc Netw 12:686–718
Epstein JI (2006) What’s new in prostate cancer disease assessment in 2006? Curr Opin Urol 16:146–151
Epstein JI, Allsbrook WC, Jr, Amin MB, Egevad LL, Committee IG (2005) The 2005 International Society of Urological Pathology (ISUP) Consensus Conference on Gleason Grading of Prostatic Carcinoma. Am J Surg Pathol 29:1228–1242
Gleason DF (1966) Classification of prostatic carcinomas. Cancer Chemother Rep 50:125–128
Gleason DF, Mellinger GT (1974) Prediction of prognosis for prostatic adenocarcinoma by combined histological grading and clinical staging. J Urol 111:58–64
Itou Y, Nakanishi K, Narumi Y, Nishizawa Y, Tsukuma H (2011) Clinical utility of apparent diffusion coefficient (ADC) values in patients with prostate cancer: can ADC values contribute to assess the aggressiveness of prostate cancer? J Magn Reson Imaging 33:167–172
Kobus T, Vos PC, Hambrock T et al (2012) Prostate cancer aggressiveness: in vivo assessment of MR spectroscopy and diffusion-weighted imaging at 3 T. Radiology 265:457–467
Vaché T, Bratan F, Mège-Lechevallier F, Roche S, Rabilloud M, Rouvière O (2014) Characterization of prostate lesions as benign or malignant at multiparametric MR imaging: comparison of three scoring systems in patients treated with radical prostatectomy. Radiology 272:446–455
Vargas HA, Akin O, Franiel T et al (2011) Diffusion-weighted endorectal MR imaging at 3 T for prostate cancer: tumor detection and assessment of aggressiveness. Radiology 259:775–784
Verma S, Rajesh A, Morales H et al (2011) Assessment of aggressiveness of prostate cancer: correlation of apparent diffusion coefficient with histologic grade after radical prostatectomy. AJR Am J Roentgenol 196:374–381
Litjens GJ, Barentsz JO, Karssemeijer N, Huisman HJ (2015) Clinical evaluation of a computer-aided diagnosis system for determining cancer aggressiveness in prostate MRI. Eur Radiol 25:318–3199
Zhang YD, Wang Q, Wu CJ et al (2015) The histogram analysis of diffusion-weighted intravoxel incoherent motion (IVIM) imaging for differentiating the Gleason grade of prostate cancer. Eur Radiol 25:994–1004
Zhou J, Blakeley JO, Hua J et al (2008) Practical data acquisition method for human brain tumor amide proton transfer (APT) imaging. Magn Reson Med 60:842–849
Zhou J, Payen JF, Wilson DA, Traystman RJ, van Zijl PC (2003) Using the amide proton signals of intracellular proteins and peptides to detect pH effects in MRI. Nat Med 9:1085–1090
Togao O, Yoshiura T, Keupp J et al (2014) Amide proton transfer imaging of adult diffuse gliomas: correlation with histopathological grades. Neuro Oncol 16:441–448
Dula AN, Arlinghaus LR, Dortch RD et al (2013) Amide proton transfer imaging of the breast at 3 T: establishing reproducibility and possible feasibility assessing chemotherapy response. Magn Reson Med 70:216–224
Jia G, Abaza R, Williams JD et al (2011) Amide proton transfer MR imaging of prostate cancer: a preliminary study. J Magn Reson Imaging 33:647–654
Tamada T, Sone T, Jo Y et al (2008) Prostate cancer: relationships between postbiopsy hemorrhage and tumor detectability at MR diagnosis. Radiology 248:531–539
Keupp J, Baltes C, Harver P, Van den Brink J (2011) Parallel RF Transmission based MRI Technique for Highly Sensitive Detection of Amide Proton Transfer in the Human Brain at 3. In: Proceedings of 19th Annual Meeting of International Society of Magnetic Resonance in Medicine, Québec, p 710
Agarwal HK, Keupp J, Bornardo M, Turkbey B, Choyke PL (2015) Saturation duration and power optimization for APT MRI of prostate cancer. In: Proceedings of 23rd Annual Meeting of International Society of Magnetic Resonance in Medicine, Toronto, p 3851
Nagarajan R, Margolis D, Raman S et al (2012) MR spectroscopic imaging and diffusion-weighted imaging of prostate cancer with Gleason scores. J Magn Reson Imaging 36:697–703
Oto A, Yang C, Kayhan A et al (2011) Diffusion-weighted and dynamic contrast-enhanced MRI of prostate cancer: correlation of quantitative MR parameters with Gleason score and tumor angiogenesis. AJR Am J Roentgenol 197:1382–1390
Tamada T, Sone T, Jo Y et al (2008) Apparent diffusion coefficient values in peripheral and transition zones of the prostate: comparison between normal and malignant prostatic tissues and correlation with histologic grade. J Magn Reson Imaging 28:720–726
Turkbey B, Shah VP, Pang Y et al (2011) Is apparent diffusion coefficient associated with clinical risk scores for prostate cancers that are visible on 3-T MR images? Radiology 258:488–495
Donati OF, Mazaheri Y, Afaq A et al (2014) Prostate cancer aggressiveness: assessment with whole-lesion histogram analysis of the apparent diffusion coefficient. Radiology 271:143–152
Mazaheri Y, Shukla-Dave A, Hricak H et al (2008) Prostate cancer: identification with combined diffusion-weighted MR imaging and 3D 1H MR spectroscopic imaging—correlation with pathologic findings. Radiology 246:480–488
Wang XZ, Wang B, Gao ZQ et al (2009) Diffusion-weighted imaging of prostate cancer: correlation between apparent diffusion coefficient values and tumor proliferation. J Magn Reson Imaging 29:1360–1366
Langer DL, van der Kwast TH, Evans AJ et al (2010) Prostate tissue composition and MR measurements: investigating the relationships between ADC, T2, K(trans), v(e), and corresponding histologic features. Radiology 255:485–494
Tan CH, Wang J, Kundra V (2011) Diffusion weighted imaging in prostate cancer. Eur Radiol 21:593–603
Zelhof B, Pickles M, Liney G et al (2009) Correlation of diffusion-weighted magnetic resonance data with cellularity in prostate cancer. BJU Int 103:883–888
Humphrey PA (2004) Gleason grading and prognostic factors in carcinoma of the prostate. Mod Pathol 17:292–306
Nguyen K, Sarkar A, Jain AK (2014) Prostate cancer grading: use of graph cut and spatial arrangement of nuclei. IEEE Trans Med Imaging 33:2254–2270
Freedland SJ, Kane CJ, Amling CL et al (2007) Upgrading and downgrading of prostate needle biopsy specimens: risk factors and clinical implications. Urology 69:495–499
Sakr WA, Tefilli MV, Grignon DJ et al (2000) Gleason score 7 prostate cancer: a heterogeneous entity? Correlation with pathologic parameters and disease-free survival. Urology 56:730–734
Acknowledgments
This work was supported by a Grant-in-Aid for Scientific Research (C), JSPS KAKENHI Grant Number 25461833.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Jochen Keupp declares that he receives a salary from Philips Research. The other authors have no conflict of interest.
Ethical standards
This prospective study was approved by our institutional review board and complied with ethics committee standards. All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Written informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Takayama, Y., Nishie, A., Sugimoto, M. et al. Amide proton transfer (APT) magnetic resonance imaging of prostate cancer: comparison with Gleason scores. Magn Reson Mater Phy 29, 671–679 (2016). https://doi.org/10.1007/s10334-016-0537-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10334-016-0537-4