Abstract
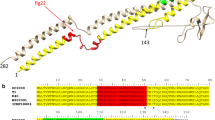
Flagellin in Pseudomonas syringae is a potent elicitor of defense responses including hypersensitive cell death in dicot plants. The oligopeptides flg22 consisting of 22 conserved amino acids near the N-terminus of flagellins is reported to induce plant defense responses. Because glycosylation of the central domain of flagellin affects its elicitor activity, we investigated whether any peptide sequence in addition to flg22 is required for flagellin-induced hypersensitive reaction. A study of recombinant flagellin polypeptides indicated that the N-terminal domain including the conserved flg22 is required for flagellin-induced hypersensitive cell death in Arabidopsis thaliana.



Similar content being viewed by others
Abbreviations
- HR:
-
Hypersensitive reaction
- PAMP:
-
Pathogen-associated molecular pattern
References
Adam L, Somerville SC (1996) Genetic characterization of five powdery mildew disease resistance loci in Arabidopsis thaliana. Plant J 9:341–356
Alonso JM, Stepanova AN, Leisse TJ, Kim CJ, Chen H, Shinn P, Stevenson DK, Zimmerman J, Barajas P, Cheuk R, Gadrinab C, Heller C, Jeske A, Koesema E, Meyers CC, Parker H, Prednis L, Ansari Y, Choy N, Deen H, Geralt M, Hazari N, Hom E, Karnes M, Mulholland C, Ndubaku R, Schmidt I, Guzman P, Aguilar-Henonin L, Schmid M, Weigel D, Carter DE, Marchand T, Risseuw E, Brogden D, Zeko A, Crosby WL, Berry CC, Ecker JR (2003) Genome-wide insertional mutagenesis of Arabidopsis thaliana. Science 301:653–657
Chisholm ST, Coaker G, Day B, Staskawicz BJ (2006) Host–microbe interactions: shaping the evolution of the plant immune response. Cell 124:803–814
Dangl JL, Jones JDG (2001) Plant pathogens and integrated defense responses to infection. Nature 411:826–833
Felix G, Duran JD, Volko S, Boller T (1999) Plants have a sensitive perception system for the most conserved domain of bacterial flagellin. Plant J 18:265–276
Gómez-Gómez L (2004) Plant perception systems for pathogen recognition and defence. Mol Immunol 41:1055–1062
Gómez-Gómez L, Boller T (2000) FLS2: an LRR receptor-like kinase involved in the perception of the bacterial elicitor flagellin in Arabidopsis. Mol Cell 5:1003–1011
Gómez-Gómez L, Boller T (2002) Flagellin perception: a paradigm for innate immunity. Trends Plant Sci 7:251–256
Ichinose Y, Taguchi F, Takeuchi K, Marutani M, Ishiga Y, Inagaki Y, Toyoda K, Shiraishi T (2004) Bacterial flagellins as elicitors of the defense response. In: Tsuyumu et al. (ed) Genomic and genetic analysis of plant parasitism and defense. APS Press, St Paul, pp 83–91
Ishiga Y, Takeuchi K, Taguchi F, Inagaki Y, Toyoda K, Shiraishi T, Ichinose Y (2005) Defense responses of Arabidopsis thaliana inoculated with Pseudomonas syringae pv. tabaci wild type and defective mutants for flagellin (ΔfliC) and flagellin-glycosylation (Δorf1). J Gen Plant Pathol 71:302–307
Jones DA, Takemoto D (2004) Plant innate immunity – direct and indirect recognition of general and specific pathogen-associated molecules. Curr Opin Immunol 16:48–62
Koch E, Slusarenko A (1990) Arabidopsis is susceptible to infection by a downy mildew fungus. Plant Cell 2:437–445
Mysore KS, Ryu C-M (2004) Nonhost resistance: how much do we know? Trend Plant Sci 9:97–104
Ramos HC, Rumbo M, Sirard J-C (2004) Bacterial flagellins: mediators of pathogenicity and host immune responses in mucosa. Trends Microbiol 12:509–517
Robatzek S, Chinchilla D, Boller T (2006) Ligand-induced endocytosis of the pattern recognition receptor FLS2 in Arabidopsis. Genes Dev 20:537–542
Sun W, Dunning FM, Pfund C, Weingarten R, Bent AF (2006) Within-species flagellin polymorphism in Xanthomonas campestris pv. campestris and its impact on elicitation of Arabidopsis FLAGELLIN SENSING2-dependent defenses. Plant Cell 18:764–779
Taguchi F, Shimizu R, Inagaki Y, Toyoda K, Shiraishi T, Ichinose Y (2003a) Post-translational modification of flagellin determines the specificity of HR induction. Plant Cell Physiol 44:342–349
Taguchi F, Shimizu R, Nakajima R, Toyoda K, Shiraishi T, Ichinose Y (2003b) Differential effects of flagellin from Pseudomonas syringae pv. tabaci, tomato, glycinea on plant defense response. Plant Physiol Biochem 41:165–174
Taguchi F, Takeuchi K, Katoh E, Murata K, Suzuki T, Marutani M, Kawasaki T, Eguchi M, Katoh S, Kaku H, Yasuda C, Inagaki Y, Toyoda K, Shiraishi T, Ichinose Y (2006) Identification of glycosylation genes and glycosylated amino acids of flagellin in Pseudomonas syringae pv. tabaci. Cell Microbiol 8:923–938
Zipfel C, Felix G (2005) Plants and animals: a different taste for microbes? Curr Opin Plant Biol 8:353–360
Acknowledgments
We thank the Leaf Tobacco Research Laboratory, Japan Tobacco Inc. and the Salk Institute for providing P. syringae pv. tabaci 6605 and T-DNA tagged Arabidopsis lines for fls2 mutants, respectively. This work was supported in part by Grants-in-Aid for Scientific Research (B) (No. 18380035) and (S) (No. 15108001) from the Ministry of Education, Culture, Sports, Science and Technology of Japan, and the Okayama University COE program “Establishment of Plant Health Science”.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Naito, K., Ishiga, Y., Toyoda, K. et al. N-terminal domain including conserved flg22 is required for flagellin-induced hypersensitive cell death in Arabidopsis thaliana . J Gen Plant Pathol 73, 281–285 (2007). https://doi.org/10.1007/s10327-007-0017-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10327-007-0017-9