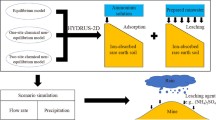
Abstract
Uranium tailings are radioactive and toxic solid wastes that could contaminate waters, soils and sediments. Actually the risk of contamination is poorly known due to the lack of knowledge on the mobility of contaminants, especially the influence of acid rain on leaching, transformation and speciation of contaminants. Here, we studied the effect of simulated rain at different pH on the transport and transformation of Mn, Sr, Pb and U from uranium tailings. Semi-dynamic leaching tests and sequential extraction were conducted for Mn, Sr, Pb and U to determine their leaching patterns and to determine their speciation in uranium tailings. Results show that Mn, Sr, Pb and U become more leachable with decreasing rain pH. The release of these metals is initially rapid and then reaches a near steady state. After leaching with simulated rain, concentrations of metal-exchangeable and metal-carbonate fractions in tailings are highly reduced. The process governing the leaching of Mn and U is mainly wash-off, whereas the leached Sr and Pb are dissolution-controlled.



Similar content being viewed by others
References
Abdelouas A (2006) Uranium mill tailings: geochemistry, mineralogy, and environmental impact. Elements 2:335–341. https://doi.org/10.2113/gselements.2.6.335
Al-Abed SR, Hageman PL, Jegadeesan G, Madhavan N, Allen D (2006) Comparative evaluation of short-term leach tests for heavy metal release from mineral processing waste. Sci Total Environ 364:14–23. https://doi.org/10.1016/j.scitotenv.2005.10.021
Al-Abed SR, Jegadeesan G, Purandare J, Allen D (2008) Leaching behavior of mineral processing waste: comparison of batch and column investigations. J Hazard Mater 153:1088–1092. https://doi.org/10.1016/j.jhazmat.2007.09.063
Alam MS, Cheng T (2014) Uranium release from sediment to groundwater: influence of water chemistry and insights into release mechanisms. J Contam Hydrol 164:72–87. https://doi.org/10.1016/j.jconhyd.2014.06.001
Appel C, Ma L (2002) Concentration, pH, and surface charge effects on cadmium and lead sorption in three tropical soils. J Environ Qual 31:581–589. https://doi.org/10.2134/jeq2002.5810
Brown GE, Hochella MF, Calas G (2017) Improving mitigation of the long-term legacy of mining activities: nano- and molecular-level concepts and methods. Elements 13:325–330. https://doi.org/10.2138/gselements.13.5.325
Coelho I, Castanheira I, Bordado JM, Donard O, Silva JAL (2017) Recent developments and trends in the application of strontium and its isotopes in biological related fields. TrAC Trend Anal Chem 90:45–61. https://doi.org/10.1016/j.trac.2017.02.005
Cumberland SA, Douglas G, Grice K, Moreau JW (2016) Uranium mobility in organic matter-rich sediments: a review of geological and geochemical processes. Earth Sci Rev 159:160–185. https://doi.org/10.1016/j.earscirev.2016.05.010
Dang DH, Wang W, Pelletier P, Poulain AJ, Evans RD (2018) Uranium dispersion from U tailings and mechanisms leading to U accumulation in sediments: insights from biogeochemical and isotopic approaches. Sci Total Environ 610–611:880–891. https://doi.org/10.1016/j.scitotenv.2017.08.156
Dove PM, Czank CA (1995) Crystal chemical controls on the dissolution kinetics of the isostructural sulfates: celestite, anglesite, and barite. Geochim Cosmochim Acta 59(10):1907–1915. https://doi.org/10.1016/0016-7037(95)00116-6
Dutta BK, Khanra S, Mallick D (2009) Leaching of elements from coal fly ash: assessment of its potential for use in filling abandoned coal mines. Fuel 88(7):1314–1323. https://doi.org/10.1016/j.fuel.2009.01.005
El-Kamash AM, El-Naggar MR, El-Dessouky MI (2006) Immobilization of cesium and strontium radionuclides in zeolite-cement blends. J Hazard Mater 136(2):310–316. https://doi.org/10.1016/j.jhazmat.2005.12.020
Feng M, Peng T, Sun H, Wang P (2016) Effect of oxidation degree on structure and cation exchange capacity of graphite oxide. Chin J Inorg Chem 32(3):427–433. https://doi.org/10.11862/CJIC.2016.047
Finneran KT, Anderson RT, Nevin KP, Lovley DR (2002) Potential for bioremediation of uranium-contaminated aquifers with microbial U(VI) reduction. Soil Sediment Contam 11(3):339–357. https://doi.org/10.1080/20025891106781
Frisbie SH, Mitchell EJ, Sarkar B (2013) World health organization increases its drinking-water guideline for uranium. Environ Sci Process Impacts 15(10):1817–1823. https://doi.org/10.1039/C3EM00381G
Fuentes A, Lloréns M, Sáez J, Isabel Aguilar M, Ortuño JF, Meseguer VF (2008) Comparative study of six different sludges by sequential speciation of heavy metals. Bioresour Technol 99(3):517–525. https://doi.org/10.1016/j.biortech.2007.01.025
Ghariani RA, Gržetić I, Antić M, Mandić SN (2010) Distribution and availability of potentially toxic metals in soil in central area of Belgrade, Serbia. Environ Chem Lett 8(3):261–269. https://doi.org/10.1007/s10311-009-0215-0
Gong B, Wu P, Huang Z, Li Y, Yang S, Dang Z, Ruan B, Kang C (2016) Efficient inhibition of heavy metal release from mine tailings against acid rain exposure by triethylenetetramine intercalated montmorillonite (TETA-Mt). J Hazard Mater 318:396–406. https://doi.org/10.1016/j.jhazmat.2016.07.011
Gustafsson JP, Persson I, Oromieh AG, van Schaik JW, Kleja DB (2014) Chromium(III) complexation to natural organic matter: mechanisms and modeling. Environ Sci Technol 48:1753–1761. https://doi.org/10.1021/es404557e
Hageman PL, Seal RR, Diehl SF, Piatak NM, Lowers HA (2015) Evaluation of selected static methods used to estimate element mobility, acid-generating and acid-neutralizing potentials associated with geologically diverse mining wastes. Appl Geochem 57:125–139. https://doi.org/10.1016/j.apgeochem.2014.12.007
Harter RD, Naidu R (2001) An assessment of environmental and solution parameter impact on trace-metal sorption by soils. Soil Sci Soc Am J 65(3):597–612. https://doi.org/10.2136/sssaj2001.653597x
Kaniki AT, Tumba K (2019) Management of mineral processing tailings and metallurgical slags of the congolese copperbelt: environmental stakes and perspectives. J Clean Prod 210:1406–1413. https://doi.org/10.1016/j.jclepro.2018.11.131
Kushwaha A, Hans N, Kumar S, Rani R (2018) A critical review on speciation, mobilization and toxicity of lead in soil-microbe-plant system and bioremediation strategies. Ecotoxicol Environ Saf 147:1035–1045. https://doi.org/10.1016/j.ecoenv.2017.09.049
Lasheen MR, Ammar NS (2009) Assessment of metals speciation in sewage sludge and stabilized sludge from different wastewater treatment plants, Greater Cairo, Egypt. J Hazard Mater 164(2–3):740–749. https://doi.org/10.1016/j.jhazmat.2008.08.068
Li J, Jia C, Lu Y, Tang S, Shim H (2015) Multivariate analysis of heavy metal leaching from urban soils following simulated acid rain. Microchem J 122:89–95. https://doi.org/10.1016/j.microc.2015.04.015
Lin C, Yu R, Hu G, Yang Q, Wang X (2016) Contamination and isotopic composition of Pb and Sr in offshore surface sediments from Jiulong River, Southeast China. Environ Pollut 218:644–650. https://doi.org/10.1016/j.envpol.2016.07.051
Liu B, Peng T, Sun H (2017a) Leaching behavior of U, Mn, Sr, and Pb from different particle-size fractions of uranium mill tailings. Environ Sci Pollut Res 24(18):15804–15815. https://doi.org/10.1007/s11356-017-8921-9
Liu B, Peng T, Sun H, Yue H (2017b) Release behavior of uranium in uranium mill tailings under environmental conditions. J Environ Radioact 171:160–168. https://doi.org/10.1016/j.jenvrad.2017.02.016
Mahar A, Wang P, Ali A, Awasthi MK, Lahori AH, Wang Q, Li R, Zhang Z (2016) Challenges and opportunities in the phytoremediation of heavy metals contaminated soils: a review. Ecotoxicol Environ Saf 126:111–121. https://doi.org/10.1016/j.ecoenv.2015.12.023
McArthur JM, Sikdar PK, Nath B, Grassineau N, Marshall JD, Banerjee DM (2012) Sedimentological control on Mn, and other trace elements, in groundwater of the Bengal Delta. Environ Sci Technol 46(2):669–676. https://doi.org/10.1021/es202673n
Meca S, Giménez J, Casas I, Martí V, de Pablo J (2012) Uranium speciation in river sediments contaminated by phosphate ores. Environ Chem Lett 10(1):49–53. https://doi.org/10.1007/s10311-011-0327-1
Mitchell GN (1991) Water quality issues in the British uplands. Appl Geogr 11(3):201–214. https://doi.org/10.1016/0143-6228(91)90030-D
Ospina-Alvarez N, Głaz Ł, Dmowski K, Krasnodębska-Ostręga B (2014) Mobility of toxic elements in carbonate sediments from a mining area in Poland. Environ Chem Lett 12(3):435–441. https://doi.org/10.1007/s10311-014-0468-0
Othmane G, Allard T, Morin G, Sélo M, Brest J, Llorens I, Chen N, Bargar JR, Fayek M, Calas G (2013) Uranium association with iron-bearing phases in mill tailings from Gunnar, Canada. Environ Sci Technol 47(22):12695–12702. https://doi.org/10.1021/es401437y
Pan Z, Giammar DE, Mehta V, Troyer LD, Catalano JG, Wang Z (2016) Phosphate-induced immobilization of uranium in Hanford sediments. Environ Sci Technol 50(24):13486–13494. https://doi.org/10.1021/acs.est.6b02928
Patra AC, Sumesh CG, Mohapatra S, Sahoo SK, Tripathi RM, Puranik VD (2011) Long-term leaching of uranium from different waste matrices. J Environ Manag 92(3):919–925. https://doi.org/10.1016/j.jenvman.2010.10.046
Pöykiö R, Mäkelä M, Nurmesniemi H, Dahl O, Oguchi M (2013) Application of the BRC sequential extraction scheme for assessing the leaching of elements in wood-based ash fractions from a large-sized (115 MW) industrial power plant of a pulp and board mill. Waste Biomass Valorization 4(4):821–830. https://doi.org/10.1007/s12649-012-9170-2
Ram R, Charalambous F, McMaster S, Tardio J, Bhargava SK (2013) An investigation on the effects of several anions on the dissolution of synthetic uraninite (UO2). Hydrometallurgy 136:93–104. https://doi.org/10.1016/j.hydromet.2013.03.003
Santos EA, Ladeira AC (2011) Recovery of uranium from mine waste by leaching with carbonate-based reagents. Environ Sci Technol 45(8):3591–3597. https://doi.org/10.1021/es2002056
Shaw SA, Hendry MJ, Essilfie-Dughan J, Kotzer T, Wallschläger D (2011) Distribution, characterization, and geochemical controls of elements of concern in uranium mine tailings, Key Lake, Saskatchewan, Canada. Appl Geochem 26(12):2044–2056. https://doi.org/10.1016/j.apgeochem.2011.07.002
Steinhausler F, Zaitseva L (2007) Uranium mining and milling: material security and risk assessment. Int J Nucl Gov Econ Ecol 1(3):286–304. https://doi.org/10.1504/IJNGEE.2007.014675
Szecsody JE, Truex MJ, Qafoku NP, Wellman DM, Resch T, Zhong L (2013) Influence of acidic and alkaline waste solution properties on uranium migration in subsurface sediments. J Contam Hydrol 151:155–175. https://doi.org/10.1016/j.jconhyd.2013.05.009
Tałałaj IA, Biedka P, Bartkowska I (2019) Treatment of landfill leachates with biological pretreatments and reverse osmosis. Environ Chem Lett 17(3):1177–1193. https://doi.org/10.1007/s10311-019-00860-6
Tessier A, Campbell PG, Bisson M (1979) Sequential extraction procedure for the speciation of particulate trace metals. Anal Chem 51(7):844–851. https://doi.org/10.1021/ac50043a017
Wang G, Um W, Wang Z, Reinoso-Maset E, Washton NM, Mueller KT, Perdrial N, O’Day PA, Chorover J (2017) Uranium release from acidic weathered hanford sediments: single-pass flow-through and column experiments. Environ Sci Technol 51(19):11011–11019. https://doi.org/10.1021/acs.est.7b03475
Xie Z, Du Y, Zeng Y, Li Y, Yan M, Jiao S (2009) Effects of precipitation variation on severe acid rain in Southern China. J Geogr Sci 19(4):489–501. https://doi.org/10.1007/s11442-009-0489-y
Yang Z, Tian S, Liu L, Wang X, Zhang Z (2018) Recycling ground MSWI bottom ash in cement composites: long-term environmental impacts. Waste Manag 78:841–848. https://doi.org/10.1016/j.wasman.2018.07.002
Yin M, Sun J, Chen Y, Wang J, Shang J, Belshaw N, Shen C, Liu J, Li H, Linghu W, Xiao T, Dong X, Song G, Xiao E, Chen D (2019) Mechanism of uranium release from uranium mill tailings under long-term exposure to simulated acid rain: geochemical evidence and environmental implication. Environ Pollut 244:174–181. https://doi.org/10.1016/j.envpol.2018.10.018
Yue YC, Li MH, Wang HB, Zhang BL, He W (2018) The toxicological mechanisms and detoxification of depleted uranium exposure. Environ Health Prev 23:18. https://doi.org/10.1186/s12199-018-0706-3
Acknowledgements
This research was financially supported by the National Key R&D Program of China (2018YFC1802902), the National Natural Science Foundation of China (41372052) and the doctoral foundation of Southwest University of Science and Technology (Grant No. 18zx7104).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Liu, B., Sun, H., Peng, T. et al. Transport and transformation of uranium and heavy metals from uranium tailings under simulated rain at different pH. Environ Chem Lett 18, 495–503 (2020). https://doi.org/10.1007/s10311-019-00951-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10311-019-00951-4