Abstract
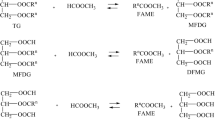
Biodiesel emerged again recently as an alternative for fossil fuels. Besides energy, biodiesel can be used as raw material to synthesize high value products such as epoxides. Indeed, epoxides are versatile intermediates in organic synthesis for numerous reactions due to the high reactivity of the oxirane ring. Actually common epoxidation by peracids in organic solvent can last several hours according to the double bonds number. The solvent confers selectivity to the reaction, preventing di-hydroxylation. Alternatively solvent-free reactions can be done in shorter times, but hydroxylation is less controlled. Here, we set up the synthesis of epoxides from methyl and ethyl esters of waste cooking oil, without solvent or metal catalyst. We tested molar ratio of hydrogen peroxide and formic acid, double bond number, reaction time, and temperature. Results show that the highest epoxide yields and selectivity, with high conversion of the double bonds, were obtained for temperature reactions between 50 and 65 °C, reaction times from 2 to 3 h, and molar ratio of 20/2/1. For those conditions, the double bonds conversion is equal or near 100 %, with epoxide yield and selectivity between 85 and 93 %. Organic solvent suppression, besides being environmentally friendly, also saves reaction time and subsequent purification steps.


Similar content being viewed by others
References
Alves CT, Oliveira A, Carneiro SAV, Silva AG, Andrade HMC, Vieira de Melo SAB, Torres EA (2013) Transesterification of waste frying oil using a zinc aluminate catalyst. Fuel Process Technol 106:102–107
Bitencourt TB, Nascimento MG (2010) The influence of organic solvent and ionic liquids on the selective formation of 2-(2-ethylhexyl)-3-phenyl-1,2-oxaziridine mediated by lipases. J Phys Org Chem 23:995–999
Campanella A, Fontanini C, Baltanás MA (2008) High yield epoxidation of fatty acid methyl esters with performic acid generated in situ. Chem Eng J 144:466–475
De Torres M, Arends IWCE, Mayoral JA, Pires E, Jiménez-Osés G (2012a) (a) A highly efficient, green and recoverable catalytic system for the epoxidation of fatty esters and biodiesel with H2O2. Appl Catal A: Gen 425–426:91–96
De Torres M, Jiménez-Osés G, Mayoral JA, Pires E, Blanco RM, Fernández O (2012b) (b) Evaluation of several catalytic systems for the epoxidation of methyl oleate using H2O2 as oxidant. Catal Today 195:76–82
Fazeelat T, Hussain ST, Ali SD, Iqbal Z, Ahmad W (2012) Homogeneous Catalysis of High Free Fatty Acid Waste Cooking Oil to Fatty Acid Methyl Esters (Biodiesel). Int J Energ Pow 1:31–36
Guzatto R, De Martini TL, Samios D (2011) The use of a modified TDSP for biodiesel production from soybean, linseed and waste cooking oil. Fuel Process Technol 92:2083–2088
Guzatto R, Deferrari D, Reiznautt QB, Cadore IR, Samios D (2012) Transesterification double step process modification for ethyl ester biodiesel production from vegetable and waste oils. Fuel 92:197–203
Hilker I, Bothe D, Prüss J, Warnecke H-J (2001) Chemo-enzymatic epoxidation of unsaturated plant oils. Chem Eng Sci 56:427–432
Kirk O, Christensen MW, Damhus T, Godtfredsen SE (1994) Enzyme catalyzed degradation and formation of peroxycarboxylic acids. Biocatal Biotransform 11:65–77
Lee K-W, Hailan C, Yinhua J, Kim Y-W, Chung K-W (2008) Modification of soybean oil for intermediates by epoxidation, alcoholysis and amidation. Korean J Chem Eng 25:474–482
Lu H, Sun S, Bi Y, Yang G (2012) Enzymatic epoxidation of biodiesel optimized by response surface methodology. Afr J Biotechnol 11:12356–12363
Mata TM, Sousa IRBG, Vieira SS, Caetano NS (2012) Biodiesel production from corn oil via enzymatic catalysis with ethanol. Energ Fuel 26:3034–3041
Nicolau A, Samios D, Piatnick CMS, Reiznautt QB, Martini DD, Chagas AL (2012) On the polymerisation of the epoxidised biodiesel: the importance of the epoxy rings position, the process and the products. Eur Polym J 48:1266–1278
Orellana-Coca C, Adlercreutz D, Andersson MM, Mattiasson B, Hatti-Kaul R (2005) Analysis of fatty acid epoxidation by high performance liquid chromatography coupled with evaporative light scattering detection and mass spectrometry. Chem Phys Lipids 135:189–199
Overeem A, Buisman GJH, Derksen JTP, Cuperus FP, Molhoek L, Grisnich W, Goemans C (1999) Seed oils rich in linolenic acid as renewable feedstock for environment-friendly crosslinkers in powder coatings. Ind Crop Prod 10:157–165
Petrovic ZS, Zlatanic A, Lava CC, Sinadinovic-Fišer S (2002) Epoxidation of soybean oil in toluene with peroxyacetic and peroxyformic acids: kinetics and side reactions. Eur J Lipid Sci Technol 104:293–299
Samios D, Pedrotti F, Nicolau A, Reiznautt QB, Martini DD, Dalcin FM (2009) A transesterification double step process: TDSP for biodiesel preparation from fatty acids triglycerides. Fuel Process Technol 90:599–605
Sander MM, Nicolau A, Guzatto R, Samios D (2012) Plasticiser effect of oleic acid polyester on polyethylene and polypropylene. Polym Test 31:1077–1082
Santacesaria E, Tesser R, Di Serio M, Turco R, Russo V, Verde D (2011) A biphasic model describing soybean oil epoxidation with H2O2 in a fed-batch reactor. Chem Eng J 173:198–209
Schneider RCS, Lara LRS, Bitencourt TB, Nascimento MG, Nunes MRS (2009) Chemo-enzymatic epoxidation of sunflower oil methyl esters. J Braz Chem Soc 20:1473–1477
Schulz GAS, da Silveira KC, Libardi DB, Peralba MCR, Samios D (2011) Synthesis and characterization of mono-acylglycerols through the glycerolysis of methyl esters obtained from linseed oil. Eur J Lipid Sci Technol 113:1533–1540
Silva WSD, Lapis AAM, Suarez PAZ, Neto BAD (2011) Enzyme-mediated epoxidation of methyl oleate supported by imidazolium-based ionic liquids. J Mol Cat B: Enzym 68:98–103
Supanchaiyamat N, Shuttleworth PS, Hunt AJ, Clark JH, Matharu AS (2012) Thermosetting resin based on epoxidised linseed oil and bio-derived crosslinker. Green Chem 14:1759–1765
Von Holleben MLA, Schuch CM (1997) Ativação do peróxido de hidrogênio para a epoxidação de olefinas não-funcionalizadas. Quim Nova 20:58–71
Yaakob Z, Mohammad M, Alherbawi M, Alam Z, Sopian K (2013) Overview of the production of biodiesel from waste cooking oil. Renew Sust Energ Rev 18:184–193
Yaliwal VS, Banapurmath NR, Tewari PG, Kundagol SI, Daboji SR, Galveen SC (2011) Production of renewable liquid fuels for diesel engine applications: a review. Cyber J: JRSE 1–9
Acknowledgments
The authors would like to thank FINEP, CNPq, and CAPES for their financial support.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lehnen, D.R., Guzatto, R., Defferrari, D. et al. Solvent-free biodiesel epoxidation. Environ Chem Lett 12, 335–340 (2014). https://doi.org/10.1007/s10311-013-0448-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10311-013-0448-9