Abstract
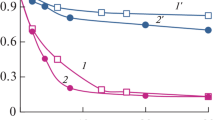
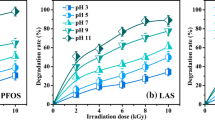
This paper describes a study of the treatment of surfactant synthetic solutions by chemical and photolytic oxidation. Synthetic solutions of linear alkylbenzene sulfonates (LAS) are treated in this work as this is a model compound commonly used in the formulation of detergents, with a great presence in urban and industrial waste-waters. The application of ultraviolet (UV) radiation combined with hydrogen peroxide to oxidize linear alkylbenzene sulfonates (LAS) is shown to be suitable as a primary oxidation step since conversions of about 50% of the original compounds are achieved in the most favorable cases. Initially, the influence of the operating variables on the degradation levels is analyzed in this work. A kinetic model that considers the contributions of both direct photolysis and radical attack is also worked out. Direct photolysis is performed to determine the quantum yield in the single photodecomposition reaction. In addition, the rate constant of the reaction between hydroxyl radicals and linear alkylbenzene sulfonates in the oxidizing system H2O2/UV is determined for different operational conditions. Finally, the contribution of each oxidation pathway is quantified, resulting in a higher contribution of the radical reaction than of photolysis in all cases.




Similar content being viewed by others
References
Baxendale JH, Wilson JA (1956) The photolysis of hydrogen peroxide at high light intensities. Trans Faraday Soc 53:344–356
Beltrán FJ, Ovejero G, Acedo B (1993) Oxidation of atrazine in water by UV radiation combined with hydrogen peroxide. Water Res 27:1013–1021
Benítez FJ, Beltran-Heredia J, Acero JL, Rubio FJ (2001) Oxidation of several chorophenoloic derivatives by UV irradiation and hydroxyl radicals. J Chem Tech Biotechnol 76:312–320
Cavalli L, Cassani G, Lazzarin M (1996) Biodegradation of linear alkylbenzene sulphonates (LAS) and alcohol ethoxylate (AE). Tenside Surfactants Deterg 33:158–165
Eisenberg GM (1943) Colorimetric determination of hydrogen peroxide. Ind Eng Chem 15:327–328
García-Morales JL, Nebot E, Romero LI, Sales D (2001) Comparison between acidogenic and methanogenic inhibition caused by linear alkylbenzene-sulfonate (LAS). Chem Biochem Eng 15:13–19
Glaze W, Kang JW, Chapin D (1987) The chemistry of water treatment processes involving ozone, hydrogen peroxide and ultraviolet radiation. Ozone Sci Eng 9:335–352
Hatchard CG, Parker CA (1956) A new sensitive chemical actinometer. II. Potassium ferrioxalate as a standard chemical actinometer. Proc R Soc Lond Ser A220:518–536
Kolthoff IM, Medalia AI (1949) The reaction between ferrous iron and peroxides. II. Reaction with hydrogen peroxide in the presence of oxygen. J Am Chem Soc 71:3784–3788
Nicole I, De Laat J, Doré M, Duguet JP, Bonnel C (1990) Utilisation du rayonnement ultraviolet dans les traitement des eaux: measure du flux photonique par actinometrie chimique au peroxyde d'hydrogene. (Use of UV radiation in water treatment: measurement of photonic flux by hydrogen peroxide actinometry.) Water Res 24:157–168
Perales JA, Manzano MA, Sales D, Quiroga JM (1999) Linear alkylbenzene sulphonates: biodegradability and isomeric composition. Bull Environ Contam Ecol 63:94–100
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sanz, J., Lombraña, J.I. & de Luís, A. Ultraviolet-H2O2 oxidation of surfactants. Environ Chem Lett 1, 32–37 (2003). https://doi.org/10.1007/s10311-002-0006-3
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10311-002-0006-3