Abstract
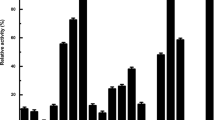
A novel lipolytic enzyme was isolated from a metagenomic library after demonstration of lipolytic activity on an LB agar plate containing 1% (w/v) tributyrin. A novel esterase gene (estIM1), encoding a lipolytic enzyme (EstIM1), was cloned using a shotgun method from a pFosEstIM1 clone of the metagenomic library, and the enzyme was characterized. The estIM1 gene had an open reading frame (ORF) of 936 base pairs and encoded a protein of 311 amino acids with a molecular mass 34 kDa and a pI value of 4.32. The deduced amino acid sequence was 62% identical to that of an esterase from an uncultured bacterium (ABQ11271). The amino acid sequence indicated that EstIM1 was a member of the family IV of lipolytic enzymes, all of which contain a GDSAG motif shared with similar enzymes of lactic acid microorganisms. EstIM1 was active over a temperature range of 1–50°C, at alkaline pH. The activation energy for hydrolysis of p-nitrophenyl propionate was 1.04 kcal/mol, within a temperature range of 1–40°C. The activity of EstIM1 was about 60% of maximal even at 1°C, suggesting that EstIM1 is efficiently cold-adapted. Further characterization of this cold-adapted enzyme indicated that the esterase may be very valuable in industrial applications.





Similar content being viewed by others
References
Arpigny JL, Jaeger KE (1999) Bacterial lipolytic enzymes: classification and properties. Biochem J 343:177–183
Aurilia V, Parracino A, D’Auria S (2008) Microbial carbohydrate esterases in cold adapted environments. Gene 410:234–240
Birnboim HC, Doly J (1979) A rapid alkaline extraction for screening recombinant plasmid DNA. Nuc Acid Res 7:1513–1523
Bornscheuer UT (2002) Microbial carboxyl esterases: classification, properties and application in biocatalysis. FEMS Microbiol Rev 26:73–81
Bradford MA (1976) Rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Cavicchioli R, Siddiqui KS, Andrews D, Sowers KR (2002) Low temperature extremophiles and their applications. Curr Opin Biotechnol 13:253–261
Choi YJ, Miguez CB, Lee BH (2004) Characterization and heterologous gene expression of a novel esterase from Lactobacillus casei CL96. Appl Environ Microbiol 70:3213–3221
Choo DW, Kurihara T, Suzuki T, Soda K, Esaki N (1998) A cold-adapted lipase of an Alaskan psychrotroph, Pseudomonas sp. strain B11–1: gene cloning and enzyme purification and characterization. Appl Environ Microbiol 64:486–491
Cowan D, Meyer Q, Stafford W, Muyanga S, Cameron R, Wittwer P (2005) Metagenomic gene discovery: past, present and future. Trends Biotechnol 23:321–329
Daniel R (2004) The soil metagenome—a rich resource for the discovery of novel natural products. Curr Opin Biotechnol 15:199–204
Elend C, Schmeisser C, Leggewie C, Babiak P, Carballeira JD, Steele HL, Reymond JL, Jaeger KE, Streit WR (2006) Isolation and biochemical characterization of two novel metagenome-derived esterases. Appl Environ Microbiol 72:3637–3645
Feller G, Gerday C (2003) Psychrophilic enzymes: hot topics in cold adaptation. Nat Rev Microbiol 1:200–208
Feller G, Narinix E, Arpigny JL, Aittaleb M, Baise E, Genicot S, Gerday C (1996) Enzymes from psychrophilic organisms. FEMS Microbiol Rev 18:189–202
Fleishmann RD, Adams MD, White O, Clayton RA, Kirkness EF, Kerlavage AR, Bult CJ, Tomb JF, Dougherty BA, Merrick JM (1995) Whole-genome random sequencing and assembly of Haemophilus influenzae Rd. Science 269:496–512
Foster FC (1988) Microenvironments of soil microorganisms. Biol Fertil Soils 6:189–203
Gerday C, Aittaleb M, Bentahir M, Chessa JP, Claverie P, Collins T, D‘Amico S, Feller G (2000) Cold-adapted enzymes: from fundamentals to biotechnology. Trends Biotechnol 18:103–107
Handelsman J (2004) Metagenomics: application of genomics to uncultured microorganisms. Microbiol Mol Biol Rev 68:669–685
Hårdeman F, Sjöling S (2007) Metagenomic approach for the isolation of a novel low-temperature-active lipase from uncultured bacteria of marine sediment. FEMS Microbiol Ecol 59:524–534
Heath C, Hu XP, Cary SC, Cowan D (2009) Identification of a novel alkaliphilic esterase active at low temperatures by screening a metagenomic library. Appl Environ Microbiol 75:4657–4659
Hong KS, Lim HK, Chung EJ, Park EJ, Lee MH, Kim JC, Choi GJ, Cho KY, Lee SW (2007) Selection and characterization of forest soil metagenome genes encoding lipolytic enzymes. J Microbiol Biotechnol 17:1655–1660
Jaeger KE, Dijkstra BW, Reetz MT (1999) Bacterial biocatalysts: molecular biology, three-dimensional structures, and biotechnological applications of lipases. Annu Rev Microbiol 53:315–351
Jaeger KE, Eggert T (2002) Lipases for biotechnology. Curr Opin Biotechnol 13:390–397
Jaeger KE, Reetz MT (1998) Microbial lipases form versatile tools for biotechnology. Trends Biotechnol 16:396–403
Jeon JH, Kim JT, Kang SG, Lee JH, Kim SJ (2009) Characterization and its potential application of two esterases derived from the arctic sediment metagenome. Mar Biotechnol 11:307–316
Jeon JH, Kim JT, Kim YJ, Kim YK, Lee HS, Kang SG, Kim SJ, Lee JH (2009) Cloning and characterization of a new cold-active lipase from a deep-sea sediment metagenome. Appl Microbiol Biotechnol 81:865–874
Joseph B, Ramteke PW, Thomas G, Shrivastava N (2007) Standard review cold-active microbial lipases: a versatile tool for industrial applications. Biotechnol Mol Biol Rev 2:39–48
Kellenberger E (2001) Exploring the unknown: the silent revolution of microbiology. EMBO Rep 2:5–7
Kim EY, Oh KH, Lee MH, Kang CH, Oh TK, Yoon JH (2009) Novel cold-adapted alkaline lipase from an intertidal flat metagenome and proposal for a new family of bacterial lipases. Appl Environ Microbiol 75:257–260
Kim YJ, Choi GS, Kim SB, Yoon GS, Kim YS, Ryu YW (2006) Screening and characterization of a novel esterase from a metagenomic library. Protein Express Purif 45:315–323
Khudary RAl, Venkatachalam R, Elleuche MS, Antranikian G (2010) A cold-adapted esterase of a novel marine isolate, Pseudoalteromonas arctica: gene cloning, enzyme purification and characterization. Extremophiles 14:273–285
Ko WH, Wang IT, Ann PJ (2005) A simple method for detection of lipolytic microorganisms in soils. Soil Biol Biochem 37:597–599
Lee SW, Won KH, Lim HK, Kim JC, Choi GJ, Cho KY (2004) Screening for novel lipolytic enzymes from uncultured soil microorganisms. Appl Microbiol Biotechnol 65:720–726
Lineweaver H, Burk D (1934) The determination of enzyme dissociation constants. J Am Chem Soc 56:658–666
Liu YY, Xu JH, Hu Y (2000) Enhancing effect of Tween-80 on lipase performance in enantioselective hydrolysis of ketoprofen ester. J Mol Catal B: Enzymatic 10:523–529
Osterlund T, Danielsson B, Degerman E, Contreras JA, Edgren G, Davis RC, Schotz MC, Holm C (1996) Domain-structure analysis of recombinant rat hormone-sensitive lipase. Biochem J 15:411–420
Panda T, Gowrishankar BS (2005) Production and applications of esterases. Appl Microbiol Biotechnol 67:160–169
Ranjan R, Grover A, Kapardar RK, Sharma R (2005) Isolation of novel lipolytic genes from uncultured bacteria of pond water. Biochem Biophys Res Commun 335:57–65
Rashamuse K, Ronneburg T, Hennessy F, Visser D, van Heerden E, Piater L, Litthauer D, Moller C, Brady D (2009) Discovery of a novel carboxylesterase through functional screening of a pre-enriched environmental library. J Appl Microbiol 106:1532–1539
Reetz MT, Jaeger KE (1998) Overexpression, immobilization and biotechnological application of Pseudomonas lipases. Chem Phys Lipids 93:3–14
Rhee JK, Ahn DG, Kim YG, Oh JW (2005) New thermophilic and thermostable esterase with sequence similarity to the hormone-sensitive lipase family, cloned from a metagenomic library. Appl Environ Microbiol 71:817–825
Rondon MR, August PR, Bettermann AD, Brady SF, Grossman TH, Liles MR, Loiacono KA, Lynch BA, MacNeil IA, Minor C, Tiong CL, Gilman M, Osburne MS, Clardy J, Handelsman J, Goodman RM (2000) Cloning the soil metagenome a strategy for accessing the genetic and functional diversity of uncultured microorganisms. Appl Environ Microbiol 66:2541–2547
Rossello-Mora R, Amann R (2001) The species concept for prokaryotes. FEMS Microbiol Rev 25:39–67
Soliman NA, Knoll M, Abdel-Fattah YR, Schmid RD, Lange S (2007) Molecular cloning and characterization of thermostable esterase and lipase from Geobacillus thermoleovorans YN isolated from desert soil in Egypt. Process Biochem 42:1090–1100
Steele HL, Streit WR (2005) Metagenomics: advances in ecology and biotechnology. FEMS Microbiol Lett 247:105–111
Streit WR, Schmitz RA (2004) Metagenomics—the key to the uncultured microbes. Curr Opin Microbiol 7:492–498
Sumby KM, Matthews AH, Grbin PR, Jiranek V (2009) Cloning and characterization of an intracellular esterase from the wine-associated lactic acid bacterium Oenococcus oeni. Appl Environ Microbiol 75:6729–6735
Verger R (1997) ‘Interfacial activation’ of lipases: facts and artifacts. Trends Biotech 15:32–38
Zhou J, Bruns MA, Tiedje JM (1996) DNA recovery from soils of diverse composition. Appl Environ Microbiol 62:316–322
Acknowledgments
This work was supported by an HTS-based Integrated Technology Development grant (no. 2008-04171) from the Ministry of Education, Science and Technology through the National Research Foundation of Korea, and a basic research grant from KRIBB.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Ko, KC., Rim, SO., Han, Y. et al. Identification and characterization of a novel cold-adapted esterase from a metagenomic library of mountain soil. J Ind Microbiol Biotechnol 39, 681–689 (2012). https://doi.org/10.1007/s10295-011-1080-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10295-011-1080-y