Abstract
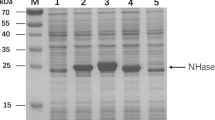
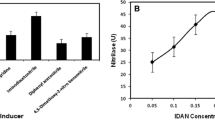
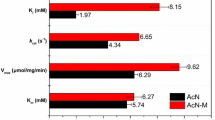
A novel enzymatic route for the synthesis of 2-amino-2,3-dimethylbutyramide (ADBA), important intermediate of highly potent and broad-spectrum imidazolinone herbicides, from 2-amino-2,3-dimethylbutyronitrile (ADBN) was developed. Strain Rhodococcus boritolerans CCTCC M 208108 harboring nitrile hydratase (NHase) towards ADBN was screened through a sophisticated colorimetric screening method and was found to be resistant to cyanide (5 mM). Resting cells of R. boritolerans CCTCC M 208108 also proved to be tolerant against high product concentration (40 g l−1) and alkaline pH (pH 9.3). A preparative scale process for continuous production of ADBA in both aqueous and biphasic systems was developed and some key parameters of the biocatalytic process were optimized. Inhibition of NHase by cyanide dissociated from ADBN was successfully overcome by temperature control (at 10°C). The product concentration, yield and catalyst productivity were further improved to 50 g l−1, 91% and 6.3 g product/g catalyst using a 30/70 (v/v) n-hexane/water biphasic system. Furthermore, cells of R. boritolerans CCTCC M 208108 could be reused for at lease twice by stopping the continuous reaction before cyanide concentration rose to 2 mM, with the catalyst productivity increasing to 12.3 g product/g catalyst. These results demonstrated that enzymatic synthesis of ADBA using whole cells of R. boritolerans CCTCC M 208108 showed potential for industrial application.






Similar content being viewed by others
References
Alister C, Kogan M (2005) Efficacy of imidazolinone herbicides applied to imidazolinone-resistant maize and their carryover effect on rotational crops. Crop Prot 24:375–379
Banerjee A, Sharma R, Banerjee UC (2002) The nitrile-degrading enzymes: current status and future prospects. Appl Microbiol Biotechnol 60:33–44
Boesten WHJ, Kamphuis J (1987) Process for the preparation of alpha-amino-alpha-methylcarboxylic acid amides and alpha-amino-alpha-cycloalkylcarboxylic acid amides. EP 0231546 (A1)
Brady D, Beeton A, Zeevaart J, Kgaje C, van Rantwijk F, Sheldon RA (2004) Characterisation of nitrilase and nitrile hydratase biocatalytic systems. Appl Microbiol Biotechnol 64:76–85
Brandão PFB, Verseck S, Syldatk C (2004) Bioconversion of D, L-tert-leucine nitrile to D-tert-leucine by recombinant cells expressing nitrile hydratase and D-selective amidase. Eng Life Sci 4:547–556
Cantarella M, Cantarella L, Gallifuoco A, Spera A (2006) Nitrile bioconversion by Microbacterium imperiale CBS 498–74 resting cells in batch and ultrafiltration membrane bioreactors. J Ind Microbiol Biotechnol 33:208–214
Deregnaucourt J, Archelas A, Barbirato F, Paris JM, Furstoss R (2007) Enzymatic transformations 63. High-concentration two liquid–liquid phase Aspergillus niger epoxide hydrolase-catalysed resolution: application to trifluoromethyl-substituted aromatic epoxides. Adv Synth Catal 349:1405–1417
Duchateau ALL, Crombach MG (1987) Determination of α-aminonitriles, α-amino acid amides and α-amino acids by means of HPLC, post-column reaction and fluorescence detection. Chromatographia 24:339–343
Effenberger F, Böhme J (1994) Enzyme-catalysed enantioselective hydrolysis of racemic naproxen nitrile. Bioorg Med Chem 2:715–721
Fallon RD, Stieglitz B, Turner I (1997) A Pseudomonas putida capable of stereoselective hydrolysis of nitriles. Appl Microbiol Biotechnol 47:156–161
Gastrock WH, Wepplo PJ (1987) Process for the resolution of certain racemic amino nitriles. US 4683324
Gerasimova T, Novikov A, Osswald S, Yanenko A (2004) Screening, characterization and application of cyanide-resistant nitrile hydratases. Eng Life Sci 4:543–546
Höllrigl V, Otto K, Schmid A (2007) Electroenzymatic asymmetric reduction of rac-3-methylcyclohexanone to (1S, 3S)-3-methylcyclohexanol in organic/aqueous media catalyzed by a thermophilic alcohol dehydrogenase. Adv Synth Catal 349:1337–1340
Hu JG, Wang YJ, Zheng YG, Shen YC (2007) Isolation of glycolonitrile-hydrolyzing microorganism based on colorimetric reaction. Enzyme Microb Technol 41:244–249
Kovacs JA (2004) Synthetic analogues of cysteinate-ligated non-heme iron and non-corrinoid cobalt enzymes. Chem Rev 104:825–848
Liang LY, Zheng YG, Shen YC (2008) Optimization of beta-alanine production from beta-aminopropionitrile by resting cells of Rhodococcus sp. G20 in a bubble column reactor using response surface methodology. Process Biochem 43:758–764
Lin ZJ, Zheng RC, Lei LH, Zheng YG, Shen YC (2011) Ferrous and ferric ions-based high-throughput screening strategy for nitrile hydratase and amidase. J Microbiol Meth 85:214–220
Martinková L, Mylerová V (2003) Synthetic applications of nitrile-converting enzymes. Curr Org Chem 7:1–7
Mersinger LJ, Hann EC, Cooling FB, Gavagan JE, Ben-Bassat A, Wu SJ, Petrillo KL, Payne MS, DiCosimo R (2005) Production of acrylamide using alginate-immobilized E. coli expressing Comamonas testosteroni 5-MGAM-4D nitrile hydratase. Adv Synth Catal 347:1125–1131
Nagasawa T, Nanba H, Ryuno K, Takeuchi K, Yamada H (1987) Nitrile hydratase of Pseudomonas chlororaphis B23. Purification and characterization. Eur J Biochem 162:691–698
Nagasawa T, Shimizu H, Yamada H (1993) The superiority of the third-generation catalyst, Rhodococcus rhodochrous J1 nitrile hydratase, for industrial production of acrylamide. Appl Microbiol Biotechnol 40:189–195
Nagasawa T, Takeuchi K, Yamada H (1991) Characterization of a new cobalt-containing nitrile hydratase purified from urea-induced cells of Rhodococcus rhodochrous J1. Eur J Biochem 196:581–589
Prasad S, Bhalla TC (2010) Nitrile hydratases (NHases): at the interface of academia and industry. Biotechnol Adv 28:725–741
Přepachalová I, Martinková L, Stolz A, Ovesná M, Bezouška K, Kopecký J, Křen V (2001) Purification and characterization of the enantioselective nitrile hydratase from Rhodococcus equi A4. Appl Microbiol Biotechnol 55:150–156
Raj J, Prasad S, Bhalla TC (2006) Rhodococcus rhodochrous PA-34: a potential biocatalyst for acrylamide synthesis. Process Biochem 41:1359–1363
Raj J, Sharma NN, Prasad S, Bhalla TC (2008) Acrylamide synthesis using agar entrapped cells of Rhodococcus rhodochrous PA-34 in a partitioned fed batch reactor. J Ind Microbiol Biotechnol 35:35–40
Ramezani MK, Oliver DP, Kookana RS, Lao WJ, Gill G, Preston C (2010) Faster degradation of herbicidally-active enantiomer of imidazolinones in soils. Chemosphere 79:1040–1045
Stepek WJ, Nigro MM (1984) Novel process for the preparation of aminonitriles useful for the preparation of herbicides. EP 0123830 (A2)
Takashima Y, Yamaga Y, Mitsuda S (1998) Nitrile hydratase from a thermophilic Bacillus smithii. J Ind Microbiol Biotechnol 20:220–226
van Pelt S, van Rantwijk F, Sheldon RA (2009) Synthesis of aliphatic (S)-alpha-hydroxycarboxylic amides using a one-pot bienzymatic cascade of immobilised oxynitrilase and nitrile hydratase. Adv Synth Catal 351:397–404
Wepplo P (1990) Imidazolinone herbicides-synthesis and novel chemistry. Pestic Sci 29:293–315
Yamada H, Shimizu S, Kobayashi M (2001) Hydratases involved in nitrile conversion: screening, characterization and application. Chem Rec 1:152–161
Zhang W, Webster EP, Pellerin KJ, Blouin DC (2006) Weed control programs in drill-seeded imidazolinone-resistant rice (Oryza sativa). Weed Technol 20:956–960
Zhou QY, Xu C, Zhang YS, Liu WP (2009) Enantioselectivity in the phytotoxicity of herbicide imazethapyr. J Agric Food Chem 57:1624–1631
Acknowledgments
This work was financially supported by the Major Basic Research Development Program of China (No. 2011CB710800), Natural Sciences Foundation of Zhejiang Province (No. Z4090612) and Doctor Program for High Education of China (No. 20103317120002).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Lin, ZJ., Zheng, RC., Wang, YJ. et al. Enzymatic production of 2-amino-2,3-dimethylbutyramide by cyanide-resistant nitrile hydratase. J Ind Microbiol Biotechnol 39, 133–141 (2012). https://doi.org/10.1007/s10295-011-1008-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10295-011-1008-6