Abstract
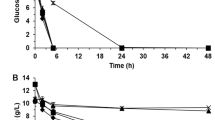
In general, it is believed that fermentation by yeast under harsh industrial conditions, especially if substrates such as wood hydrolysate or lignocellulosic substrates are used, requires the use of so-called industrial strains. In order to check whether this is always true, a comparison of performance was made using two industrial strains and four commonly used laboratory strains, the haploid and diploid versions of CEN-PK and X2180, under industrially relevant stress conditions. The industrial strains were a Swedish commercial baker’s yeast strain and a strain previously isolated from an industrial bioethanol production plant using lignocellulosic substrate. Stress conditions included, apart from growth in the lignocellulosic substrate itself, elevated concentrations of glucose, NaCl, ethanol, and lactate as well as low pH. Results showed that, indeed, the strain adapted to lignocellulosic substrate also possessed the highest growth rate as well as shortest duration of the lag phase in this type of medium. However, the higher the additional stress level, the lower the difference compared to other strains, and X2180 in particular displayed a high resistance to these additional stress conditions. Furthermore, no difference in performance could be detected between the haploid or diploid versions of the laboratory strains. It might be that, at least under some circumstances, a laboratory strain such as X2180 could be an industrially attractive production organism with the advantage of facilitating the possibilities for making controlled genetic manipulations.




Similar content being viewed by others
References
Albers E, Larsson C, Andlid T, Walsh MC, Gustafsson L (2007) Effect of nutrient starvation on the cellular composition and metabolic capacity of Saccharomyces cerevisiae. Appl Environ Microbiol 73:4839–4848. doi:10.1128/AEM.00425-07
Bencini DA, Wild JR, O’Donovan GA (1983) Linear one-step assay for the determination of orthophosphate. Anal Biochem 132:254–258. doi:10.1016/0003-2697(83)90004-0
Brandberg T, Franzen CJ, Gustafsson L (2004) The fermentation performance of nine strains of Saccharomyces cerevisiae in batch and fed-batch cultures in dilute-acid wood hydrolysate. J Biosci Bioeng 98:122–125
Gibson BR, Lawrence SJ, Leclaire JP, Powell CD, Smart KA (2007) Yeast responses to stresses associated with industrial brewery handling. FEMS Microbiol Rev 31:535–569. doi:10.1111/j.1574-6976.2007.00076.x
Hahn-Hagerdal B, Galbe M, Gorwa-Grauslund MF, Liden G, Zacchi G (2006) Bio-ethanol–the fuel of tomorrow from the residues of today. Trends Biotechnol 24:549–556. doi:10.1016/j.tibtech.2006.10.004
Hansen J, Kielland-Brandt MC (1996) Modification of biochemical pathways in industrial yeasts. J Biotechnol 49:1–12. doi:10.1016/0168-1656(96)01523-4
Herbert D, Phipps PJ, Strange RE (1971) Chemical analysis of microbial cells. In: Norris JR, Ribbons DW (eds) Methods in microbiology. Academic Press, London, pp 209–344
Mackenzie KF, Blomberg A, Brown AD (1986) Water stress plating hypersensitivity of yeasts. J Gen Microbiol 132:2053–2056
Martin C, Jönsson LJ (2003) Comparison of the resistance of industrial and laboratory strains of Saccharomyces and Zygosaccharomyces to lignocellulose-derived fermentation inhibitors. Enzyme Microb Technol 32:386–395
Olofsson K, Bertilsson G, Lidén G (2008) A short review on SSF—an interesting process option for ethanol production from lignocellulosic feedstocks. Biotechnol Biofuels 1:1–14
Purwadi R, Brandberg T, Taherzadeh M (2007) A possible industrial solution to ferment lignocellulosic hydrolyzate to ethanol: continuous cultivation with flocculating yeast. Int J Mol Sci 8:920–932. doi:10.3390/i8090920
Ragauskas AJ, Williams CK, Davison BH, Britovsek G, Cairney J, Eckert CA, Frederick WJ Jr, Hallett JP, Leak DJ, Liotta CL, Mielenz JR, Murphy R, Templer R, Tschaplinski T (2006) The path forward for biofuels and biomaterials. Science 311:484–489. doi:10.1126/science.1114736
Skinner KA, Leathers TD (2004) Bacterial contaminants of fuel ethanol production. J Ind Microbiol Biotechnol 31:401–408. doi:10.1007/s10295-004-0159-0
Taherzadeh MJ, Niklasson C, Lidén G (1999) Conversion of dilute acid hydrolyzates of spruce and birch to ethanol by fed-batch fermentation. Bioresour Technol 69:59–66. doi:10.1016/S0960-8524(98)00169-2
Walker GM (1998) Yeast physiology and biotechnology. John Wiley and Sons, Chichester
van Maris AJ, Abbott DA, Bellissimi E, van den Brink J, Kuyper M, Luttik MA, Wisselink HW, Scheffers WA, van Dijken JP, Pronk JT (2006) Alcoholic fermentation of carbon sources in biomass hydrolysates by Saccharomyces cerevisiae: current status. Antonie Leeuwenhoek 90:391–418. doi:10.1007/s10482-006-9085-7
Warringer J, Blomberg A (2003) Automated screening in environmental arrays allows analysis of quantitative phenotypic profiles in Saccharomyces cerevisiae. Yeast 20:53–67. doi:10.1002/yea.931
Wingren A, Galbe M, Zacchi G (2003) Techno-economic evaluation of producing ethanol from softwood: comparison of SSF and SHF and identification of bottlenecks. Biotechnol Prog 19:1109–1117. doi:10.1021/bp0340180
Acknowledgements
Financial support from the Swedish Energy Agency (Project No. 30188-1) and the Royal Swedish Academy of Sciences (Project No. FOA07B-086) is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Albers, E., Larsson, C. A comparison of stress tolerance in YPD and industrial lignocellulose-based medium among industrial and laboratory yeast strains. J Ind Microbiol Biotechnol 36, 1085–1091 (2009). https://doi.org/10.1007/s10295-009-0592-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10295-009-0592-1