Abstract
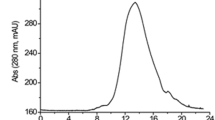
We have isolated a bacterium (TP-6) from the Indonesian fermented soybean, Tempeh, which produces a strong fibrinolytic protease and was identified as Bacillus subtilis. The protease (TPase) was purified to homogeneity by ammonium sulfate fractionation and octyl sepharose and SP sepharose chromatography. The N-terminal amino acid sequence of the 27.5 kDa enzyme was determined, and the encoding gene was cloned and sequenced. The result demonstrates that TPase is a serine protease of the subtilisin family consisting of 275 amino acid residues in its mature form. Its apparent K m and V max for the synthetic substrate N-succinyl-Ala-Ala-Pro-Phe-pNA were 259 μM and 145 μmol mg−1 min−1, respectively. The fibrinogen degradation pattern generated by TPase as a function of time was similar to that obtained with plasmin. In addition, N-terminal amino acid sequence analysis of the fibrinogen degradation products demonstrated that TPase cleaves Glu (or Asp) near hydrophobic acids as a P1 site in the α- and β-chains of fibrinogen to generate fragments D′, E′, and D′ similar to those generated by plasmin. On plasminogen-rich fibrin plates, TPase did not seem to activate fibrin clot lysis. Moreover, the enzyme converted the active plasminogen activator inhibitor-1 to the latent form.






Similar content being viewed by others
References
Astrupt T, Müllertz S (1952) The fibrin plate method for estimating fibrinolytic activity. Arch Biochem Biophys 40:346–351
Backes BJ, Harris JL, Leonetti F, Craik CS, Ellman JA (2000) Synthesis of positional-scanning libraries of fluorogenic peptide substrates to define the extended substrate specificity of plasmin and thrombin. Nat Biotech 18:187–193
Baker WF (2002) Thrombolytic therapy: clinical application. Thromb Hemost 8:291–314
Brown JH, Volkmann N, Jun G, Henschen-Edman AH, Cohen C (2000) The crystal structure of modified bovine fibrinogen. Proc Natl Acad Sci USA 97:85–90
Bryan PN (2000) Protein engineering of subtilisin. Biochim Biophys Acta 1543:203–222
Chang CT, Fan MH, Kuo FC, Sung HY (2000) Potent fibrinolytic enzyme from a mutant of Bacillus subtilis IMR-NK1. J Agric Food Chem 48:3210–3216
Cleary S, Mulkerrin MG, Kelley RF (1989) Purification and characterization of tissue plasminogen activator kringle-2 domain expressed in Escherichia coli. Biochemistry 28:1884–1891
Drapeau GR, Boily Y, Houmard J (1972) Purification and properties of an extracellular protease of Staphylococcus aureus. J Biol Chem 247:6720–6726
French JK, Hyde TA, Patel H, Amos DJ, McLaughlin SC, Webber BJ, White HD (1999) Survival 12 years after randomization to streptokinase: the influence of thrombolysis in myocardial infarction flow at three to four weeks. J Am Coll Cardiol 34:62–69
Fujita M, Nomura K, Hong K, Ito K, Asada A, Nishimuro S (1993) Purification and characterization of a strong fibrinolytic enzyme (nattokinase) in the vegetable cheese Natto, a popular soybean fermented food in Japan. Biochem Biophys Res Commun 197:1340–1347
Henschen A (1983) On the structure of functional sites in fibrinogen. Thromb Res Suppl 5:27–39
Karayadi D, Lukito W (2000) Functional food and contemporary nutrition-health paradigm: Tempeh and its potential beneficial effects in disease prevention and treatment. Nutrition 16:679
Kim WK, Choi KH, Kim YT, Park HH, Choi JY, Lee YS, Oh HI, Kwon IB, Lee SY (1996) Purification and characterization of a fibrinolytic enzyme produced from Bacillus sp. strain CK 11–4 screened from Chungkook-jang. Appl Environ Microbiol 62:2482–2488
Kunitz M (1947) Crystalline soybean trypsin inhibitor II. General properties. J Gen Physiol 30:291–310
Kunst F et al (1997) The complete genome sequence of the gram-positive bacterium Bacillus subtilis. Nature 390:249–256
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685
Lee HJ, Im H (2003) Purification of recombinant plasminogen activator-1 in the active conformation by refolding from inclusion bodies. Protein Expr Purif 31:99–107
Lijnen HR, Van Hoef B, Collen D (1993) Interaction of staphylokinase with different molecular forms of plasminogen. Eur J Biochem 211:91–97
Marder VJ, Landskroner K, Novokhatny V, Zimmerman TP, Kong M, Kanouse JJ, Jesmok G (2001) Plasmin induces local thrombolysis without causing hemorrhage: a comparison with tissue plasminogen activator in the rabbit. Thromb Haemost 86:739–745
McGrath KG, Zeffren B, Alexander J, Patterson R (1985) Allergic reactions to streptokinase consistent with anaphylactic or antigen–antibody complex-mediated damage. J Allergy Clin Immunol 76:453–457
Nakamura T, Yamagata Y, Ichishima E (1992) Nucleotide sequence of subtilisin NAT gene, aprT, of Bacillus subtilis (natto). Biosci Biotech Biochem 56:1869–1871
Pannell R, Gurewich V (1987) Activation of plasminogen by single-chain urokinase or by two-chain urokinase—a demonstration that single-chain urokinase has a low catalytic activity (pro-urokinase). Blood 69:22–26
Schleuning WD (2001) Vampire bat plasminogen activator DSPA-alpha-1 (desmoteplase): a thrombolytic drug optimized by natural selection. Haemostasis 31:118–122
Sumi H, Toki N, Sasaki K, Robbins KC (1980) Oral administration of urokinase. Thromb Res 20:711–714
Sumi H, Hamada H, Tsushima H, Mihara H, Muraki H (1987) A novel fibrinolytic enzyme (nattokinase) in the vegetable cheese Natto; a typical and popular soybean food in the Japanese diet. Experientia 43:1110–1111
Sumi H, Hamada H, Nakanishi K, Hiratani H (1990) Enhancement of the fibrinolytic activity in plasma by oral administration of natokinase. Acta Haematol 84:139–143
Thomson JD, Higgins DG, Gibson TJ (1994) Clustal W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position specific gap penalties and weight matrix choice. Nucleic Acids Res 22:4673–4680
Torr SR, Nachowiak DA, Fujii S, Sobel BE (1992) Plasminogen steal and clot lysis. J Am Coll Cardiol 19:1085–1090
Urano T, Ihara H, Umemura K, Suzuki Y, Oike M, Akita S, Tsukamoto Y, Suzuki I, Takada A (2001) The profibrinolytic enzyme subtilisin NAT purified from Bacillus subtilis cleaves and inactivates plasminogen activator inhibitor type 1. J Biol Chem 276:24690–24696
Vasantha N, Thomson LD, Rhodes C, Banner C, Nagle J, Filpula D (1984) Genes for alkaline protease and neutral protease from Bacillus amyloliquefaciens contain a large open reading frame between the regions coding for signal sequence and mature protein. J Bacteriol 159:811–819
Verstraete M (2000) Third-generation thrombolytic drugs. Am J Med 109:52–58
Walker JB, Nesheim ME (1999) The molecular weights, mass distribution, chain composition, and structure of soluble fibrin degradation products released from a fibrin clot perfused with plasmin. J Biol Chem 274:5201–5212
Weisberg WG, Barns SM, Pelletier DA, Lane DJ (1991) 16S ribosomal DNA amplification for phylogenetic study. J Bacteriol 173:697–703
Yoshimoto T, Oyama H, Honda T, Tone H, Takeshita T, Kamiyama T, Tsuru D (1988) Cloning and expression of subtilisin amylosacchariticus gene. J Biochem 103:1060–1065
Acknowledgements
This work [R11–1994-03405001-0] was supported by the Korea Science and Engineering Foundation (KOSEF). We gratefully acknowledge Prof. Jerry Eichler for editing the manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Seong-Bo Kim and Dong-Woo Lee contributed equally to the work.
Rights and permissions
About this article
Cite this article
Kim, SB., Lee, DW., Cheigh, CI. et al. Purification and characterization of a fibrinolytic subtilisin-like protease of Bacillus subtilis TP-6 from an Indonesian fermented soybean, Tempeh. J IND MICROBIOL BIOTECHNOL 33, 436–444 (2006). https://doi.org/10.1007/s10295-006-0085-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10295-006-0085-4