Abstract
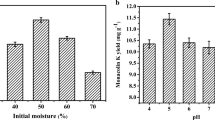
Monascus, a traditional Chinese fermentation fungus, is used as a natural dietary supplement. Its metabolic products monacolin K and γ-aminobutyric acid (GABA) have each been proven to be a cholesterol-lowering drug and a hypotensive agent. Citrinin, another secondary metabolite, is toxic to humans, thus lowering the acceptability of red mold rice to the general public. In this study, the influence of different carbon and nitrogen sources, and fatty acid or oils, on the production of monacolin K, citrinin and GABA by Monascus purpureus NTU 601 was studied. When 0.5% ethanol was added to the culture medium, the production of citrinin decreased from 813 ppb to 561 ppb while monacolin K increased from 136 mg/kg to 383 mg/kg and GABA increased from 1,060 mg/kg to 7,453 mg/kg. In addition, response surface methodology was used to optimize culture conditions for monacolin K, citrinin and GABA production, and data were collected according to a three-factor (temperature, ethanol concentration and amount of water supplemented), three-level central composite design. When 500 g rice was used as a solid substrate with 120 ml water and 0.3% ethanol, the production of monacolin K at 30°C increased from 136 mg/kg to 530 mg/kg, GABA production increased from 1,060 mg/kg to 5,004 mg/kg and citrinin decreased from 813 ppb to 460 ppb.





Similar content being viewed by others
References
Albert AW, Chen J, Springer J (1980) Mevinolin: a highly potent competitive inhibitor of hydroxymethylglutaryl-coenzyme A reductase and a cholesterol-lowering agent. Proc Natl Acad Sci USA 77:3957–3961
Blanc PJ, Loret MO, Santerre AT, Pareilleux A, Prome D, Prome JC, Laussac JP, Goma G (1994) Pigment of Monascus. J Food Sci 59:862–865
Blanc PJ, Laussac JP, Le Bars J, Le Bars P, Loret MO, Pareilleux A, Prome D, Prome JC, Santerre AL, Goma G (1995) Characterization of monascidin A from Monascus as citrinin. Int J Food Microbiol 27:201–213
Blanc PJ, Loret MO, Goma G (1995) Production of citrinin by various species of Monascus. Biotechnol Lett 17:291–294
Box GEP, Behnken DW (1960) Some new three level designs for the study of quantitative variable. Technometrics 2:455–463
Brown MS, Golstein JL (1981) How LDL receptors influence cholesterol and atherosclerosis. Sci Am 251:52–60
Budavari S, O’Neill MJ, Smith A, Heckelman PE (1989) The Merck Index vol 11. Merck, Rahway, N.J., pp 2330–2331
Comerio R, Virginia E, Fernandez P, Graciela V (1998) Influence of water activity on Penicillium citrinum growth and kinetics of citrinin accumulation in wheat. Int J Food Microbiol 42:219–223
Endo A (1979) Monacolin K, a new hypocholesterolemic agent produced by Monascus species. J Antibiot 32:852–854
Giovanni M (1983) Response surface methodology and product optimization. Food Technol 37:96–105
Hajjaj H, Blanc PJ, Groussac E, Goma G, Uribelarrea JL, Loubiere P (1999) Improvement of red pigment/ citrinin production ratio as a function of environment conditions by Monascus ruber. Biotechnol Bioeng 64:497–501
Hajjaj H, Blanc P, Groussac E, Uribelarrea JL, Goma G, Loubiere P (2000) Kinetic analysis of red pigment and citrinin production by Monascus ruber as a function of organic acid accumulation. Enzyme Microb Technol 27:619–625
Heber D, Yip I, Ashley JM, Elashoff DA, Elashoff RM, Go VLM (1999) Cholesterol-lowing effects of a proprietary Chinese red-yeast-rice dietary supplement. Am J Clin Nutr 69:231–236
Hsieh YT, Pan TM (2002) The analytic methods and assays for secondary metabolites of Monascus products. J Biomass Energy Soc China 21:63–71
Juzlova P, Martinkova L, Kren V (1996) Secondary metabolites of the fungus Monascus: a review. J Ind Microbiol Biotechnol 16:163–170
Kohama Y, Matsumoto S, Mimura T, Tanabe N, Inada A, Nakanishi T (1987) Isolation and identification of hypotensive principles in red mold rice. Chem Pharm Bull 35:2484–2489
Kycko S, Shiho SS, Yoko K, Tamio M (1998) Analytical method for citrinin in Monascus colour. Jpn J Food Chem 5:64–68
Lin YC, Ayres JC, Koehler PE (1981) Effect of temperature cycling on the production of patulin and citrinin. J Food Sci 46:974–977
Matheson GK, Freed E, Tunnicliff G (1986) Novel GABA analogues as hypotensive agent. Neuropharmacology 25:1191–1195
Monica S, Roel FM, Johanna FG (1999) Mutagenicity of commercial Monascus fermentation products and the role of citrinin contamination. Mutat Res 444:7–16
Narahara H 1994. Koji-mold and its product koji. J Brew Soc Jpn 89:873–881
Pimentel MCB, Melo EHM, Filho JLL, Duran N (1996) Production of lipase free of citrinin by Penicillium citrinum. Mycopathology 133:119–121
Rossetti V, Lombard A (1996) Determination of glutamate decarboxylase by high-performance liquid chromatography. J Chromatogr B 681:63–67
Su YC, Wang JJ, Lin TT, Pan TM (2003) Production of the secondary metabolites γ-aminobutyric acid and monacolin K by Monascus. J Ind Microbiol Biotechnol 30:40–46
Tsuji K, Ichikawa T, Tanabe N, Abe S, Tarui S, Nakagawa Y (1992) Antihypertensive activities of Beni-Koji extracts and γ-aminobutyric acid in spontaneously hypertensive rats. Jpn J Nutr 50:285–291
Ueno Y, Hayakawa K, Takahashi S, Oda K (1997) Purification and characterization of glutamate decarboxylase from Lactobacillus brevis IFO 12005. Biosci Biotechnol Biochem 61:1168–1171
Wang UL, Houng JY, Chang HS, Chien HCR, Hsu WH (1998) Selection of drug-resistant mutants Monascus pilosus for enhanced monacolin K production. J Chin Agric Chem Soc 36:192–200
Wong HC, Koehler PE (1981) Production and isolation of an antibiotic from Monascus purpureus and its relationship to pigment production. J Food Sci 46:589–592
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, JJ., Lee, CL. & Pan, TM. Improvement of monacolin K, γ-aminobutyric acid and citrinin production ratio as a function of environmental conditions of Monascus purpureus NTU 601. J IND MICROBIOL BIOTECHNOL 30, 669–676 (2003). https://doi.org/10.1007/s10295-003-0097-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10295-003-0097-2