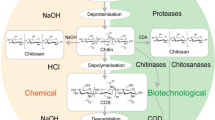
Abstract
Multiple industrial and medical uses of chitin and its derivatives have been developed in recent years. The demand for enzymes with new or desirable properties continues to grow as additional uses of chitin, chitooligosaccharides, and chitosan become apparent. Microorganisms, the primary degraders of chitin in the environment, are a rich source of valuable chitin-modifying enzymes. This review summarizes many methods that can be used to isolate and characterize chitin-modifying enzymes including chitin depolymerases, chitodextrinases, chitin deacetylases, N-acetylglucosaminidases, chitin-binding proteins, and chitosanases. Chitin analogs, zymography, detection of reducing sugars, genomic library screening, chitooligosaccharide electrophoresis, degenerate PCR primer design, thin layer chromatography, and chitin-binding assays are discussed.


Similar content being viewed by others
References
Austin P, Brine C, Castle J, Zikakis J (1981) Chitin: new facets of research. Science 212:749–753
Bassler B, Yu C, Lee Y, Roseman S (1991) Chitin utilization by marine bacteria. Degradation and catabolism of chitin oligosaccharides by Vibrio furnissii. J Biol Chem 266:24276–24286
Blumberg K, Liniere F, Pustilnik L, Bush C (1982) Fractionation of oligosaccharides containing N-acetyl amino sugars by reverse-phase high-pressure liquid chromatography. Anal Biochem 119:407–412
Bodek K (2002) Effect of microcrystalline chitosan on the solubility of ibuprofen. Acta Pol Pharm 59:105–108
Cardenas G, Orlando P, Edelio T (2001) Synthesis and applications of chitosan mercaptanes as heavy metal retention agent. Int J Biol Macromol 28:167–174
Chang K, Lee L, Fu W (2000) HPLC analysis of N-acetyl-chitooligosaccharides during the acid hydrolysis of chitin. J Food Drug Anal 8:75–83
Chong S, Mersha F, Comb D et al (1997) Single-column purification of free recombinant proteins using a self-cleavable affinity tag derived from a protein splicing element. Gene 192:271–281
Connell T, Metzger D, Lynch J, Folster J (1998) Endochitinase is transported to the extracellular milieu by the eps-encoded general secretory pathway of Vibrio cholerae. J Bacteriol 180:5591–5600
Cottrell M, Moore J, Kirchman D (1999) Chitinases from uncultured marine microorganisms. Appl Environ Microbiol 65:2553–2557
Cottrell M, Wood D, Yu Y, Kirchman D (2000) Selected chitinase genes in cultured and uncultured marine bacteria in the alpha- and gamma-subclasses of the proteobacteria. Appl Environ Microbiol 66:1195–1201
Cuero R (1999) Antimicrobial action of exogenous chitosan. EXS 87:315–333
De Boer W, Klein Gunnewiek P, Kowalchuk G, Van Veen J (2001) Growth of chitinolytic dune soil beta-subclass Proteobacteria in response to invading fungal hyphae. Appl Environ Microbiol 67:3358–3362
Drozd N, Sher A, Makarov V, Galbraikh L, Vikhoreva G, Gorbachiova I (2001) Comparison of antithrombin activity of the polysulphate chitosan derivatives in in vivo and in vitro system. Thromb Res 102:445–455
Edwards C (2000) Problems posed by natural environments for monitoring microorganisms. Mol Biotechnol 15:211–223
Fan J, Kondo A, Kato I, Lee Y (1994) High-performance liquid chromatography of glycopeptides and oligosaccharides on graphitized carbon columns. Anal Biochem 219:224–229
Felse P, Panda T (1999) Regulation and cloning of microbial chitinase genes. Appl Microbiol Biotechnol 51:141–151
Felt O, Buri P, Gurny R (1998) Chitosan: a unique polysaccharide for drug delivery. Drug Dev Ind Pharm 24:979–993
Fung K, Zhao K, He Z, Chye M (2002) Tobacco-expressed Brassica juncea chitinase BjCHI1 shows antifungal activity in vitro. Plant Mol Biol 50:283–294
Garcia E (1993) Assessment of endo-1,4-beta-d-glucanase activity by a rapid colorimetric assay using disodium-2–2′-bicinchoninate. J Food Biochem 17:135–145
Hedrick J, Smith A (1968) Size and charge isomer separation and estimation of molecular weights of proteins by disc gel electrophoresis. Arch Biochem Biophys 126:155–164
Helander I, Nurmiaho-Lassila E, Ahvenainen R, Rhoades J, Roller S (2001) Chitosan disrupts the barrier properties of the outer membrane of Gram-negative bacteria. Int J Food Microbiol 71:235–244
Henrissat B (1999) Classification of chitinases modules. EXS 87:137–156
Honda Y, Kirihata M, Fukamizo T, Kaneko S, Tokuyasu K, Brzezinski R (1999) Chitosanase-catalyzed hydrolysis of 4-methylumbelliferyl beta-chitotrioside. J Biochem (Tokyo) 126:470–474
Howard M, Ekborg N, Taylor L, Weiner R, Hutcheson S (2003) Genomic analysis and initial characterization of the chitinolytic system of Microbulbifer degradans strain 2-40. J Bacteriol 185:3352–3360
Huang Z, Prickett T, Potts M, Helm R (2000) The use of the 2-aminobenzoic acid tag for oligosaccharide gel electrophoresis. Carbohydr Res 328:77–83
Ilango R, Kavimani S, Jaykar B, Umamaheshwari G (1999) Dissolution studies on tablets of ibuprofen using chitosan. Indian J Exp Biol 37:505–508
Illum L (1998) Chitosan and its use as a pharmaceutical excipient. Pharm Res 15:1326–1331
Ingram C, Westpheling J (1995) The glucose kinase gene of Streptomyces coelicolor is not required for glucose repression of the chi63 promoter. J Bacteriol 177:3587–3588
Keyhani N, Roseman S (1996) Molecular cloning, isolation, and characterization of a periplasmic chitodextrinase. J Biol Chem 271:33414–33424
Kim Y, Liu J, Han X, Pervin A, Lindhardt R (1995) Analysis of fluorescently labeled sugars by reverse phase ion pairing HPLC. J Chrom Sci 33:162–167
Laemmli U (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685
Li H, Morimoto K, Katagiri N, Kimura T, Sakka K, Lun S, Ohmiya K (2002) A novel beta-N-acetylglucosaminidase of Clostridium paraputrificum M-21 with high activity on chitobiose. Appl Microbiol Biotechnol 60:420–427
Liu M, Cai Q, Liu H, Zhang B, Yan J, Yuan Z (2002) Chitinolytic activities in Bacillus thuringiensis and their synergistic effects on larvicidal activity. J Appl Microbiol 93:374–379
Lonhienne T, Mavromatis K, Vorgias C, Buchon L, Gerday C, Bouriotis V (2001) Cloning, sequences, and characterization of two chitinase genes from the Antarctic Arthrobacter sp. strain TAD20: isolation and partial characterization of the enzymes. J Bacteriol 183:1773–1779
Lopatin S, Ilyin M, Pustobaev V, Bezchetnikova Z, Varlamov V, Davankov V (1995) Mass-spectrometric analysis of N-acetylchitooligosaccharides prepared through enzymatic hydrolysis of chitosan. Anal Biochem 227:285–288
Matsuo Y, Kurita M, Park J, Tanaka K, Nakagawa T, Kawamukai M, Matsuda H (1999) Purification, characterization and gene analysis of N-acetylglucosaminidase from Enterobacter sp. G-1. Biosci Biotechnol Biochem 63:1261–1268
Miller G (1959) Use of dinitrosalicylic acid reagent for detection of reducing sugar. Anal Chem 31:426–428
Minhalma M, de Pinho M (2001) Flocculation/flotation/ultrafiltration integrated process for the treatment of cork processing wastewaters. Environ Sci Technol 35:4916–4921
Morimoto K, Karita S, Kimura T, Sakka K, Ohmiya K (1997) Cloning, sequencing, and expression of the gene encoding Clostridium paraputrificum chitinase ChiB and analysis of the functions of a novel cadherin-like domains and a chitin-binding domain. J Bacteriol 179:7306–7314
Muzzarelli R (1999) Native, industrial, and fossil chitins. In: Jolles P, Muzzarelli R (eds) Chitin and chitinases. Birkhauser, Basel
Muzzarelli R, Mattioli-Belmonte M, Pugnaloni A, Biagini G (1999) Biochemistry, histology and clinical uses of chitins and chitosans in wound healing. EXS 87:251–264
Nguyen J, Francou F, Virolle M, Guerineau M (1997) Amylase and chitinase genes in Streptomyces lividans are regulated by reg1, a pleiotropic regulatory gene. J Bacteriol 179:6383–6390
Nielsen K, Bojsen K, Roepstorff P, Mikkelsen J (1994) A hydroxyproline-containing class IV chitinase of sugar beet is glycosylated with xylose. Plant Mol Biol 25:241–257
Nishizawa H, Kita N, Okimura S, Takao E, Abe Y (1988) Determination of molecular weight of native proteins by polyacrylamide gradient gel electrophoresis. Electrophoresis 9:803–806
O’Brien M, Colwell R (1987) A rapid test for chitinase activity that uses 4-methylumbelliferyl-N-acetyl-beta-d-glucosaminide. Appl Environ Microbiol 53:1718–1720
Ohtakara A, Matsunaga H, Mitsutomi M (1990) Action pattern of Streptomyces griseus chitinase on partially N-acetylated chitosan. Agric Biol Chem 54:3191–3199
Pugsley A (1993) The complete general secretory pathway in Gram-negative bacteria. Microbiol Rev 57:50–108
Rajamohanan P, Ganapathy S, Vyas P, Ravikumar A, Deshpande M (1996) Solid-state CP/MASS 13C-NMR spectroscopy: a sensitive method to monitor enzymatic hydrolysis of chitin. J Biochem Biophys Methods 31:151–163
Ramaiah N, Hill R, Chun J, Ravel J, Matte M, Straube W, Colwell R (2000) Use of a chiA probe for detection of chitinase genes in bacteria from the Chesapeake Bay. FEMS Microbiol Ecol 34:63–71
Robbins P, Albright C, Benfield B (1988) Cloning and expression of a Streptomyces plicatus chitinase (chitinase-63) in Escherichia coli. J Biol Chem 263:443–447
Sakai K, Narihara M, Kasama Y, Wakayama M, Moriguchi M (1994) Purification and characterization of thermostable beta-N-acetylhexosaminidase of Bacillus stearothermophilus CH-4 isolated from chitin-containing compost. Appl Environ Microbiol 60:2911–2915
Shigemasa Y, Minami S (1996) Applications of chitin and chitosan for biomaterials. Biotechnol Genet Eng Rev 13:383–420
Singla A, Chawla M (2001) Chitosan: some pharmaceutical and biological aspects—an update. J Pharm Pharmacol 53:1047–1067
Somers P, Yao R, Doolin L, McGowan M, Fukuda D, Mynderse J (1987) Method for the detection and quantitation of chitinase inhibitors in fermentation broths; isolation and insect life cycle effect of A82516. J Antibiot (Tokyo) 40:1751–1756
Svitil A, Chadhain S, Moore J, Kirchman D (1997) Chitin degradation proteins produced by the marine bacterium Vibrio harveyii growing on different forms of chitin. Appl Environ Microbiol 63:408–413
Techkarnjanaruk S, Goodman A (1999) Multiple genes involved in chitin degradation from the marine bacterium Pseudoalteromonas sp. strain S91. Microbiology 145:925–934
Tokuyasu K, Mitsutomi M, Yamaguchi I, Hayashi K, Mori Y (2000) Recognition of chitooligosaccharides and their N-acetyl groups by putative subsites of chitin deacetylase from a deuteromycete, Colletotrichum lindemuthianum. Biochemistry 39:8837–8843
Trudel J, Asselin A (1989) Detection of chitinase activity after polyacrylamide gel electrophoresis. Anal Biochem 178:362–366
Trudel J, Asselin A (1990) Detection of chitin deacetylase activity after polyacrylamide gel electrophoresis. Anal Biochem 189:249–253
Tsigos I, Martinou A, Kafetzopoulos D, Bouriotis V (2000) Chitin deacetylases: new, versatile tools in biotechnology. Trends Biotechnol 18:305–312
Tsujibo H, Orikoshi H, Baba N, Miyahara M, Miyamoto K, Yasuda M, Inamori Y (2002) Identification and characterization of the gene cluster involved in chitin degradation in a marine bacterium, Alteromonas sp. strain O-7. Appl Environ Microbiol 68:263–270
Wang L, Keyhani N, Roseman S, Lee Y (1997) 4-Methylumbelliferyl glycosides of N-acetyl 4-thiochito-oligosaccharides as fluorogenic substrates for chitodextrinase from Vibrio furnissii. Glycobiology 7:855–860
Wang L, Keyhani N, Roseman S, Lee Y (1997) 4-Methylumbelliferyl glycosides of N-acetyl 4-thiochito-oligosaccharides as fluorogenic substrates for chitodextrinase from Vibrio furnissii. Glycobiology 7:855–860
Yamada H, Imoto T (1981) A convenient synthesis of glycolchitin, a substrate of lysozyme. Carbohydr Res 92:160–162
Ylitalo R, Lehtinen S, Wuolijoki E, Ylitalo P, Lehtimaki T (2002) Cholesterol-lowering properties and safety of chitosan. Arzneimittelforschung 52:1–7
Zhang H, Du Y, Yu X, Mitsutomi M, Aiba S (1999) Preparation of chitooligosaccharides from chitosan by a complex enzyme. Carbohydr Res 320:257–260
Zobell C, Rittenberg S (1937) The occurrence and characteristics of chitinoclastic bacteria in the sea. J Bacteriol 35:275–287
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Howard, M.B., Ekborg, N.A., Weiner, R.M. et al. Detection and characterization of chitinases and other chitin-modifying enzymes. J IND MICROBIOL BIOTECHNOL 30, 627–635 (2003). https://doi.org/10.1007/s10295-003-0096-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10295-003-0096-3