Abstract
Purpose
Congenital central hypoventilation syndrome (CCHS) and rapid-onset obesity with hypothalamic dysfunction, hypoventilation, and autonomic dysregulation (ROHHAD) are rare disorders of autonomic regulation with risk for disrupted neurocognitive development. Our aim is to summarize research on neurocognitive outcomes in these conditions, advance understanding of how to best support these individuals throughout development, and facilitate future research.
Methods
We conducted a narrative review of literature on neurocognitive outcomes in CCHS and ROHHAD, supplemented with previously unpublished data from patients with CCHS and ROHHAD at our Center for Autonomic Medicine in Pediatrics (CAMP).
Results
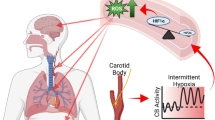
Individuals with CCHS and ROHHAD experience a wide range of neurocognitive functioning ranging from above average to below average, but are at particular risk for difficulties with working memory, processing speed, perceptual reasoning, and visuographic skills. An assessment framework emphasizing fluid cognition seems especially appropriate for these conditions. Owing to small cohorts and varied methods of data collection, it has been difficult to identify associations between disease factors (including CCHS PHOX2B genotypes) and cognitive outcomes. However, results suggest that early childhood is a period of particular vulnerability, perhaps due to the disruptive impact of recurrent intermittent hypoxic episodes on brain and cognitive development.
Conclusion
Neurocognitive monitoring is recommended as a component of routine clinical care in CCHS and ROHHAD as a marker of disease status and to ensure that educational support and disability accommodations are provided as early as possible. Collaborative efforts will be essential to obtain samples needed to enhance our understanding of neurocognitive outcomes in CCHS and ROHHAD.

Similar content being viewed by others
References
Saiyed R, Rand C, Carroll MS, Weese-Mayer DE (2014) Hypoventilation syndromes of infancy, childhood, and adulthood: congenital central hypoventilation syndrome (CCHS), later-onset CCHS, and rapid-onset obesity with hypothalamic dysfunction, hypoventilation, and autonomic dysregulation. Sleep Med Clin 9:P425-439
Weese-Mayer DE, Rand CM, Khaytin I, Slattery SM, Yap KL, Marazita ML, Berry-Kravis EM (2021) Congenital Central Hypoventilation Syndrome. In: Adam MP, Ardinger HH, Pagon RA, Wallace SE, Bean LJH, Gripp KW, Mirzaa GM, Amemiya A (eds). GeneReviews((R)). Seattle (WA)
Weese-Mayer DE, Berry-Kravis EM, Ceccherini I, Keens TG, Loghmanee DA, Trang H, Subcommittee ATSCCHS (2010) An official ATS clinical policy statement: congenital central hypoventilation syndrome: genetic basis, diagnosis, and management. Am J Respir Crit Care Med 181:626–644
Weese-Mayer DE, Berry-Kravis EM, Zhou L, Maher BS, Silvestri JM, Curran ME, Marazita ML (2003) Idiopathic congenital central hypoventilation syndrome: analysis of genes pertinent to early autonomic nervous system embryologic development and identification of mutations in PHOX2b. Am J Med Genet A 123A:267–278
Matera I, Bachetti T, Puppo F, Di Duca M, Morandi F, Casiraghi GM, Cilio MR, Hennekam R, Hofstra R, Schober JG, Ravazzolo R, Ottonello G, Ceccherini I (2004) PHOX2B mutations and polyalanine expansions correlate with the severity of the respiratory phenotype and associated symptoms in both congenital and late onset central hypoventilation syndrome. J Med Genet 41:373–380
Zhou A, Rand CM, Hockney SM, Niewijk G, Reineke P, Speare V, Berry-Kravis EM, Zhou L, Jennings LJ, Yu M, Ceccherini I, Bachetti T, Pennock M, Yap KL, Weese-Mayer DE (2021) Paired-like homeobox gene (PHOX2B) nonpolyalanine repeat expansion mutations (NPARMs): genotype–phenotype correlation in congenital central hypoventilation syndrome (CCHS). Genet Med 23:1656–1663
Khaytin I, Stewart TM, Zelko FA, Kee MAL, Osipoff JN, Slattery SM, Weese-Mayer DE (2022) Evolution of physiologic and autonomic phenotype in rapid-onset obesity with hypothalamic dysfunction, hypoventilation, and autonomic dysregulation over a decade from age at diagnosis. J Clin Sleep Med 18:937–944
Ize-Ludlow D, Gray JA, Sperling MA, Berry-Kravis EM, Milunsky JM, Farooqi IS, Rand CM, Weese-Mayer DE (2007) Rapid-onset obesity with hypothalamic dysfunction, hypoventilation, and autonomic dysregulation presenting in childhood. Pediatrics 120:e179–188
Carroll MS, Patwari PP, Kenny AS, Brogadir CD, Stewart TM, Weese-Mayer DE (2015) Rapid-onset obesity with hypothalamic dysfunction, hypoventilation, and autonomic dysregulation (ROHHAD): response to ventilatory challenges. Pediatr Pulmonol 50:1336–1345
Jacobson LA, Rane S, McReynolds LJ, Steppan DA, Chen AR, Paz-Priel I (2016) Improved behavior and neuropsychological function in children with ROHHAD after high-dose cyclophosphamide. Pediatrics 138(1): e20151080
Chow C, Fortier MV, Das L, Menon AP, Vasanwala R, Lam JC, Ng ZM, Ling SR, Chan DW, Choong CT, Liew WK, Thomas T (2015) Rapid-onset obesity with hypothalamic dysfunction, hypoventilation, and autonomic dysregulation (ROHHAD) syndrome may have a hypothalamus-periaqueductal gray localization. Pediatr Neurol 52:521–525
Vanderlaan M, Holbrook CR, Wang M, Tuell A, Gozal D (2004) Epidemiologic survey of 196 patients with congenital central hypoventilation syndrome. Pediatr Pulmonol 37:217–229
Montirosso R, Morandi F, D’Aloisio C, Berna A, Provenzi L, Borgatti R (2009) International classification of functioning, disability and health in children with congenital central hypoventilation syndrome. Disabil Rehabil 31(Suppl 1):S144–152
Oren J, Kelly DH, Shannon DC (1987) Long-term follow-up of children with congenital central hypoventilation syndrome. Pediatrics 80:375–380
Marcus CL, Jansen MT, Poulsen MK, Keens SE, Nield TA, Lipsker LE, Keens TG (1991) Medical and psychosocial outcome of children with congenital central hypoventilation syndrome. J Pediatr 119:888–895
Kaufman AS, Kaufman NL (1983) Kaufman Assessment Battery for Children (K-ABC). American Guidance Service, Circle Pines, MN
Dunn LM (1997) Peabody Picture Vocabulary Test—third edition (PPVT-III). American Guidance Service, Circle Pine, MN
Beery KE, Buktenica NA, Beery MA (2010) The Beery-Buktenica Developmental Test of Visual-Motor Integration: administaration, scoring, and teaching manual, 6th edn. Pearson, Minneapolis, MN
Silvestri JM, Weese-Mayer DE, Nelson MN (1992) Neuropsychologic abnormalities in children with congenital central hypoventilation syndrome. J Pediatr 120:388–393
Ruof H, Hammer J, Tillmann B, Ghelfi D, Weber P (2008) Neuropsychological, behavioral, and adaptive functioning of Swiss children with congenital central hypoventilation syndrome. J Child Neurol 23:1254–1259
Zelko FA, Nelson MN, Leurgans SE, Berry-Kravis EM, Weese-Mayer DE (2010) Congenital central hypoventilation syndrome: neurocognitive functioning in school age children. Pediatr Pulmonol 45:92–98
Wechsler D (2008) Wechsler Adult Intelligence Scale, 4th edn. Pearson, San Antonio, TX
Wechsler D (2014) Wechsler Intelligence Scale for Children, 5th edn. Pearson, San Antonio, TX
Bayley N (2006) Bayley scales of infant and toddler development® 3rd edition (Bayley-III®). The Psychological Corporation, San Antonio, TX
Charnay AJ, Antisdel-Lomaglio JE, Zelko FA, Rand CM, Le M, Gordon SC, Vitez SF, Tse JW, Brogadir CD, Nelson MN, Berry-Kravis EM, Weese-Mayer DE (2016) Congenital central hypoventilation syndrome: neurocognition already reduced in preschool-aged children. Chest 149:809–815
Lagercrantz R, Bergman K, Lagercrantz H, Markström A, Böhm B (2020) Neurocognitive function and quality of life with congenital central hypoventilation syndrome. J Sleep Med Disord 6:1097–1101
Macdonald KT, Mosquera RA, Yadav A, Caldas-Vasquez MC, Emanuel H, Rennie K (2020) Neurocognitive functioning in individuals with congenital central hypoventilation syndrome. BMC Pediatr 20:194
Ogata T, Muramatsu K, Miyana K, Ozawa H, Iwasaki M, Arakawa H (2020) Neurodevelopmental outcome and respiratory management of congenital central hypoventilation syndrome: a retrospective study. BMC Pediatr 20:342
Trang H, Bourgeois P, Cheliout-Heraut F (2020) Neurocognition in congenital central hypoventilation syndrome: influence of genotype and ventilation method. Orphanet J Rare Dis 15:322
Akshoomoff N, Newman E, Thompson WK, McCabe C, Bloss CS, Chang L, Amaral DG, Casey BJ, Ernst TM, Frazier JA, Gruen JR, Kaufmann WE, Kenet T, Kennedy DN, Libiger O, Mostofsky S, Murray SS, Sowell ER, Schork N, Dale AM, Jernigan TL (2014) The NIH toolbox cognition battery: results from a large normative developmental sample (PING). Neuropsychology 28:1–10
Welbel RZ, Rand CM, Zhou A, Fadl-Alla A, Chen ML, Weese-Mayer DE, Zelko FA (2022) Neurocognitive monitoring in congenital central hypoventilation syndrome with the NIH Toolbox(R). Pediatr Pulmonol 57:2040–2047
Zelko FA, Stewart TM, Brogadir CD, Rand CM, Weese-Mayer DE (2018) Congenital central hypoventilation syndrome: broader cognitive deficits revealed by parent controls. Pediatr Pulmonol 53:492–497
Shimokaze T, Sasaki A, Meguro T, Hasegawa H, Hiraku Y, Yoshikawa T, Kishikawa Y, Hayasaka K (2015) Genotype-phenotype relationship in Japanese patients with congenital central hypoventilation syndrome. J Hum Genet 60:473–477
Enjoji M, Yanai N (1961) Analytic test for development in infancy and childhood. Pediatr Int 4:2–6
Ikuzawa M, Matsushita Y, Nakase A (2002) The Kyoto Scale of Psychological Development test. Kyoto International Social Welfare Exchange Centre, Kyoto, Japan
Welbel RZ, Rand CM, Zhou A, Fadl-Alla A, Chen ML, Weese-Mayer DE, Zelko FA (2022) Neurocognitive monitoring in congenital central hypoventilation syndrome with the NIH Toolbox®. Pediatr Pulmonol 57:2040–2047.
Cave SN, Wright M, von Stumm S (2022) Change and stability in the association of parents’ education with children’s intelligence. Intelligence 90:1–8
Shipley W, Gruber C, Martin T, Klein A (2009) Shipley-2 Manual. Western Psychological Services, Los Angeles, CA
Maguire MJ, Schneider JM, Middleton AE, Ralph Y, Lopez M, Ackerman RA, Abel AD (2018) Vocabulary knowledge mediates the link between socioeconomic status and word learning in grade school. J Exp Child Psychol 166:679–695
Grudnikoff E, Foley C, Poole C, Theodosiadis E (2013) Nocturnal anxiety in a youth with rapid-onset obesity, hypothalamic dysfunction, hypoventilation, and autonomic dysregulation (ROHHAD). J Can Acad Child Adolesc Psychiatry 22:235–237
Kocaay P, Siklar Z, Camtosun E, Kendirli T, Berberoglu M (2014) ROHHAD syndrome: reasons for diagnostic difficulties in obesity. J Clin Res Pediatr Endocrinol 6:254–257
Erensoy H, Ceylan ME, Evrensel A (2016) Psychiatric symptoms in rapid-onset obesity with hypothalamic dysfunction, hypoventilation, and autonomic dysregulation syndrome and its treatment: a case report. Chin Med J (Engl) 129:242–243
Kot K, Moszczynska E, Lecka-Ambroziak A, Migdal M, Szalecki M (2016) ROHHAD in a 9 year-old boy—clinical case. Endokrynol Pol 67:226–231
Al-Harbi AS, Al-Shamrani A, Al-Shawwa BA (2016) Rapid-onset obesity, hypothalamic dysfunction, hypoventilation, and autonomic dysregulation in Saudi Arabia. Saudi Med J 37:1258–1260
Lee JM, Shin J, Kim S, Gee HY, Lee JS, Cha DH, Rim JH, Park SJ, Kim JH, Ucar A, Kronbichler A, Lee KH, Shin JI (2018) Rapid-onset obesity with hypoventilation, hypothalamic, autonomic dysregulation, and neuroendocrine tumors (ROHHADNET) syndrome: a systematic review. Biomed Res Int 2018:1250721
Warner JJ, Zelko FA, Weese-Mayer DE (2018) Rapid-onset obesity with hypothalamic dysfunction, hypoventilation, and autonomic dysregulation (ROHHAD): dichotomy of Neurocognitive Performance. In: American Thoracic Society. San Diego, CA
Harrison P, Oakland T (2003) Adaptive Behavior Assessment System, 2nd edn. Pearson, San Antonio, IX
Gershon RC, Fox RS, Manly JJ, Mungas DM, Nowinski CJ, Roney EM, Slotkin J (2020) The NIH Toolbox: overview of development for use with Hispanic populations. J Int Neuropsychol Soc 26:567–575
Duffey MM, Ayuku D, Ayodo G, Abuonji E, Nyalumbe M, Giella AK, Hook JN, Tran TM, McHenry MS (2022) Translation and cultural adaptation of NIH Toolbox cognitive tests into Swahili and Dholuo languages for use in children in western Kenya. J Int Neuropsychol Soc 28:414–423
Funding
This project was supported by the PHOX2B Patent Fund Chicago Community Trust Foundation and the National Institutes of Health National Center for Advancing Translational Sciences (NUCATS) Pilot and Voucher Grant. The authors have no financial relationships relevant to this article to disclose.
Author information
Authors and Affiliations
Contributions
Conceptualization: F.A.Z., R.Z.W., C.M.R., D.W.-M.; literature search: F.A.Z., R.Z.W., C.M.R., D.W.-M.; data collection/curation: F.A.Z., C.M.R., T.S., A.F.A., I.K., S.M.S., D.W.-M.; writing, original draft: F.A.Z., R.Z.W., C.M.R.; writing, review and editing: F.A.Z., R.Z.W., C.M.R., T.S., A.F.A., I.K., S.M.S., D.W.-M.; all authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that they have no conflicts of interest.
Ethical approval
Data presented from the Center for Autonomic Medicine in Pediatrics were collected under research approved by the Internal Review Board of the Ann & Robert H. Lurie Children’s Hospital of Chicago (protocols 2013–15230, 2016–137, 2017–1399). Consent for participation was obtained from subjects age 18 years and older, and from parents of individuals under 18 years of age. Assent for participation was also obtained from individuals between 12 and 17 years of age.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zelko, F.A., Welbel, R.Z., Rand, C.M. et al. Neurocognition as a biomarker in the rare autonomic disorders of CCHS and ROHHAD. Clin Auton Res 33, 217–230 (2023). https://doi.org/10.1007/s10286-022-00901-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10286-022-00901-1