Abstract
Objective
Electrochemical skin conductance (ESC) is a non-invasive test of sweat function developed as a potential marker of small fiber neuropathy. Here we systematically review the evolution of this device and the data obtained from studies of ESC across different diseases.
Methods
Electronic databases, including MEDLINE, and Google Scholar were searched through to February 2018. The search strategy included the following terms: “electrochemical skin conductance,” “EZSCAN,” and “Sudoscan.” The data values provided by each paper were extracted, where available, and input into tabular and figure data for direct comparison.
Results
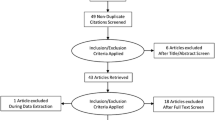
Thirty-seven studies were included this systematic review. ESC did not change by age or gender, and there was significant variability in ESC values between diseases, some of which exceeded control values. Longitudinal studies of disease demonstrated changes in ESC that were not biologically plausible. Of the 37 studies assessed, 25 received support from the device manufacturer. The extracted data did not agree with other published normative values. Prior studies do not support claims that ESC is a measure of small fiber sensory function or autonomic function.
Conclusions
Although many papers report significant differences in ESC values between disease and control subjects, the compiled data assessed in this review raises questions about the technique. Many of the published results violate biologic plausibility. A single funding source with a vested interest in the study outcomes has supported most of the studies. Normative values are inconsistent across publications, and large combined data sets do not support a high sensitivity and specificity. Finally, there is insufficient evidence supporting the claim that Sudoscan tests sudomotor or sensory nerve fiber function.



Similar content being viewed by others
References
Scarr D Lovblom LE, Lovshin JA et al (2017) Lower corneal nerve fibre length identifies diabetic neuropathy in older adults with diabetes: results from the Canadian Study of Longevity in Type 1 Diabetes. Diabetologia 60(12):2529–2531
Kass-Iliyya L, Javed S, Gosal D et al (2015) Small fiber neuropathy in Parkinson’s disease: a clinical, pathological and corneal confocal microscopy study. Parkinsonism Relat Disord 21(12):1454–1460
Scarr D, Lovblom LE, Ostrovski I et al (2017) Agreement between automated and manual quantification of corneal nerve fiber length: implications for diabetic neuropathy research. J Diabetes Complications 31(6):1066–1073
Selvarajah D, Cash T, Davies J et al (2015) SUDOSCAN: a simple, rapid, and objective method with potential for screening for diabetic peripheral neuropathy. PLoS One 10(10):e0138224
Luk AO, Fu WC, Li X et al (2015) The clinical utility of SUDOSCAN in chronic kidney disease in Chinese patients with type 2 diabetes. PLoS One 10(8):e0134981
Mao F, Liu S, Qiao X et al (2016) Sudoscan is an effective screening method for asymptomatic diabetic neuropathy in Chinese type 2 diabetes mellitus patients. J Diabetes Investig. 2016
Novak P (2017) Electrochemical skin conductance: a systematic review. Clin Auton Res. doi: https://doi.org/10.1007/s10286-017-0467-x.
Khalfallah K, Ayoub H, Calvet JH, Neveu X, Brunswick P, Griveau S, Lair V, Casir M, Bedioui F (2010) Noninvasive galvanic skin sensor for early diagnosis of sudomotor dysfunction: application to diabetes. IEEE Sens J 12(3):8
Vinik AI, Smith AG, Singleton JR et al (2016) Normative values for electrochemical skin conductances and impact of ethnicity on quantitative assessment of sudomotor function. Diabetes Technol Ther 18(6):391–398
Ang L, Jaiswal M, Callaghan B et al (2017) Sudomotor dysfunction as a measure of small fiber neuropathy in type 1 diabetes. Auton Neurosci 205:87–92
Zhu L, Zhao X, Zeng P et al (2016) Study on autonomic dysfunction and metabolic syndrome in Chinese patients. J Diabetes Invest 7(6):901–907
Chahal S, Vohra K, Syngle A (2016) Association of sudomotor function with peripheral artery disease in type 2 diabetes. Neurol Sci 38:151–156
Syngle V, Syngle A, Garg N, Krishan P, Verma I (2016) Predictors of autonomic neuropathy in rheumatoid arthritis. Auton Neurosci 201:54–59
Castro J, Miranda B, Castro I, de Carvalho M, Conceicao I (2016) The diagnostic accuracy of Sudoscan in transthyretin familial amyloid polyneuropathy. Clin Neurophysiol 127(5):2222–2227
Sahuc P, Chiche L, Dussol B, Pouget J, Franques J (2016) Sudoscan as a noninvasive tool to assess sudomotor dysfunction in patients with Fabry disease: results from a case–control study. Ther Clin Risk Manag 12:135–138
Casellini CM, Parson HK, Hodges K et al (2016) Bariatric surgery restores cardiac and sudomotor autonomic C-fiber dysfunction towards normal in obese subjects with type 2 diabetes. PLoS One 11(5):e0154211
Freedman BI, Smith SC, Bagwell BM et al (2015) Electrochemical skin conductance in diabetic kidney disease. Am J Nephrol 41(6):438–447
Névoret ML, Vinik AI (2015) CIDP variants in diabetes: measuring treatment response with a small nerve fiber test. J Diabetes Complications 29(2):313–317
Yajnik CS, Kantikar VV, Pande AJ, Deslypere JP et al (2012) Quick and simple evaluation of sudomotor function for screening of diabetic neuropathy. ISRN Endocrinol 2012:103714
Casellini CM, Parson HK, Richardson MS, Nevoret ML, Vinik AI (2013) Sudoscan, a noninvasive tool for detecting diabetic small fiber neuropathy and autonomic dysfunction. Diabetes Technol Ther 15(11):948–953
Eranki VG, Santosh R, Rajitha K et al (2013) Sudomotor function assessment as a screening tool for microvascular complications in type 2 diabetes. Diabetes Res Clin Pract 101(3):e11–e13
Syngle A, Verma I, Garg N, Krishan P et al (2013) Autonomic dysfunction in psoriatic arthritis. Clin Rheumatol 32(7):1059–1064
Yajnik CS, Kantikar V, Pande A et al (2013) Screening of cardiovascular autonomic neuropathy in patients with diabetes using non-invasive quick and simple assessment of sudomotor function. Diabetes Metab 39(2):126–131
Saad M, Psimaras D, Tafani C et al (2016) Quick, non-invasive and quantitative assessment of small fiber neuropathy in patients receiving chemotherapy. J Neurooncol 127(2):373–380
Calvet JH, Dupin J, Winiecki H, Schwarz PE (2013) Assessment of small fiber neuropathy through a quick, simple and non invasive method in a German diabetes outpatient clinic. Exp Clin Endocrinol Diabetes 121(2):80–83
Sheshah E, Madanat A, Al-Greesheh F et al (2015) Electrochemical skin conductance to detect sudomotor dysfunction, peripheral neuropathy and the risk of foot ulceration among Saudi patients with diabetes mellitus. J Diabetes Metab Disord 15:29
Hubert D, Brunswick P, Calvet JH, Dusser D, Fajac I et al (2011) Abnormal electrochemical skin conductance in cystic fibrosis. J Cyst Fibros 10(1):15–20
Mayaudon H, Miloche PO, Bauduceau B (2010) A new simple method for assessing sudomotor function: relevance in type 2 diabetes. Diabetes Metab 36(6 Pt 1):450–454
Gin H, Baudoin R, Raffaitin CH, Rigalleau V, Gonzalez C (2011) Non-invasive and quantitative assessment of sudomotor function for peripheral diabetic neuropathy evaluation. Diabetes Metab 37(6):527–532
Syngle A, Verma I, Krishan P, Garg N, Syngle V (2015) Disease-modifying anti-rheumatic drugs improve autonomic neuropathy in arthritis: DIANA study. Clin Rheumatol 34(7):1233–1241
Raisanen A, Eklund J, Calvet JH, Tuomilehto J (2014) Sudomotor function as a tool for cardiorespiratory fitness level evaluation: comparison with maximal exercise capacity. Int J Environ Res Public Health 11(6):5839–5848
Freedman BI, Bowden DW, Smith SC, Xu J, Divers J (2014) Relationships between electrochemical skin conductance and kidney disease in type 2 diabetes. J Diabetes Complications 28(1):56–60
Smith AG, Lessard M, Reyna S, Doudova M, Singleton JR (2014) The diagnostic utility of Sudoscan for distal symmetric peripheral neuropathy. J Diabetes Complications 28(4):511–516
Zeng Q, Dong SY, Wang ML et al (2016) Cardiac autonomic neuropathy risk estimated by sudomotor function and arterial stiffness in Chinese subjects. J Hum Hypertens 30(11):720–725
Lefaucheur JP, Wahab A, Plante-Bordeneuve V et al (2015) Diagnosis of small fiber neuropathy: a comparative study of five neurophysiological tests. Neurophysiol Clin 45(6):445–455
Callaghan BC, Xia R, Reynolds E et al (2018) Better diagnostic accuracy of neuropathy in obesity: a new challenge for neurologists. Clin Neurophysiol 129(3):654–662
Novak P (2016) Electrochemical skin conductance correlates with skin nerve fiber density. Front Aging Neurosci 8:199
Mao F, Liu S, Qiao X et al (2017) SUDOSCAN, an effective tool for screening chronic kidney disease in patients with type 2 diabetes. Exp Ther Med 14(2):1343–1350
Mao F, Zhu X, Lu B, Li Y (2018) Detection of relationships between SUDOSCAN with estimated glomerular filtration rate (eGFR) in Chinese patients with type 2 diabetes. Diabetes Res Clin Pract 138:113–118
Chae CS, Park GY, Choi YM et al (2017) Rapid, objective and non-invasive diagnosis of sudomotor dysfunction in patients with lower extremity dysesthesia: a cross-sectional study. Ann Rehabil Med 41(6):1028–1038
Ozaki R, Cheung KK, Wu E et al (2011) A new tool to detect kidney disease in Chinese type 2 diabetes patients: comparison of EZSCAN with standard screening methods. Diabetes Technol Ther 13(9):937–943
Jin J, Wang W, Gu T et al (2017) The application of SUDOSCAN for Screening Diabetic Peripheral Neuropathy in Chinese Population Screening DPN by SUDOSCAN. Exp Clin Endocrinol Diabetes. https://doi.org/10.1055/s-0043-116673
Wang D, Shen B, Wu C, Xue Y, Liu Y (2017) The relationship between cardiovascular autonomic dysfunction and ocular abnormality in Chinese T2DM. J Diabetes Res 2017:7125760
Hupin D, Pichot V, Celle S et al (2015) Sudomotor function and obesity-related risk factors in an elderly healthy population: the PROOF–Synapse Study. Int J Cardiol 186:247–249
Ramachandran A, Moses A, Shetty S et al (2010) A new non-invasive technology to screen for dysglycaemia including diabetes. Diabetes Res Clin Pract 88(3):302–306
ESHRE Capri Workshop (ECW) Group (2018) Protect us from poor-quality medical research. Hum Reprod 33(5):770–776
Abdel-Rahman TA, Collins KJ, Cowen T, Rustin M (1992) Immunohistochemical, morphological and functional changes in the peripheral sudomotor neuro-effector system in elderly people. J Auton Nerv Syst 37(3):187–197
Hanewinckel R, Drenthen J, van Oijen M et al (2016) Prevalence of polyneuropathy in the general middle-aged and elderly population. Neurology 87(18):1892–1898
Hanewinckel R, van Oijen M, Ikram MA, van Doorn PA (2016) The epidemiology and risk factors of chronic polyneuropathy. Eur J Epidemiol 31(1):5–20
Lauria G, Bakkers M, Schmitz C et al (2010) Intraepidermal nerve fiber density at the distal leg: a worldwide normative reference study. J Peripher Nerv Syst 15(3):202–207
Low PA, Denq JC, Opfer-Gehrking TL et al (1997) Effect of age and gender on sudomotor and cardiovagal function and blood pressure response to tilt in normal subjects. Muscle Nerve 20(12):1561–1568
Albers JW, Brown MB, Sima AA, Greene DA (1996) Nerve conduction measures in mild diabetic neuropathy in the Early Diabetes Intervention Trial: the effects of age, sex, type of diabetes, disease duration, and anthropometric factors. Neurology 46(1):85–91
Miras AD, Chuah LL, Khalil N et al (2015) Type 2 diabetes mellitus and microvascular complications 1 year after Roux-en-Y gastric bypass: a case–control study. Diabetologia 58(7):1443–1447
Muller-Stich BP, Fischer L, Kenngott HG et al (2013) Gastric bypass leads to improvement of diabetic neuropathy independent of glucose normalization—results of a prospective cohort study (DiaSurg 1 study). Ann Surg 258(5):760–765 (discussion 765-6)
Muller-Stich BP, Billeter AT, Fleming T et al (2015) Nitrosative stress but not glycemic parameters correlate with improved neuropathy in nonseverely obese diabetic patients after Roux-Y gastric bypass. Surg Obes Relat Dis 11(4):847–854
Pop-Busui R, Boulton AJ, Feldman EL et al (2017) Diabetic neuropathy: a position statement by the American Diabetes Association. Diabetes Care 40(1):136–154
Martin CL, Albers JW, Pop-Busui R (2014) Neuropathy and related findings in the diabetes control and complications trial/epidemiology of diabetes interventions and complications study. Diabetes Care 37(1):31–38
Penza P, Lombardi R, Camozzi F, Ciano C, Lauria G (2009) Painful neuropathy in subclinical hypothyroidism: clinical and neuropathological recovery after hormone replacement therapy. Neurol Sci 30(2):149–151
Singleton JR, Marcus RL, Lessard MK, Jackson JE, Smith AG (2015) Supervised exercise improves cutaneous reinnervation capacity in metabolic syndrome patients. Ann Neurol 77(1):146–153
Grimes DA, Schulz KF (2002) Uses and abuses of screening tests. Lancet 359(9309):881–884
Papanas N, Ziegler D (2014) New vistas in the diagnosis of diabetic polyneuropathy. Endocrine 47(3):690–698
Low VA, Sandroni P, Fealey RD, Low PA (2006) Detection of small-fiber neuropathy by sudomotor testing. Muscle Nerve 34(1):57–61
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Rights and permissions
About this article
Cite this article
Rajan, S., Campagnolo, M., Callaghan, B. et al. Sudomotor function testing by electrochemical skin conductance: does it really measure sudomotor function?. Clin Auton Res 29, 31–39 (2019). https://doi.org/10.1007/s10286-018-0540-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10286-018-0540-0