Abstract
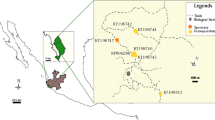
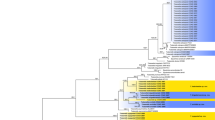
Nervilia nipponica is a tuberous terrestrial orchid that has a highly restricted distribution within common secondary evergreen forest communities in central and western Japan. Such a limited occurrence could be attributable to a requirement for a specific mycorrhizal fungus. As part of a broader examination of this hypothesis, we sought to elucidate the mycorrhizal associations of N. nipponica. Seventy-five samples of mycorrhizae from forty individuals were collected at ten populations throughout the orchid’s range in Japan. The identity of mycorrhizal fungi was investigated by sequencing two genetic markers (nrDNA ITS and nrDNA 28S LSU) and their relationships were assessed via phylogenetic analyses. The most frequently encountered mycorrhizal fungi consisted of four closely related Agaricomycetes that infected an average of 78.7 % of individuals per population. All four formed a discrete, monophyletic clade with low sequence homology to other fungi registered in GenBank, indicating that they belong to a novel, unnamed family. Two additional fungal groups, belonging to Ceratobasidiaceae and “Group B” Sebacinales, were found in 22.0 and 21.5 % of individuals per population, respectively. The orchid probably uses these two groups opportunistically, because they were found at lower densities and always in combination with the unidentified Agaricomycete. These findings suggest that a group of novel Agaricomycete fungi constitutes the dominant mycobiont of N. nipponica.




Similar content being viewed by others
References
Altschul SF, Madden TL, Schaffer AA, Zhang J, Zhang Z, Miller W, Lipman DJ (1997) Gapped BLAST and PSI-BLAST: a new generation of protein database search programs. Nucleic Acids Res 25:3389–3402
Arditti J, Ghani AKA (2000) Numerical and physical properties of orchid seeds and their biological implications. New Phytol 145:367–421
Batty AL, Dixon KW, Sivasithamparam K (2000) Soil seed-bank dynamics of terrestrial orchids. Lindleyana 15:227–236
Binder M, Hibbett DS, Larsoon KH, Larsoon E, Langer E, Langer G (2005) The phylogenetic distribution of resupinate forms across the major clades of mushroom-forming fungi (Homobasidiomycetes). Sys Biodiv 3:113–157
Bonnardeaux Y, Brundrett M, Batty A, Dixon K, Koch J, Sivasithamparam K (2007) Diversity of mycorrhizal fungi in terrestrial orchids: compatibility webs, brief encounters, lasting relationships and alien invasions. Mycol Res 111:51–61
Castresana J (2000) Selection of conserved blocks from multiple alignments for their use in phylogenetic analysis. Mol Biol Evol 17:540–552
Cribb PJ, Kell SP, Dixon KW, Barrett RL (2003) Orchid conservation: a global perspective. In: Dixon KW, Kell SP, Barrett RL, Cribb PJ (eds) Orchid conservation. Natural History Publications, Kota Kinabalu
Eum SM, Gale S, Yukawa T, Lee NS (2011) Phylogenetic and conservation status of the endangered terrestrial orchid Nervilia nipponica (Orchidaceae) in Korea. Biochem Sys Ecol 39:635–642
Felsenstein J (1985) Confidence limits on phylogenies: an approach using the bootstrap. Evol 39:783–791
Felsenstein J (2009) Phylip (Phylogeny Inference Package) version 3.69. Department of Genome Sciences and Department of Biology, University of Washington, Seattle
Gale S (2007) Autogamous seed set in a critically endangered orchid in Japan: pollination studies for the conservation of Nervilia nipponica. Plant Syst Evol 268:59–73
Gale S, Maeda A, Kuroiwa N (2006) Observations on the phenology and reproductive success of the critically endangered Nervilia nipponica (Orchidaceae) in Kochi Prefecture, Japan. Acta Phytotaxon Geobot 57:81–93
Gale S, Yukawa T, Kuroiwa N (2007) Studies in Asian Nervilia (Orchidaceae) I: neotypification and circumscription of N. nipponica in Japan. Kew Bull 62:85–94
Gale S, Maeda A, Chen CI, Yukawa T (2010a) Inter-specific relationships and hierarchical spatial genetic structuring in Nervilia nipponica, an endangered orchid in Japan. J Plant Res 123:625–637
Gale S, Yamazaki J, Hutchings MJ, Yukawa T, Miyoshi K (2010b) Constraints on establishment in an endangered terrestrial orchid: a comparative study of in vitro and in situ seed germinability and seedling development in Nervilia nipponica. Bot J Linn Soc 163:166–180
Gardes M, Bruns TD (1993) ITS primers with enhanced specificity for basidiomycetes—application to the identification of mycorrhizae and rusts. Mol Ecol 2:113–118
Gonzalez D, Carling DE, Kuninaga S, Vilgalys R, Cubeta MA (2001) Ribosomal DNA systematics of Ceratobasidium and Thanatephorus with Rhizoctonia anamorphs. Mycologia 93:1138–1150
Govaerts R, Bernet P, Kratochvil K, Gerlach G, Carr G, Alrich P, Pridgeon AM, Pfahl J, Campacci MA, Holland Baptista D, Tigges H, Shaw J, Cribb P, George A, Kreuz K, Wood J (2011) World checklist of Ochidaceae. Facilitated by the Royal Botanic Gardens, Kew. http://apps.kew.org/wcsp/. Accessed 25 Dec 2011
IUCN (2001) IUCN red list categories and criteria, Version 3.1. IUCN, Gland & Cambridge
Jacquemyn H, Brys R, Hermy M, Willems JH (2004) Does nectar reward affect rarity and extinction probabilities of orchid species? An assessment using historical records from Belgium and the Netherlands. Biol Conserv 121:257–263
Jacquemyn H, Honnay O, Cammue BP, Brys R, Lievens B (2010) Low specificity and nested subset structure characterize mycorrhizal associations in five closely related species of the genus Orchis. Mol Ecol 19:4086–4095
Katoh K, Toh H (2010) Parallelization of the MAFFT multiple sequence alignment program. Bioinformatics 26:1899–1900
Kimura M (1980) A simple method for estimating evolutionary rates of base substitutions through comparative studies of nucleotide sequences. J Mol Evol 16:111–120
Kull T, Hutchings MJ (2006) A comparative analysis of decline in the distribution ranges of orchid species in Estonia and the United Kingdom. Biol Conserv 129:31–39
Larsson KH, Larsson E, Kõljalg U (2004) High phylogenetic diversity among corticioid homobasidiomycetes. Mycol Res 108:983–1002
Lutzoni F, Kauff F, Cox JC, McLaughlin D, Celio G, Dentinger B, Padamsee M, Hibbett D, James TY, Baloch E, Grube M, Reeb V, Hofstetter V, Shcoch C, Arnold AE, Miadlikowska J, Spatafora J, Johnson D, Hambleton S, Crockett M, Shoemaker R, Sung GH, Lucking R, Lumbsch T, O’Donnell K, Binder M, Diederich P, Ertz D, Gueidan C, Hansen K, Harris RC, Hosaka K, Lim YW, Matheny B, Nishida H, Pfister D, Rogers J, Rossman A, Schmitt I, Sipman H, Stone J, Sugiyama J, Yahr R, Vilgalys R (2004) Assembling the fungal tree of life: progress, classification and evolution of subcellular traits. Am J Bot 91:1446–1480
Matheny PB, Wang Z, Binder M, Curtis JM, Lim YW, Nilsson RH, Hughes KW, Hofstetter V, Ammirati JF, Schoch CL, Langer E, Langer G, McLaughlin DJ, Wilson AW, Frøslev T, Ge ZW, Kerrigan RW, Slot JC, Yang ZL, Baroni TJ, Fischer M, Hosaka K, Matsuura K, Seidl MT, Vauras J, Hibbett DS (2007) Contributions of rpb2 and tef1 to the phylogeny of mushrooms and allies (Basidiomycota, Fungi). Mol Phyl Evol 43:430–451
Ministry of the Environment (Government of Japan) 2007. Revised Red Data List. http://www.env.go.jp/press/press.php?serial=8648. Accessed 5 June 2008
Moncalvo JM, Lutzoni FM, Rehner SA, Johnson J, Vilgalys R (2000) Phylogenetic relationships of agaric fungi based on nuclear large subunit ribosomal DNA sequences. Syst Biol 49:278–305
Moncalvo JM, Nilsson RH, Koster B, Dunham SM, Bernauer T, Matheny PB, Porter TM, Margaritescu S, Weiß M, Garnica S, Danell E, Langer G, Langer E, Larsson E, Larsson KH (2006) The cantharelloid clade: dealing with incongruent gene trees and phylogenetic reconstruction methods. Mycologia 98:937–948
Nylander JAA (2004) MrModeltest v2. Program distributed by the author. Evolutionary Biology Centre, Uppsala University
Ogura-Tsujita Y, Yukawa T (2008) High mycorrhizal specificity in a widespread mycoheterotrophic plant, Eulophia zollingeri (Orchidaceae). Am J Bot 95:93–97
Phillips RD, Barrett MD, Dixon KW, Hopper SD (2011a) Do mycorrhizal symbioses cause rarity in orchids? J Ecol 99:858–869
Phillips RD, Brown AP, Dixon KW, Hopper SD (2011b) Orchid biogeography and the factors associated with rarity in a biodiversity hotspot: the Southwest Australian Floristic Region. J Biogeogr 38:487–501
Pridgeon AM, Cribb P, Chase MW, Rasmussen FN (2006) Genera Orchidacearum: Epidendroideae (Part 1), vol 4. Oxford University Press, USA
Ramsay RR, Sivasithamparam K, Dixon KW (1986) Patterns of infection and endophytes associated with Western Australian orchids. Lindleyana 1:203–214
Rasmussen HN (1995) Terrestrial orchids: from seed to mycotrophic plant. Cambridge University Press, Cambridge
Rasmussen HN (2002) Recent developments in the study of orchid mycorrhiza. Plant Soil 244:149–163
Rasmussen HN, Rasmussen FN (2009) Orchid mycorrhiza: implications of a mycophagous life cycle. Oikos 118:334–345
Rasmussen HN, Whigham DF (1993) Seed ecology of dust seeds in situ: a new study technique and its application in terrestrial ecology. Am J Bot 80:1374–1378
Roberts DL (2003) Pollination biology: the role of sexual reproduction in orchid conservation. In: Dixon KW, Kell SP, Barrett RL, Cribb PJ (eds) Orchid conservation. Natural History Publications, Kota Kinabalu, pp 113–136
Ronquist F, Huelsenbeck JP (2003) MrBayes 3: bayesian phylogenetic inference under mixed models. Bioinformatics 19:1572–1574
Saitou N, Nei M (1987) The neighbor-joining method: a new method for reconstructing phylogenetic trees. Mol Biol Evol 4:406–425
Selosse MA, Setaro S, Glatard F, Urcelay C, Weiß M (2007) Sebacinales are common mycorrhizal associates of Ericaceae. New Phytol 174:864–878
Swarts ND, Dixon KW (2009) Terrestrial orchid conservation in the age of extinction. Ann Bot 104:543–556
Swarts ND, Sinclair EA, Francis A, Dixon KW (2010) Ecological specialization in mycorrhizal symbiosis leads to rarity in an endangered orchid. Mol Ecol 19:3226–3242
Swofford DL (2001) PAUP*: phylogenetic analysis using parsimony (*and other methods), version 4. 0b6. Sinauer, Sunderland
Taylor DL, McCormick MK (2008) Internal transcribed spacer primers and sequences for improved characterization of basidiomycetous orchid mycorrhizas. New Phytol 177:1020–1033
Thompson JD, Gibson TJ, Plewniak F, Jeanmougin F, Higgins DG (1997) The CLUSTAL_X windows interface: flexible strategies for multiple sequence alignment aided by quality analysis tools. Nucleic Acids Res 25:4876–4882
Veldre V (2011) Insights into the ecology of the Ceratobasidium–Thanatephorus complex based on phylogeny reconstruction using all publicly available ITS sequences. Master Thesis, Tartu University
Warcup JH (1971) Specificity of mycorrhizal association in some Australian terrestrial orchids. New Phytol 70:41–46
Weiß M, Selosse MA, Rexer KH, Urban A, Oberwinkler F (2004) Sebacinales: a hitherto overlooked cosm of heterobasidiomycetes with a broad mycorrhizal potential. Mycol Res 108:1003–1010
Weiß M, Sýkorová Z, Garnica S, Reiss K, Martos F, Krause C, Oberwinkler F, Bauer R, Redecker D (2011) Sebacinales everywhere: previously overlooked ubiquitous fungal endophytes. PLoS ONE, 6e16793
White TJ, Bruns TD, Taylor LS (1990) Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In: McInnis MA, Gelfand DH, Sninsky JJ, White TJ (eds) PCR protocols. A guide to method and application. Academic Press, San Diego, pp 315–322
Yukawa T, Ogura-Tsujita Y, Shefferson RP, Yokoyama J (2009) Mycorrhizal diversity in Apostasia (Orchidaceae) indicates the origin and evolution of orchid mycorrhiza. Am J Bot 96:1997–2009
Acknowledgments
The authors thank H. Abe and S. Fujimori for technical support in the laboratory, and M. Kubo and M. Saito for field assistance. This study was conducted as a part of research project of Ministry of the Environment, Japan, and is financially supported by a Grants-in-Aid to Scientific Research from the Japan Society for Promotion of Science (No. 21370038).
Author information
Authors and Affiliations
Corresponding author
Additional information
N. Nomura and Y. Ogura-Tsujita contributed equally.
Rights and permissions
About this article
Cite this article
Nomura, N., Ogura-Tsujita, Y., Gale, S.W. et al. The rare terrestrial orchid Nervilia nipponica consistently associates with a single group of novel mycobionts. J Plant Res 126, 613–623 (2013). https://doi.org/10.1007/s10265-013-0552-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10265-013-0552-8