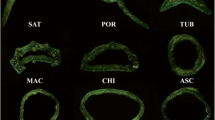
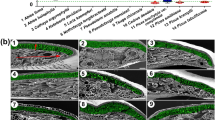
Abstract
An important objective in evolutionary developmental biology is to understand the molecular genetic mechanisms that have given rise to morphological diversity. Leaves in angiosperms generally develop as a flattened structure with clear adaxial–abaxial polarity. In monocots, however, a unifacial leaf has evolved in a number of divergent species, in which leaf blades consist of only the abaxial identity. The mechanism of unifacial leaf development has long been a matter of debate for comparative morphologists. However, the underlying molecular genetic mechanism remains unknown. Unifacial leaves would be useful materials for developmental studies of leaf-polarity specification. Moreover, these leaves offer unique opportunities to investigate important phenomena in evolutionary biology, such as repeated evolution or convergent evolution of similar morphological traits. Here we describe the potential of unifacial leaves for evolutionary developmental studies and present our recent approaches to understanding the mechanisms of unifacial leaf development and evolution using Juncus as a model system.



Similar content being viewed by others
References
Barkoulas M, Hay A, Kougioumoutzi E, Tsiantis M (2008) A developmental framework for dissected leaf formation in the Arabidopsis relative Cardamine hirsuta. Nat Genet 40:1136–1141
Blein T, Pulido A, Vialette-Guiraud A, Nikovics K, Morin H, Hay A, Johansen IE, Tsiantis M, Laufs P (2008) A conserved molecular framework for compound leaf development. Science 322:1835–1839
Bowman JL, Floyd SK (2008) Patterning and polarity in seed plant shoots. Annu Rev Plant Biol 59:67–88
Bowman JL, Eshed Y, Baum SF (2002) Establishment of polarity in angiosperm lateral organs. Trends Genet 18:134–141
Cameron KM (2005) Leave it to the leaves: a molecular phylogenetic study of Malaxideae (Epidendroideae, Orchidaceae). Am J Bot 92:1025–1032
Candela H, Johnston R, Gerhold A, Foster T, Hake S (2008) The milkweed pod1 gene encodes a KANADI protein that is required for abaxial/adaxial patterning in maize leaves. Plant Cell 20:2073–2087
Carlsward BS, Stern WL, Judd WS, Lucansky TW (1997) Comparative leaf anatomy and systematics in Dendrobium, sections Aporum and Rhizobium (Orchidaceae). Int J Plant Sci 158:332–342
Champagne C, Sinha N (2004) Compound leaves: equal to the sum of their parts? Development 131:4401–4412
Chitwood DH, Guo M, Nogueira FT, Timmermans MC (2007) Establishing leaf polarity: the role of small RNAs and positional signals in the shoot apex. Development 134:813–823
Cutler D (1969) Anatomy of the monocotyledons. IV. Juncales. Clarendon, Oxford
Gould SJ (2002) The structure of evolutionary biology. Belknap, Cambridge
Irish VF, Benfey PN (2004) Beyond Arabidopsis. Translational biology meets evolutionary developmental biology. Plant Physiol 135:611–614
Kaplan DR (1970) Comparative foliar histogenesis in Acorus calamus and its bearing on the phyllode theory of monocotyledonous leaves. Am J Bot 57:331–361
Kaplan DR (1975) Comparative developmental evaluation of the morphology of unifacial leaves in the monocotyledons. Bot Jahrb Syst 95:1–105
Kirschner J (2002a) Juncaceae 2: Juncus subg. Juncus, Species plantarum: flora of the world part 7. ABRS, Canberra
Kirschner J (2002b) Juncaceae 3: Juncus subg. Agathryon, Species plantarum: flora of the world part 8. ABRS, Canberra
Nandakumar R, Li C, Rogers S (2007) A stable and reproducible transformation system for the wetland monocot Juncus accuminatus (bulrush) mediated by Agrbacterium tumefaciens. In Vitro Plant 43:187–194
Piazza P, Jasinski S, Tsiantis M (2005) Evolution of leaf developmental mechanisms. New Phytol 167:693–710
Prud’homme B, Gompel N, Rokas A, Kassner VA, Williams TM, Yeh SD, True JR, Carroll SB (2006) Repeated morphological evolution through cis-regulatory changes in a pleiotropic gene. Nature 440:1050–1053
Raff RA (2000) Evo-devo: the evolution of a new discipline. Nat Rev Genet 1:74–79
Rudall PJ, Buzgo M (2002) Evolutionary history of the monocot leaf. In: Cronk QCB, Bateman RM, Hawkins JA (eds) Developmental genetics and plant evolution. Taylor & Francis, London, pp 431–458
Waites R, Hudson A (1995) Phantastica: a gene required for dorsoventrality of leaves in Antirrhinum majus. Development 121:2143–2154
West-Eberhard MJ (2003) Developmental plasticity and evolution. Oxford University Press, Oxford
Yoon HS, Baum DA (2004) Transgenic study of parallelism in plant morphological evolution. Proc Natl Acad Sci USA 101:6524–6529
Acknowledgments
We thank Dr. Futoshi Miyamoto (Tokyo University of Agriculture, Atsugi); the Royal Botanic Gardens, Kew; and the Botanical Gardens, Graduate School of Science, the University of Tokyo for providing plant material. This work was supported by a grant-in-aid for Creative Scientific Research to HT from the Japan Society for the Promotion of Science; by a grant-in-aid for Scientific Research on Priority Areas to HT; and a grant-in-aid for Young Scientists (B) to TY, from the Ministry of Education, Culture, Sports, Science and Technology of Japan; and by a Grant from the Sumitomo Foundation to TY.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yamaguchi, T., Tsukaya, H. Evolutionary and developmental studies of unifacial leaves in monocots: Juncus as a model system. J Plant Res 123, 35–41 (2010). https://doi.org/10.1007/s10265-009-0255-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10265-009-0255-3