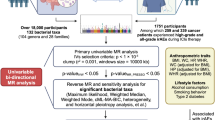
Abstract
Identifying patients at increased risk of immune-related adverse events (irAEs) facilitates safe application of immune checkpoint inhibitors (ICIs). This retrospective study aimed to determine the effect of age on the risk of irAEs in patients receiving ICIs and to identify potential mechanisms underlying age-related irAE risk differences. We analyzed reports of FDA Adverse Event Reporting System from July 1, 2014, to September 30, 2021. The information component ratio (ICΔ) was used to compare the irAE risk between older adults (> 65 years) and younger adults (25–65 years), of which the 95% confidential interval lower limit (ICΔ025) exceeding zero indicated significantly increased risk. We found that older adults had a significantly higher overall irAE risk than younger adults (ICΔ025 0.38), which was observed in almost all organ systems. We further analyzed the correlation between age-related irAE risks and age-related transcriptional changes to identify potential genes and pathways underlying age-related irAE risk differences. We found that genes significantly correlated with ICΔ were enriched in processes including extracellular matrix organization, regulation of myeloid leukocyte mediated immunity, and regulation of c-Jun N-terminal kinase (JNK) cascade. In addition, single-cell RNA sequencing analysis confirmed that genes involved in collagen-containing extracellular matrix and JNK cascade were significantly upregulated in myeloid cells from ICI-associated colitis tissues compared with ICI-treated colon tissues without colitis. In conclusion, older adults receiving ICIs have higher irAE risks than younger adults. Upregulation of genes involved in JNK cascade and collagen-containing extracellular matrix in myeloid cells may contribute to increased irAE risks in older adults.





Similar content being viewed by others
References
Ramos-Casals M, Brahmer JR, Callahan MK, et al. Immune-related adverse events of checkpoint inhibitors. Nat Rev Dis Primers. 2020;6(1):38.
Martins F, Sofiya L, Sykiotis GP, et al. Adverse effects of immune-checkpoint inhibitors: epidemiology, management and surveillance. Nat Rev Clin Oncol. 2019;16(9):563–80.
Wang DY, Salem JE, Cohen JV, et al. Fatal toxic effects associated with immune checkpoint inhibitors: a systematic review and meta-analysis. JAMA Oncol. 2018;4(12):1721–8.
Pilleron S, Alqurini N, Ferlay J, et al. International trends in cancer incidence in middle-aged and older adults in 44 countries. J Geriatr Oncol. 2022;13(3):346–55.
Hurria A, Togawa K, Mohile SG, et al. Predicting chemotherapy toxicity in older adults with cancer: a prospective multicenter study. J Clin Oncol. 2011;29(25):3457–65.
Hurria A, Mohile S, Gajra A, et al. Validation of a prediction tool for chemotherapy toxicity in older adults with cancer. J Clin Oncol. 2016;34(20):2366–71.
Shah KP, Song H, Ye F, et al. Demographic factors associated with toxicity in patients treated with anti-programmed cell death-1 therapy. Cancer Immunol Res. 2020;8(7):851–5.
Baldini C, Martin Romano P, Voisin AL, et al. Impact of aging on immune-related adverse events generated by anti-programmed death (ligand)PD-(L)1 therapies. Eur J Cancer. 2020;129:71–9.
Ksienski D, Wai ES, Croteau NS, et al. Association of age with differences in immune related adverse events and survival of patients with advanced nonsmall cell lung cancer receiving pembrolizumab or nivolumab. J Geriatr Oncol. 2020;11(5):807–13.
Thompson JA, Schneider BJ, Brahmer J, et al. Management of immunotherapy-related toxicities, version 1.2022, NCCN clinical practice guidelines in oncology. J Natl Compr Canc Netw. 2022;20(4):387–405.
Jing Y, Liu J, Ye Y, et al. Multi-omics prediction of immune-related adverse events during checkpoint immunotherapy. Nat Commun. 2020;11(1):4946.
Jing Y, Zhang Y, Wang J, et al. Association between sex and immune-related adverse events during immune checkpoint inhibitor therapy. J Natl Cancer Inst. 2021;113(10):1396–404.
Jing Y, Chen X, Li K, et al. Association of antibiotic treatment with immune-related adverse events in patients with cancer receiving immunotherapy. J Immunother Cancer. 2022;10(1):3779.
Jing Y, Yang J, Johnson DB, Moslehi JJ, Han L. Harnessing big data to characterize immune-related adverse events. Nat Rev Clin Oncol. 2022;19(4):269–80.
von Elm E, Altman DG, Egger M, et al. The strengthening the reporting of observational studies in epidemiology (STROBE) statement: guidelines for reporting observational studies. PLoS Med. 2007;4(10): e296.
Sarangdhar M, Tabar S, Schmidt C, et al. Data mining differential clinical outcomes associated with drug regimens using adverse event reporting data. Nat Biotechnol. 2016;34(7):697–700.
Schneider BJ, Naidoo J, Santomasso BD, et al. Management of immune-related adverse events in patients treated with immune checkpoint inhibitor therapy: ASCO guideline update. J Clin Oncol. 2021;39(36):4073–126.
Brown EG, Wood L, Wood S. The medical dictionary for regulatory activities (MedDRA). Drug Saf. 1999;20(2):109–17.
Lindquist M, Ståhl M, Bate A, Edwards IR, Meyboom RH. A retrospective evaluation of a data mining approach to aid finding new adverse drug reaction signals in the WHO international database. Drug Saf. 2000;23(6):533–42.
Norén GN, Hopstadius J, Bate A. Shrinkage observed-to-expected ratios for robust and transparent large-scale pattern discovery. Stat Methods Med Res. 2013;22(1):57–69.
Zhai Y, Ye X, Hu F, et al. Endocrine toxicity of immune checkpoint inhibitors: a real-world study leveraging US Food and drug administration adverse events reporting system. J Immunother Cancer. 2019;7(1):286.
Hopstadius, J., G.N. Norén, Robust discovery of local patterns: subsets and stratification in adverse drug reaction surveillance. In: Proceedings of the 2nd ACM SIGHIT international health informatics symposium. 2012, Association for computing machinery: Miami, Florida, USA. p. 265–274.
Sandberg L, Taavola H, Aoki Y, Chandler R, Norén GN. Risk factor considerations in statistical signal detection: using subgroup disproportionality to uncover risk groups for adverse drug reactions in VigiBase. Drug Saf. 2020;43(10):999–1009.
Wakao R, Lönnstedt IM, Aoki Y, Chandler RE. The use of subgroup disproportionality analyses to explore the sensitivity of a global database of individual case safety reports to known pharmacogenomic risk variants common in Japan. Drug Saf. 2021;44(6):681–97.
The genotype-tissue expression (GTEx) project. Nat Genet, 2013; 45(6): 580–5.
Ritchie ME, Phipson B, Wu D, et al. limma powers differential expression analyses for RNA-sequencing and microarray studies. Nucleic Acids Res. 2015;43(7): e47.
Wu T, Hu E, Xu S, et al. clusterProfiler 4.0: a universal enrichment tool for interpreting omics data. Innovation (Camb). 2021;2(3):100141.
Newman AM, Liu CL, Green MR, et al. Robust enumeration of cell subsets from tissue expression profiles. Nat Methods. 2015;12(5):453–7.
Hänzelmann S, Castelo R, Guinney J. GSVA: gene set variation analysis for microarray and RNA-seq data. BMC Bioinform. 2013;14:7.
Luoma AM, Suo S, Williams HL, et al. Molecular pathways of colon inflammation induced by cancer immunotherapy. Cell. 2020;182(3):655-671.e22.
Butler A, Hoffman P, Smibert P, Papalexi E, Satija R. Integrating single-cell transcriptomic data across different conditions, technologies, and species. Nat Biotechnol. 2018;36(5):411–20.
McGinnis CS, Murrow LM, Gartner ZJ. DoubletFinder: doublet detection in single-cell RNA sequencing data using artificial nearest neighbors. Cell Syst. 2019;8(4):329-337.e4.
Korsunsky I, Millard N, Fan J, et al. Fast, sensitive and accurate integration of single-cell data with harmony. Nat Methods. 2019;16(12):1289–96.
Kondo T, Okamoto I, Sato H, et al. Age-based efficacy and safety of nivolumab for recurrent or metastatic head and neck squamous cell carcinoma: a multicenter retrospective study. Asia Pac J Clin Oncol. 2020;16(6):340–7.
Quach HT, Dewan AK, Davis EJ, et al. Association of anti-programmed cell death 1 cutaneous toxic effects with outcomes in patients with advanced melanoma. JAMA Oncol. 2019;5(6):906–8.
Huang X, Tian T, Zhang Y, et al. Age-associated changes in adverse events arising from anti-PD-(L)1 therapy. Front Oncol. 2021;11: 619385.
Mikami T, Liaw B, Asada M, et al. Neuroimmunological adverse events associated with immune checkpoint inhibitor: a retrospective, pharmacovigilance study using FAERS database. J Neurooncol. 2021;152(1):135–44.
Solimando AG, Crudele L, Leone P, et al. Immune checkpoint inhibitor-related myositis: from biology to bedside. Int J Mol Sci. 2020;21(9):3054.
Anquetil C, Salem JE, Lebrun-Vignes B, et al. Immune checkpoint inhibitor-associated myositis: expanding the spectrum of cardiac complications of the immunotherapy revolution. Circulation. 2018;138(7):743–5.
Morad G, Helmink BA, Sharma P, Wargo JA. Hallmarks of response, resistance, and toxicity to immune checkpoint blockade. Cell. 2021;184(21):5309–37.
Henderson Berg MH, Del Rincón SV, Miller WH. Potential therapies for immune-related adverse events associated with immune checkpoint inhibition: from monoclonal antibodies to kinase inhibition. J Immunother Cancer. 2022;10(1):e003551.
Saferding V, Blüml S. Innate immunity as the trigger of systemic autoimmune diseases. J Autoimmun. 2020;110: 102382.
Dougan M, Luoma AM, Dougan SK, Wucherpfennig KW. Understanding and treating the inflammatory adverse events of cancer immunotherapy. Cell. 2021;184(6):1575–88.
Hu H, Zakharov PN, Peterson OJ, Unanue ER. Cytocidal macrophages in symbiosis with CD4 and CD8 T cells cause acute diabetes following checkpoint blockade of PD-1 in NOD mice. Proc Natl Acad Sci U S A. 2020;117(49):31319–30.
Santoro A, Bientinesi E, Monti D. Immunosenescence and inflammaging in the aging process: age-related diseases or longevity? Ageing Res Rev. 2021;71: 101422.
Dong C, Davis RJ, Flavell RA. MAP kinases in the immune response. Annu Rev Immunol. 2002;20:55–72.
Rincón M, Davis RJ. Regulation of the immune response by stress-activated protein kinases. Immunol Rev. 2009;228(1):212–24.
Okada M, Matsuzawa A, Yoshimura A, Ichijo H. The lysosome rupture-activated TAK1-JNK pathway regulates NLRP3 inflammasome activation. J Biol Chem. 2014;289(47):32926–36.
Hammouda, M.B., A.E. Ford, Y. Liu, J.Y. Zhang, The JNK Signaling Pathway in Inflammatory Skin Disorders and Cancer. Cells, 2020. 9(4).
Lai B, Wu CH, Lai JH. Activation of c-Jun N-terminal kinase, a potential therapeutic target in autoimmune arthritis. Cells. 2020;9(11):2466.
Li G, Qi W, Li X, et al. Recent advances in c-Jun N-terminal kinase (JNK) inhibitors. Curr Med Chem. 2021;28(3):607–27.
Popmihajlov Z, Sutherland DJ, Horan GS, et al. CC-90001, a c-Jun N-terminal kinase (JNK) inhibitor, in patients with pulmonary fibrosis: design of a phase 2, randomised, placebo-controlled trial. BMJ Open Respir Res. 2022;9(1):e001060.
Arthur JS, Ley SC. Mitogen-activated protein kinases in innate immunity. Nat Rev Immunol. 2013;13(9):679–92.
Hara H, Tsuchiya K, Kawamura I, et al. Phosphorylation of the adaptor ASC acts as a molecular switch that controls the formation of speck-like aggregates and inflammasome activity. Nat Immunol. 2013;14(12):1247–55.
Jo EK, Kim JK, Shin DM, Sasakawa C. Molecular mechanisms regulating NLRP3 inflammasome activation. Cell Mol Immunol. 2016;13(2):148–59.
Cirillo N, Prime SS. A scoping review of the role of metalloproteinases in the pathogenesis of autoimmune pemphigus and pemphigoid. Biomolecules. 2021;11(10):1506.
Costa S, Ragusa MA, Lo Buglio G, Scilabra SD, Nicosia A. The repertoire of tissue inhibitors of metalloproteases: evolution, regulation of extracellular matrix proteolysis, engineering and therapeutic challenges. Life (Basel). 2022;12(8):1145.
Smyth P, Sasiwachirangkul J, Williams R, Scott CJ. Cathepsin S (CTSS) activity in health and disease - a treasure trove of untapped clinical potential. Mol Aspects Med. 2022;88: 101106.
Bui TM, Wiesolek HL, Sumagin R. ICAM-1: a master regulator of cellular responses in inflammation, injury resolution, and tumorigenesis. J Leukoc Biol. 2020;108(3):787–99.
Pyka-Fościak G, Lis GJ, Litwin JA. Adhesion molecule profile and the effect of Anti-VLA-4 mAb treatment in experimental autoimmune encephalomyelitis, a mouse model of multiple sclerosis. Int J Mol Sci. 2022;23(9):4637.
da Rosa Franchi Santos LF, Costa NT, Maes M, Simão ANC, Dichi I. Influence of treatments on cell adhesion molecules in patients with systemic lupus erythematosus and rheumatoid arthritis: a review. Inflammopharmacology. 2020;28(2):363–84.
Márquez EJ, Chung CH, Marches R, et al. Sexual-dimorphism in human immune system aging. Nat Commun. 2020;11(1):751.
Bruijnen CP, Koldenhof JJ, Verheijden RJ, et al. Frailty and checkpoint inhibitor toxicity in older patients with melanoma. Cancer. 2022;128(14):2746–52.
Gomes F, Lorigan P, Woolley S, et al. A prospective cohort study on the safety of checkpoint inhibitors in older cancer patients - the ELDERS study. ESMO Open. 2021;6(1): 100042.
Kuksin M, Morel D, Aglave M, et al. Applications of single-cell and bulk RNA sequencing in onco-immunology. Eur J Cancer. 2021;149:193–210.
Acknowledgements
None
Funding
This work was supported by the National High Level Hospital Clinical Research Funding (2022-PUMCH-B-051).
Author information
Authors and Affiliations
Contributions
Conceptualization contributed by JRL, KLY, LZ, ZS, and CMB. Data curation and analysis contributed by KLY and JRL. Original draft writing contributed by KLY and JRL. Manuscript review and editing contributed by KLY, JRL, LZ, ZS, and CMB.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Ethical approval
This study was exempt from institutional board review because it only involved publicly available deidentified data.
Data availability
This study only involved publicly available deidentified data. Data of retrospective pharmacovigilance analysis were collected from the FDA Adverse Event Reporting System and were aggregated and downloaded from the AERSMine database (https://research.cchmc.org/aers/home). Data of tissue transcriptional analysis were downloaded from the GTEx portal (https://gtexportal.org). Data of single-cell RNA sequencing analysis were downloaded from the GEO database via the access number GSE144469 (https://www.ncbi.nlm.nih.gov/geo/).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yang, K., Li, J., Sun, Z. et al. Effect of age on the risk of immune-related adverse events in patients receiving immune checkpoint inhibitors. Clin Exp Med 23, 3907–3918 (2023). https://doi.org/10.1007/s10238-023-01055-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10238-023-01055-8