Abstract
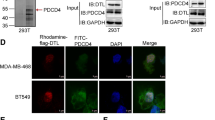
Carboxyl terminus of Hsc-70-interacting protein (CHIP), as U-box-type ubiquitin ligase, connects the chaperone and proteasome systems and plays a pivotal role in maintaining protein homeostasis in the cytoplasm. CHIP induces the ubiquitination and degradation of diverse oncogenic substrate proteins and therefore involves in the progression of tumorigenesis. In this study, the CHIP expression was examined in different human breast cancer cell lines and a group of breast cancer tissues. We found, for the first time, that CHIP expression was correlated with the molecular subtyping of breast cancer. CHIP was least expressed in the base-like subtype of breast cancer, which are triple-negative breast cancer (TNBC) breast cancer predominantly. Accordingly, CHIP expression was evidently decreased in the TNBC MDA-MB-231 breast cancer cell line. Enforced induction of CHIP in the MDA-MB-231 cells exerted no obvious influences on cellular growth and cell cycle. The apoptotic and proliferation cells in hCHIP cells were both reduced compared to the ctrl cells. The mRNA and protein expressions of the anti-apoptotic Bcl-2 and Bcl-xL were markedly increased in the hCHIP cells compared to that of the ctrl cells. The expression of RelA was significantly reduced in the nuclear extract in hCHIP cells compared to that in the ctrl cells. The protein expressions of IKKβ were markedly decreased in the hCHIP cells compared to the ctrl cells. The reduced cellular proliferation was largely due to the attenuated IKKβ-p65/NF-κB activity. Meanwhile, the invasion ability but not the migration ability was diminished when CHIP was overexpressed in the MDA-MB-231 cells. The activity of MMP2 but not MMP9 was significantly decreased in the hCHIP cells compared to the ctrl cells. Taken together, these observations here provide functional evidence for CHIP behaved as a tumor suppressor in the TNBC breast cancer cells. CHIP influenced diverse biological aspects of the MDA-MB-231 breast cancer cells. Importantly, CHIP expression is a useful indicator of the molecular subtyping of breast cancer.





Similar content being viewed by others
References
Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68(6):394–424. https://doi.org/10.3322/caac.21492.
Siegel RL, Miller KD, Jemal A. Cancer statistics, 2018. CA Cancer J Clin. 2018;68(1):7–30. https://doi.org/10.3322/caac.21442.
Oakman C, Viale G, Di Leo A. Management of triple negative breast cancer. Breast. 2010;19(5):312–21. https://doi.org/10.1016/j.breast.2010.03.026.
de Ruijter TC, Veeck J, de Hoon JP, van Engeland M, Tjan-Heijnen VC. Characteristics of triple-negative breast cancer. J Cancer Res Clin Oncol. 2011;137(2):183–92. https://doi.org/10.1007/s00432-010-0957-x.
Lee A, Djamgoz MBA. Triple negative breast cancer: emerging therapeutic modalities and novel combination therapies. Cancer Treat Rev. 2018;62:110–22. https://doi.org/10.1016/j.ctrv.2017.11.003.
Tomao F, Papa A, Zaccarelli E, Rossi L, Caruso D, Minozzi M, et al. Triple-negative breast cancer: new perspectives for targeted therapies. Onco Targets Ther. 2015;8:177–93. https://doi.org/10.2147/OTT.S67673.
Ballinger CA, Connell P, Wu Y, Hu Z, Thompson LJ, Yin LY, et al. Identification of CHIP, a novel tetratricopeptide repeat-containing protein that interacts with heat shock proteins and negatively regulates chaperone functions. Mol Cell Biol. 1999;19(6):4535–45. https://doi.org/10.1128/mcb.19.6.4535.
Sun C, Li HL, Shi ML, Liu QH, Bai J, Zheng JN. Diverse roles of C-terminal Hsp70-interacting protein (CHIP) in tumorigenesis. J Cancer Res Clin Oncol. 2014;140(2):189–97. https://doi.org/10.1007/s00432-013-1571-5.
Paul I, Ghosh MK. A CHIPotle in physiology and disease. Int J Biochem Cell Biol. 2015;58:37–52. https://doi.org/10.1016/j.biocel.2014.10.027.
Patani N, Jiang W, Newbold R, Mokbel K. Prognostic implications of carboxyl-terminus of Hsc70 interacting protein and lysyl-oxidase expression in human breast cancer. J Carcinog. 2010;9:9. https://doi.org/10.4103/1477-3163.72505.
Kajiro M, Hirota R, Nakajima Y, Kawanowa K, So-ma K, Ito I, et al. The ubiquitin ligase CHIP acts as an upstream regulator of oncogenic pathways. Nat Cell Biol. 2009;11(3):312–9. https://doi.org/10.1038/ncb1839.
Xu W, Marcu M, Yuan X, Mimnaugh E, Patterson C, Neckers L. Chaperone-dependent E3 ubiquitin ligase CHIP mediates a degradative pathway for c-ErbB2/Neu. Proc Natl Acad Sci U S A. 2002;99(20):12847–52. https://doi.org/10.1073/pnas.202365899.
Fan M, Park A, Nephew KP. CHIP (carboxyl terminus of Hsc70-interacting protein) promotes basal and geldanamycin-induced degradation of estrogen receptor-alpha. Mol Endocrinol. 2005;19(12):2901–14. https://doi.org/10.1210/me.2005-0111.
Yi X, Wei W, Wang SY, Du ZY, Xu YJ, Yu XD. Histone deacetylase inhibitor SAHA induces ERalpha degradation in breast cancer MCF-7 cells by CHIP-mediated ubiquitin pathway and inhibits survival signaling. Biochem Pharmacol. 2008;75(9):1697–705. https://doi.org/10.1016/j.bcp.2007.10.035.
Jang KW, Lee KH, Kim SH, Jin T, Choi EY, Jeon HJ, et al. Ubiquitin ligase CHIP induces TRAF2 proteasomal degradation and NF-kappaB inactivation to regulate breast cancer cell invasion. J Cell Biochem. 2011;112(12):3612–20. https://doi.org/10.1002/jcb.23292.
Hiyoshi H, Goto N, Tsuchiya M, Iida K, Nakajima Y, Hirata N, et al. 2-(4-Hydroxy-3-methoxyphenyl)-benzothiazole suppresses tumor progression and metastatic potential of breast cancer cells by inducing ubiquitin ligase CHIP. Sci Rep. 2014;4:7095. https://doi.org/10.1038/srep07095.
Xu J, Zhou J, Dai H, Liu F, Li W, Wang W, et al. CHIP functions as an oncogene by promoting colorectal cancer metastasis via activation of MAPK and AKT signaling and suppression of E-cadherin. J Transl Med. 2018;16(1):169. https://doi.org/10.1186/s12967-018-1540-5.
McDonough H, Patterson C. CHIP: a link between the chaperone and proteasome systems. Cell Stress Chaperones. 2003;8(4):303–8. https://doi.org/10.1379/1466-1268(2003)008%3c0303:calbtc%3e2.0.co;2.
Paul I, Ahmed SF, Bhowmik A, Deb S, Ghosh MK. The ubiquitin ligase CHIP regulates c-Myc stability and transcriptional activity. Oncogene. 2013;32(10):1284–95. https://doi.org/10.1038/onc.2012.144.
Hayden MS, Ghosh S. NF-kappaB, the first quarter-century: remarkable progress and outstanding questions. Genes Dev. 2012;26(3):203–34. https://doi.org/10.1101/gad.183434.111.
Wang S, Wu X, Zhang J, Chen Y, Xu J, Xia X, et al. CHIP functions as a novel suppressor of tumour angiogenesis with prognostic significance in human gastric cancer. Gut. 2013;62(4):496–508. https://doi.org/10.1136/gutjnl-2011-301522.
Wang Y, Ren F, Wang Y, Feng Y, Wang D, Jia B, et al. CHIP/Stub1 functions as a tumor suppressor and represses NF-kappaB-mediated signaling in colorectal cancer. Carcinogenesis. 2014;35(5):983–91. https://doi.org/10.1093/carcin/bgt393.
Liu F, Zhou J, Zhou P, Chen W, Guo F. The ubiquitin ligase CHIP inactivates NF-kappaB signaling and impairs the ability of migration and invasion in gastric cancer cells. Int J Oncol. 2015;46(5):2096–106. https://doi.org/10.3892/ijo.2015.2893.
Bozzuto G, Ruggieri P, Molinari A. Molecular aspects of tumor cell migration and invasion. Ann Ist Super Sanita. 2010;46(1):66–80. https://doi.org/10.4415/ANN_10_01_09.
Wang T, Yang J, Xu J, Li J, Cao Z, Zhou L, et al. CHIP is a novel tumor suppressor in pancreatic cancer through targeting EGFR. Oncotarget. 2014;5(7):1969–86. https://doi.org/10.18632/oncotarget.1890.
Cao Z, Xu J, Huang H, Shen P, You L, Zhou L, et al. MiR-1178 promotes the proliferation, G1/S transition, migration and invasion of pancreatic cancer cells by targeting CHIP. PLoS ONE. 2015;10(1):e0116934. https://doi.org/10.1371/journal.pone.0116934.
Choi YN, Lee SK, Seo TW, Lee JS, Yoo SJ. C-Terminus of Hsc70-interacting protein regulates profilin1 and breast cancer cell migration. Biochem Biophys Res Commun. 2014;446(4):1060–6. https://doi.org/10.1016/j.bbrc.2014.03.061.
Ding Z, Joy M, Bhargava R, Gunsaulus M, Lakshman N, Miron-Mendoza M, et al. Profilin-1 downregulation has contrasting effects on early vs late steps of breast cancer metastasis. Oncogene. 2014;33(16):2065–74. https://doi.org/10.1038/onc.2013.166.
Patterson C, Ronnebaum S. Breast cancer quality control. Nat Cell Biol. 2009;11(3):239–41. https://doi.org/10.1038/ncb0309-239.
Guiu S, Michiels S, Andre F, Cortes J, Denkert C, Di Leo A, et al. Molecular subclasses of breast cancer: how do we define them? The IMPAKT 2012 working group statement. Ann Oncol. 2012;23(12):2997–3006. https://doi.org/10.1093/annonc/mds586.
Parker JS, Mullins M, Cheang MC, Leung S, Voduc D, Vickery T, et al. Supervised risk predictor of breast cancer based on intrinsic subtypes. J Clin Oncol. 2009;27(8):1160–7. https://doi.org/10.1200/JCO.2008.18.1370.
Goldhirsch A, Wood WC, Coates AS, Gelber RD, Thurlimann B, Senn HJ, et al. Strategies for subtypes—dealing with the diversity of breast cancer: highlights of the St. Gallen International Expert Consensus on the Primary Therapy of Early Breast Cancer 2011. Ann Oncol. 2011;22(8):1736–47. https://doi.org/10.1093/annonc/mdr304.
Avalos-Navarro G, Munoz-Valle JF, Daneri-Navarro A, Quintero-Ramos A, Franco-Topete RA, Moran-Mendoza AJ, et al. Circulating soluble levels of MIF in women with breast cancer in the molecular subtypes: relationship with Th17 cytokine profile. Clin Exp Med. 2019;19(3):385–91. https://doi.org/10.1007/s10238-019-00559-6.
Haritos C, Michaelidou K, Mavridis K, Missitzis I, Ardavanis A, Griniatsos J, et al. Kallikrein-related peptidase 6 (KLK6) expression differentiates tumor subtypes and predicts clinical outcome in breast cancer patients. Clin Exp Med. 2018;18(2):203–13. https://doi.org/10.1007/s10238-018-0487-4.
Zhao X, Rodland EA, Tibshirani R, Plevritis S. Molecular subtyping for clinically defined breast cancer subgroups. Breast Cancer Res. 2015;17:29. https://doi.org/10.1186/s13058-015-0520-4.
Foulkes WD, Smith IE, Reis-Filho JS. Triple-negative breast cancer. N Engl J Med. 2010;363(20):1938–48. https://doi.org/10.1056/NEJMra1001389.
Acknowledgements
This study was funded by grants from Suzhou Natural Science Foundation (Grant Number SS201875), Special Technical Project of Diagnosis and Treatment of Key Clinical Diseases of Suzhou (Grant Number LCZX201813), the National Natural Science Foundation of China (Grant Number 81400154), the Project of invigorating Health Care through Science, Technology and Education, Jiangsu Provincial Medical Youth Talent (Grant Number QNRC2016725), and Guiding Projects of Suzhou Science and Technology Plan (Grant Number SYSD2016112).
Author information
Authors and Affiliations
Contributions
FG and ZJH designed the research. JJX and HW performed the research and analyzed the data. WJL, KLL and TLZ collected the samples and performed the research. FG and JJX wrote the paper. All authors have read and approved the final version of this manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the Ethics Committee of Nanjing Medical University Affiliated Suzhou Hospital.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Xu, J., Wang, H., Li, W. et al. E3 ubiquitin ligase CHIP attenuates cellular proliferation and invasion abilities in triple-negative breast cancer cells. Clin Exp Med 20, 109–119 (2020). https://doi.org/10.1007/s10238-019-00594-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10238-019-00594-3