Abstract
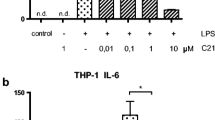
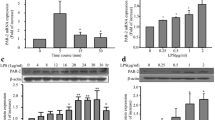
Chemokines play essential roles during inflammatory responses and in pathogenesis of inflammatory diseases. Monocyte chemotactic protein-1 (MCP-1) is a critical chemokine in the development of atherosclerosis and acute cardiovascular syndromes. MCP-1, by its chemotactic activity, causes diapedesis of monocytes from the lumen to the subendothelial space that leads to atherosclerotic plaque formation. Prostaglandin I2 (PGI2) analogs are used clinically for patients with pulmonary hypertension and have anti-inflammatory effects. However, little is known about the effect of PGI2 analogs on the MCP-1 production in human monocytes and macrophages. We investigated the effects of three conventional (iloprost, beraprost and treprostinil) and one new (ONO-1301) PGI2 analogs, on the expression of MCP-1 expression in human monocytes and macrophages. Human monocyte cell line, THP-1 cell, was treated with PGI2 analogs after LPS stimulation. Supernatants were harvested to measure MCP-1 levels and measured by ELISA. To explore which receptors involved the effects of PGI2 analogs on the expression of MCP-1 expression, IP and EP, PPAR-α and PPAR-γ receptor antagonists were used. Forskolin, a cAMP activator, was used to further confirm the involvement of cAMP on MCP-1 production in human monocytes. Three PGI2 analogs suppressed LPS-induced MCP-1 production in THP-1 cells and THP-1-induced macrophages. Higher concentrations of ONO-1301 also had the suppressive effect. CAY 10449, an IP receptor antagonist, could reverse the effects on MCP-1 production of iloprost on THP-1 cells. Other reported PGI2 receptor antagonists including EP1, EP2, EP4, PPAR-α and PPAR-γ antagonists could not reverse the effect. Forskolin, a cAMP activator, also suppressed MCP-1 production in THP-1 cells. PGI2 analogs suppressed LPS-induced MCP-1 production in human monocytes and macrophages via the IP receptor and cAMP pathway. The new PGI2 analog (ONO-1301) was not better than conventional PGI2 analog in the suppression of MCP-1 production in human monocytes.








Similar content being viewed by others
References
Luster AD. Chemokines: chemotactic cytokines that mediate inflammation. N Engl J Med. 1998;338:436–45.
Davies MJ. The pathophysiology of acute coronary syndromes. Heart. 2000;83:361–6.
Zhong L, Chen WQ, Ji XP, Zhang M, Zhao YX, Yao GH, et al. Dominant-negative mutation of monocyte chemoattractant protein-1 prevents vulnerable plaques from rupture in rabbits independent of serum lipid levels. J Cell Mol Med. 2008;12:2362–71.
Mori E, Komori K, Yamaoka T, Tanii M, Kataoka C, Takeshita A, et al. Essential role of monocyte chemoattractant protein-1 in development of restenotic changes (neointimal hyperplasia and constrictive remodeling) after balloon angioplasty in hypercholesterolemic rabbits. Circulation. 2002;105:2905–10.
Li J, Guo Y, Luan X, Qi T, Li D, Chen Y, Ji X, Zhang Y, Chen W. Independent roles of monocyte chemoattractant protein-1, regulated on activation, normal T-cell expressed and secreted and fractalkine in the vulnerability of coronary atherosclerotic plaques. Circ J. 2012;76:2167–73.
Kaya Z, Katus HA, Rose NR. Cardiac troponins and autoimmunity: their role in the pathogenesis of myocarditis and of heart failure. Clin Immunol. 2010;134:80–8.
Niu J, Kolattukudy PE. Role of MCP-1 in cardiovascular disease: molecular mechanisms and clinical implications. Clin Sci (Lond). 2009;117:95–109.
Warner TD, Mitchell JA. Cyclooxygenases: new forms, new inhibitors, and lessons from the clinic. FASEB J. 2004;18:790–804.
Helliwell RJ, Adams LF, Mitchell MD. Prostaglandin synthases: recent developments and a novel hypothesis. Prostaglandins Leukot Essent Fatty Acids. 2004;70:101–13.
Kunikata T, Yamane H, Segi E, et al. Suppression of allergic inflammation by the prostaglandin E receptor subtype EP3. Nat Immunol. 2005;6:524–31.
Aronoff DM, Peres CM, Serezani CH, Ballinger MN, Carstens JK, Coleman N, Moore BB, Peebles RS, Faccioli LH, Peters-Golden M. Synthetic prostacyclin analogs differentially regulate macrophage function via distinct analog-receptor binding specificities. J Immunol. 2007;178:1628–34.
Idzko M, Hammad H, van Nimwegen M, Kool M, Vos N, Hoogsteden HC, Lambrecht BN. Inhaled iloprost suppresses the cardinal features of asthma via inhibition of airway dendritic cell function. J Clin Invest. 2007;117:464–72.
Hung CH, Chu YT, Suen JL, Lee MS, Chang HW, Lo YC, Jong YJ. Regulation of cytokine expression in human plasmacytoid dendritic cells by prostaglandin I2 analogues. Eur Respir J. 2009;33:405–10.
Suzuki J, Ogawa M, Sakai Y, Hirata Y, Isobe M, Nagai R. A prostacycline analog prevents chronic myocardial remodeling in murine cardiac allografts. Int Heart J. 2012;53:64–7.
Daigneault M, Preston JA, Marriott HM, Whyte MK, Dockrell DH. The identification of markers of macrophage differentiation in PMA-stimulated THP-1 cells and monocyte-derived macrophages. PLoS ONE. 2010;5:e8668.
Kuo CH, Ko YC, Yang SN, Chu YT, Wang WL, Huang SK, Chen HN, Wei WJ, Jong YJ, Hung CH. Effects of PGI2 analogues on Th1- and Th2-related chemokines in monocytes via epigenetic regulation. J Mol Med. 2011;89:29–41.
Zhou W, Hashimoto K, Goleniewska K, O’Neal JF, Ji S, Blackwell TS, Fitzgerald GA, Egan KM, Geraci MW, Peebles RS Jr. Prostaglandin I2 analogs inhibit proinflammatory cytokine production and T cell stimulatory function of dendritic cells. J Immunol. 2007;178:702–10.
Warner TD, Mitchell JA. Cyclooxygenases: new forms, new inhibitors, and lessons from the clinic. FASEB J. 2004;18:790–804.
Idzko M, Hammad H, van Nimwegen M, Kool M, Vos N, Hoogsteden HC, Lambrecht BN. Inhaled iloprost suppresses the cardinal features of asthma via inhibition of airway dendritic cell function. J Clin Invest. 2007;117:464–72.
Hoeper MM, et al. Long-term treatment of primary pulmonary hypertension with aerosolized iloprost, a prostacyclin analogue. N Engl J Med. 2000;342:1866–70.
Ohman MK, Eitzman DT. Targeting MCP-1 to reduce vascular complications of obesity. Recent Pat Cardiovasc Drug Discov. 2009;4:164–76.
Daissormont IT, Kraaijeveld AO, Biessen EA. Chemokines as therapeutic targets for atherosclerotic plaque destabilization and rupture. Future Cardiol. 2009;5:273–84.
Grant SM, Goa KL. Iloprost: a review of its pharmacodynamic and pharmacokinetic properties, and therapeutic potential in peripheral vascular disease, myocardial ischaemia and extracorporeal circulation procedures. Drugs. 1992;43:889–924.
Aronoff DM, Peres CM, Serezani CH, Ballinger MN, Carstens JK, Coleman N, Moore BB, Peebles RS, Faccioli LH, Peters-Golden M. Synthetic prostacyclin analogs differentially regulate macrophage function via distinct analog–receptor binding specificities. J Immunol. 2007;178:1628–34.
Narumiya S, Sugimoto Y, Ushikubi F. Prostanoid receptors: structures, properties, and functions. Physiol Rev. 1999;79:1193–226.
Suzuki J, Ogawa M, Sakai Y, Hirata Y, Isobe M, Nagai R. A prostacycline analog prevents chronic myocardial remodeling in murine cardiac allografts. Int Heart J. 2012;53:64–7.
Acknowledgments
This study was supported by grants from Medical Research Fund (No. 101-06 and 101-03) of Kaohsiung Armed Forces General Hospital and from National Science Council (NSC 102-2314-B-037-052 and NSC 102-2314-B-037-048) of the Republic of China, and Kaohsiung Medical University Hospital Research Foundation KMUH102-2T04, grants from Kaohsiung Municipal Ta-Tung Hospital KMTTH-101-009, KMTTH-102-002, KMTTH 102-008 and grants from Kaohsiung Municipal Hsiao-Kang Hospital, Kmhk-102-007.
Conflict of Interest
All authors declare no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Ming-Kai Tsai, Chong-Chao Hsieh, Chang-Hung Kuo and Chih-Hsing Hung have contributed equally to this work.
Rights and permissions
About this article
Cite this article
Tsai, MK., Hsieh, CC., Kuo, HF. et al. Effect of prostaglandin I2 analogs on monocyte chemoattractant protein-1 in human monocyte and macrophage. Clin Exp Med 15, 245–253 (2015). https://doi.org/10.1007/s10238-014-0304-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10238-014-0304-7