Abstract
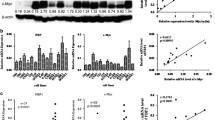
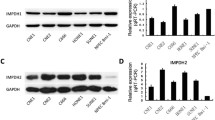
Nasopharyngeal carcinoma (NPC) is a very common head and neck cancer in southern china. Despite advances in surgical and chemotherapeutic approaches, its prognosis is still not promising. Hedgehog signaling pathway was reported to be involved in a number of cancers including head and neck. However, it remains unclear regarding the role of this pathway in NPC. By real-time PCR, we found Ptch1, Smo, and Gli-1 were expressed in all human nasopharyngeal epithelial tissues and cell lines. Compared with nasopharyngeal normal epithelial tissues, the mRNA expression level of Gli-1 was higher in carcinoma and nasopharyngitis (NPI) epithelial tissues. While compared with nasopharyngitis epithelia, the mRNA expression level of Ptch1 was lower in carcinoma epithelia and normal epithelia. The expressions of Smo mRNA were not significantly different among these epithelial tissues. Immunohistochemistry analysis revealed that the expression level of Gli-1 was higher in NPC than NPI. Thus, our data indicated that aberrant activation of hedgehog pathway in NPC. Furthermore, blocking the pathway with cyclopamine inhibited the proliferation of NPC epithelia cell lines. In addition, blockade of the pathway in three NPC cell lines with cyclopamine-induced tumor cell apoptosis. The transcription of hedgehog target genes also is inhibited by cyclopamine. These data suggested that hedgehog pathway may sustain nasopharyngeal tumor growth. Our data demonstrated that hedgehog signaling pathway was involved in NPC pathogenesis and might be a novel therapeutic target for NPC.





Similar content being viewed by others
References
Yu MC, Yuan JM (2002) Epidemiology of nasopharyngeal carcinoma. Semin Cancer Biol 12:421–429
Zülch KJ (1979) Histological typing of tumours of the central nervous system. World Health Organization, Geneva
Xiong W, Zeng ZY, Xia JH, Xia K, Shen SR, Li XL, Hu DX, Tan C, Xiang JJ, Zhou J, Deng H, Fan SQ, Li WF, Wang R, Zhou M, Zhu SG, Lu HB, Qian J, Zhang BC, Wang JR, Ma J, Xiao BY, Huang H, Zhang QH, Zhou YH, Luo XM, Zhou HD, Yang YX, Dai HP, Feng GY, Pan Q, Wu LQ, He L, Li GY (2004) A susceptibility locus at chromosome 3p21 linked to familial nasopharyngeal carcinoma. Cancer Res 64:1972–1974
Zeng YX, Jia WH (2002) Familial nasopharyngeal carcinoma. Semin Cancer Biol 12:443–450
Raab-Traub N (1992) Epstein-Barr virus infection in nasopharyngeal carcinoma. Infect Agents Dis 1:173–184
Hording U, Nielsen HW, Albeck H, Daugaard S (1993) Nasopharyngeal carcinoma: histopathological types and association with Epstein-Barr virus. Eur J Cancer B Oral Oncol 29B:137–139
Raab-Traub N, Flynn K, Pearson G, Huang A, Levine P, Lanier A, Pagano J (1987) The differentiated form of nasopharyngeal carcinoma contains Epstein-Barr virus DNA. Int J Cancer 39:25–29
Cheng SH, Jian JJ, Tsai SY, Yen KL, Chu NM, Chan KY, Tan TD, Cheng JC, Leu SY, Hsieh CY, Huang AT (2000) Long-term survival of nasopharyngeal carcinoma following concomitant radiotherapy and chemotherapy. Int J Radiat Oncol Biol Phys 48:1323–1330
Yamashita S, Kondo M, Hashimoto S (1985) Squamous cell carcinoma of the nasopharynx. An analysis of failure patterns after radiation therapy. Acta Radiol Oncol 24:315–320
Nusslein-Volhard C, Wieschaus E (1980) Mutations affecting segment number and polarity in Drosophila. Nature 287:795–801
Pasca DMM, Hebrok M (2003) Hedgehog signalling in cancer formation and maintenance. Nat Rev Cancer 3:903–911
Taipale J, Beachy PA (2001) The Hedgehog and Wnt signalling pathways in cancer. Nature 411:349–354
Marigo V, Roberts DJ, Lee SM, Tsukurov O, Levi T, Gastier JM, Epstein DJ, Gilbert DJ, Copeland NG, Seidman CE, Et A (1995) Cloning, expression, and chromosomal location of SHH and IHH: two human homologues of the Drosophila segment polarity gene hedgehog. Genomics 28:44–51
Echelard Y, Epstein DJ, St-Jacques B, Shen L, Mohler J, McMahon JA, McMahon AP (1993) Sonic hedgehog, a member of a family of putative signaling molecules, is implicated in the regulation of CNS polarity. Cell 75:1417–1430
Ingham PW, McMahon AP (2001) Hedgehog signaling in animal development: paradigms and principles. Genes Dev 15:3059–3087
Sheng T, Li C, Zhang X, Chi S, He N, Chen K, McCormick F, Gatalica Z, Xie J (2004) Activation of the hedgehog pathway in advanced prostate cancer. Mol Cancer 3:29
Thayer S (2004) The emerging role of the hedgehog signaling pathway in gastrointestinal cancers. Clin Adv Hematol Oncol 2:17, 20, 63
Watkins DN, Berman DM, Burkholder SG, Wang B, Beachy PA, Baylin SB (2003) Hedgehog signalling within airway epithelial progenitors and in small-cell lung cancer. Nature 422:313–317
Thayer SP, di Magliano MP, Heiser PW, Nielsen CM, Roberts DJ, Lauwers GY, Qi YP, Gysin S, Fernandez-del CC, Yajnik V, Antoniu B, McMahon M, Warshaw AL, Hebrok M (2003) Hedgehog is an early and late mediator of pancreatic cancer tumorigenesis. Nature 425:851–856
Patil MA, Zhang J, Ho C, Cheung ST, Fan ST, Chen X (2006) Hedgehog signaling in human hepatocellular carcinoma. Cancer Biol Ther 5:111–117
Xiao BY, Dang H, Gan JY, Cai Q, Zhang GP, Chang H (2010) Expression of PTCH-1 and SMO mRNA in nasopharyngeal carcinoma. Xi Bao Yu Fen Zi Mian Yi Xue Za Zhi 26:955–958
Chen JK, Taipale J, Cooper MK, Beachy PA (2002) Inhibition of Hedgehog signaling by direct binding of cyclopamine to Smoothened. Genes Dev 16:2743–2748
Kubo M, Nakamura M, Tasaki A, Yamanaka N, Nakashima H, Nomura M, Kuroki S, Katano M (2004) Hedgehog signaling pathway is a new therapeutic target for patients with breast cancer. Cancer Res 64:6071–6074
Lee KR, Kozukue N, Han JS, Park JH, Chang EY, Baek EJ, Chang JS, Friedman M (2004) Glycoalkaloids and metabolites inhibit the growth of human colon (HT29) and liver (HepG2) cancer cells. J Agric Food Chem 52:2832–2839
Berman DM, Karhadkar SS, Hallahan AR, Pritchard JI, Eberhart CG, Watkins DN, Chen JK, Cooper MK, Taipale J, Olson JM, Beachy PA (2002) Medulloblastoma growth inhibition by hedgehog pathway blockade. Science 297:1559–1561
Lee J, Platt KA, Censullo P, Ruiz IAA (1997) Gli1 is a target of Sonic hedgehog that induces ventral neural tube development. Development 124:2537–2552
Marigo V, Tabin CJ (1996) Regulation of patched by sonic hedgehog in the developing neural tube. Proc Natl Acad Sci USA 93:9346–9351
Duman-Scheel M, Weng L, Xin S, Du W (2002) Hedgehog regulates cell growth and proliferation by inducing cyclin D and cyclin E. Nature 417:299–304
Brennan B (2006) Nasopharyngeal carcinoma. Orphanet J Rare Dis 1:23
Bei JX, Li Y, Jia WH, Feng BJ, Zhou G, Chen LZ, Feng QS, Low HQ, Zhang H, He F, Tai ES, Kang T, Liu ET, Liu J, Zeng YX (2010) A genome-wide association study of nasopharyngeal carcinoma identifies three new susceptibility loci. Nat Genet 42:599–603
Kasper M, Regl G, Frischauf AM, Aberger F (2006) GLI transcription factors: mediators of oncogenic Hedgehog signalling. Eur J Cancer 42:437–445
Rubin LL, de Sauvage FJ (2006) Targeting the Hedgehog pathway in cancer. Nat Rev Drug Discov 5:1026–1033
Liao X, Siu MK, Au CW, Wong ES, Chan HY, Ip PP, Ngan HY, Cheung AN (2009) Aberrant activation of hedgehog signaling pathway in ovarian cancers: effect on prognosis, cell invasion and differentiation. Carcinogenesis 30:131–140
Dahmane N, Lee J, Robins P, Heller P, Ruiz IAA (1997) Activation of the transcription factor Gli1 and the Sonic hedgehog signalling pathway in skin tumours. Nature 389:876–881
Chung CH, Dignam JJ, Hammond ME, Klimowicz AC, Petrillo SK, Magliocco A, Jordan R, Trotti A, Spencer S, Cooper JS, Le QT, Ang KK (2011) Glioma-associated oncogene family zinc finger 1 expression and metastasis in patients with head and neck squamous cell carcinoma treated with radiation therapy (RTOG 9003). J Clin Oncol 29:1326–1334
Buttitta L, Mo R, Hui CC, Fan CM (2003) Interplays of Gli2 and Gli3 and their requirement in mediating Shh-dependent sclerotome induction. Development 130:6233–6243
Aza-Blanc P, Lin HY, Ruiz IAA, Kornberg TB (2000) Expression of the vertebrate Gli proteins in Drosophila reveals a distribution of activator and repressor activities. Development 127:4293–4301
Steg A, Amm HM, Novak Z, Frost AR, Johnson MR (2010) Gli3 mediates cell survival and sensitivity to cyclopamine in pancreatic cancer. Cancer Biol Ther 10:893–902
Acknowledgments
This work was partially supported by the Science and Technology Foundation of Technology-bureau of Guangdong province (2008B030301136, 2010B031500028). We thank the Center Laboratory of Guangzhou First Municipal People’s Hospital, for technical support in our experiment and analysis. Thanks to Guangzhou Institute of Respiratory Disease for real-time PCR and Immunohistochemistry experiment and analysis. Thanks to all of the hospital provide nasopharyngeal epithelia samples. Thanks to Doctor Xiangbin Xing for the help of writing the paper. We are grateful to all the participants that made this study possible.
Conflict of interest
All authors disclose any association that poses a conflict of interest in connection with the manuscript.
Author information
Authors and Affiliations
Corresponding authors
Additional information
The authors Yongjian Yue, Weide Zhong, and Genwang Pei contributed equally to this work.
Rights and permissions
About this article
Cite this article
Yue, Y., Zhong, W., Pei, G. et al. Aberrant activation of Hedgehog pathway in Nasopharyngeal carcinoma. Clin Exp Med 13, 315–322 (2013). https://doi.org/10.1007/s10238-012-0198-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10238-012-0198-1