Abstract
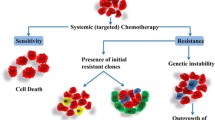
Oncologic diseases are among leading cause of mortality in developed countries. Despite significant progress, the use of standard cytotoxic chemotherapy has reached a therapeutical plateau. Currently, the process of selecting chemotherapy represents a trial and error method neglecting biological individuality of tumor and its bearer. The improvement of treatment results is expected from ex vivo drug sensitivity testing which may allow to choose the most effective drug for individual patient and to exclude agents to which the tumor cells exert resistance. New techniques and rapidly increasing knowledge about the molecular basis of malignant diseases provide important opportunities for the future of chemotherapy. This paper reviews current methods used to test the resistance of tumor cells to a panel of anticancer agents in vitro. In addition, we focused on the in vitro MTT assay which represents one of major technique for testing of tumor cell resistance to anticancer agents.

Similar content being viewed by others
References
Edward C, DeVita VT (2005) Principles of medical oncology. In: DeVita VT, Hellman S, Rosenberg S. Cancer principles & practices of oncology, vol 1. Lippincott, Williams & Wilkins, McGraw-Hill Companies, Philadelphia
Hatok J, Račay P, Hudeček J, Dobrota D (2006) Genes of multidrug resistance in haematological malignancies. Biologia 61:247–256
Bown NP, Reid MM, Malcolm AJ, Davison EV, Craft AW, Pearson AD (1994) Cytogenetic abnormalities of small round cell tumours. Med Pediatr Oncol 23:124–129
Zubor P, Kajo K, Stanclova A, Szunyogh N, Galo S, Dussan CA, Minarik G, Visnovsky J, Danko J (2008) Human epithelial growth factor receptor 2[Ile655Val] polymorphism and risk of breast fibroadenoma. Eur J Cancer Prev 17:33–38
Hamburger AW (1981) Use of in vitro tests in predictive cancer chemotherapy. J Natl Cancer Inst 66:981–988
Massaro EJ, Elstein KH, Zucker RM, Bair KW (1989) Limitations of the fluorescent probe viability assay. Mol Toxicol 2:271–284
Pieters R, Loonen AH, Huismans DR, Broekema GJ, Dirven MW, Heyenbrok MW, Hählen K, Veerman AJ (1990) In vitro drug sensitivity of cells from children with leukemia using the MTT assay with improved culture conditions. Blood 76:2327–2336
Möllgård L, Prenkert M, Smolowicz A, Paul C, Tidefelt U (2003) In vitro chemosensitivity testing of selected myeloid cells in acute myeloid leukemia. Leuk Lymphoma 44:783–789
Von Hoff DD, Sandbach JF, Clark GM, Turner JN, Forseth BF, Piccart MJ, Colombo N, Muggia FM (1990) Selection of cancer chemotherapy for a patient by an in vitro assay versus a clinician. J Natl Cancer Inst 82:110–116
Kern DH (1998) Heterogeneity of drug resistance in human breast and ovarian cancers. Cancer J Sci Am 4:41–45
Samson DJ, Seidenfeld J, Ziegler K, Aronson N (2004) Chemotherapy sensitivity and resistance assays: a systematic review. J Clin Oncol 22:3618–3630
Schrag D, Garewal HS, Burstein HJ, Samson DJ, Von Hoff DD, Somerfield MR, ASCO Working Group on Chemotherapy Sensitivity and Resistance Assays (2004) American Society of Clinical Oncology Technology Assessment: chemotherapy sensitivity and resistance assays. J Clin Oncol 22:3631–3638
Bosanquet AG, Bell PB (1996) Enhanced ex vivo drug sensitivity testing of chronic lymphocytic leukaemia using refined DiSC assay methodology. Leuk Res 20:143–153
Weisenthal LM, Dill PL, Kurnick NB, Lippman ME (1983) Comparison of dye exclusion assays with a clonogenic assay in the determination of drug-induced cytotoxicity. Cancer Res 43:258–264
Mason JM, Drummond MF, Bosanquet AG, Sheldon TA, The DiSC assay (1999) A cost-effective guide to treatment for chronic lymphocytic leukemia? Int J Technol Assess Health Care 15:173–184
Rhedin AS, Tidefelt U, Jönsson K, Lundin A, Paul C (1993) Comparison of a bioluminescence assay with differential staining cytotoxicity for cytostatic drug testing in vitro in human leukemic cells. Leuk Res 17:271–276
DeVita VT, Hellman S, Rosenberg SA (2004) Cancer: principles and practice of oncology. Lippincott Williams & Wilkins, Philadelphia
Ichihashi H, Kondo T, Sakakibara S, Akiyama S, Watanabe T (1984) Application of radioactive precursors for the evaluation of sensitivity of cancer cells to anticancer drugs. Oncology 41:88–95
Sanfilippo O, Silvestrini R, Zaffaroni N, Piva L, Pizzocaro G (1986) Application of an in vitro antimetabolic assay to human germ cell testicular tumors for the preclinical evaluation of drug sensitivity. Cancer 58:1441–1447
Hanuaske AR (author) (2003) In vitro and in vivo predictive test. In: Kuffe DW, Pollock RE, Weichselbaum RR, Bast RC, Gansler TS, Holland JF, Frei IE (eds) Cancer medicine, 6th edn. BC Decker, Hamilton, Sect. 13:43
Hamburger AW, Salmon SE (1977) Primary bioassay of human tumor stem cells. Science 197:461–463
Von Hoff DD, Clark GM, Stogdill BJ, Sarosdy MF, O’Brien MT, Casper JT, Mattox DE, Page CP, Cruz AB, Sandbach JF (1983) Prospective clinical trial of a human tumor cloning system. Cancer Res 43:1926–1931
Federico M, Alberts DS, Garcia DJ, Emerson J, Fanta P, Liu R, Salmon SE (1994) In vitro drug testing of ovarian cancer using the human tumor colony-forming assay: comparison of in vitro response and clinical outcome. Gynecol Oncol 55:156–163
Kern DH, Weisenthal LM (1990) Highly specific prediction of antineoplastic drug resistance with an in vitro assay using suprapharmacologic drug exposures. J Natl Cancer Inst 82:582–588
Rubinstein LV, Shoemaker RH, Paull KD, Simon RM, Tosini S, Skehan P, Scudiero DA, Monks A, Boyd MR (1990) Comparison of in vitro anticancer-drug-screening data generated with a tetrazolium assay versus a protein assay against a diverse panel of human tumor cell lines. J Natl Cancer Inst 82:1113–1118
Kurbacher CM, Cree IA (2005) Chemosensitivity testing using microplate adenosine triphosphate-based luminescence measurements. Methods Mol Med 110:101–120
Roche Diagnostics Corporation (2005) Technical guide to cell proliferation and apoptosis methods (Roche), 3rd edition, pp 58–78
Martin A, Clynes M (1991) Acid phosphatase: endpoint for in vitro toxicity tests. In Vitro Cell Dev Biol 27(3 Pt 1):183–184
Abe K, Matsuki N (2000) Measurement of cellular 3-(4,5-dimethylthiazol–2-yl)-2,5-diphenyltetrazolium bromide (MTT) reduction activity and lactate dehydrogenase release using MTT. Neurosci Res 38:325–329
Black MM, Speer FD (1953) Effects of cancer chemotherapeutic agents on dehydrogenase activity of human cancer tissue in vitro. Am J Clin Pathol 23:218–227
Mosmann T (1983) Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J Immunol Methods 65:55–63
Veerman AJ, Pieters R (1990) Drug sensitivity assays in leukaemia and lymphoma. Br J Haematol 74:381–384
Pieters R, Huismans DR, Leyva A, Veerman AJ (1988) Adaptation of the rapid automated tetrazolium dye based (MTT) assay for chemosensitivity testing in childhood leukemia. Cancer Lett 41:323–332
Hwang WS, Chen LM, Huang SH, Wang CC, Tseng MT (1993) Prediction of chemotherapy response in human leukemia using in vitro chemosensitivity test. Leuk Res 17:685–688
Taylor CG, Sargent JM, Elgie AW, Williamson CJ, Lewandowicz GM, Chappatte O, Hill JG (2001) Chemosensitivity testing predicts survival in ovarian cancer. Eur J Gynaecol Oncol 22:278–282
Sargent JM (2003) The use of the MTT assay to study drug resistance in fresh tumour samples. Recent Results Cancer Res 161:13–25
Larsson R, Kristensen J, Sandberg C, Nygren P (1992) Laboratory determination of chemotherapeutic drug resistance in tumor cells from patients with leukemia, using a fluorometric microculture cytotoxicity assay (FMCA). Int J Cancer 50:177–185
Santini V, Bernabei PA, Silvestro L, Dal Pozzo O, Bezzini R, Viano I, Gattei V, Saccardi R, Ferrini PR (1989) In vitro chemosensitivity testing of leukemic cells: prediction of response to chemotherapy in patients with acute non-lymphocytic leukemia. Hematol Oncol 7:287–293
Shaw GL, Gazdar AF, Phelps R, Steinberg SM, Linnoila RI, Johnson BE, Oie HK, Russell EK, Ghosh BC, Pass HI, Minna JD, Mulshine JL, Ihde DC (1996) Correlation of in vitro drug sensitivity testing results with response to chemotherapy and survival: comparison of non-small cell lung cancer and small cell lung cancer. J Cell Biochem Suppl 24:173–185
Xu JM, Song ST, Tang ZM, Jiang ZF, Liu XQ, Zhou L, Zhang J, Liu XW (1999) Predictive chemotherapy of advanced breast cancer directed by MTT assay in vitro. Breast Cancer Res Treat 53:77–85
Sharma S, Neale MH, Di Nicolantonio F, Knight LA, Whitehouse PA, Mercer SJ, Higgins BR, Lamont A, Osborne R, Hindley AC, Kurbacher CM, Cree IA (2003) Outcome of ATP-based tumor chemosensitivity assay directed chemotherapy in heavily pre-treated recurrent ovarian carcinoma. BMC Cancer 3:19
Geisler JP, Linnemeier GC, Thomas AJ, Manahan KJ (2007) Extreme drug resistance is common after prior exposure to paclitaxel. Gynecol Oncol 106:538–540
Acknowledgments
Supported by grant aAV/1106/2004 and AV 4/0013/05 from Ministry of Education of Slovak Republic.
Conflict of interest statement
The authors declare that they have no conflict of interest related to the publication of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hatok, J., Babusikova, E., Matakova, T. et al. In vitro assays for the evaluation of drug resistance in tumor cells. Clin Exp Med 9, 1–7 (2009). https://doi.org/10.1007/s10238-008-0011-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10238-008-0011-3