Abstract
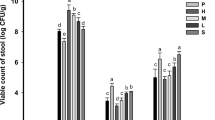
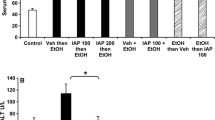
The objective is to study the possible mechanism by which lactic acid bacteria (LAB) prevent alcohol-induced steatohepatitis in rats. A total of 25 Wistar rats were divided into three groups: a LAB-fed group, an alcohol-treated group and a control group. Both the LAB-fed group and the alcohol-treated group received alcohol (10 g kg−1 per day) orally for up to 5 days (125 h). Before exposure to alcohol, the LAB-fed group were first treated daily with 1.5 ml/100 g of a mixture comprising 4 × 1010 ml−1 of Lactobacillus acidophilus and 2.5 × 107 ml−1 of Bifidobacterium longum, while the control group was treated with normal saline only. Biochemical data, alcohol dehydrogenase (ADH) activity and histology of the liver and stomach were evaluated. The ADH activity in the LAB mixture was 3.52 ± 0.45 μmol mg−1 protein (109 CFU ml−1), and was dose-dependent. By 30 min after taking alcohol, serum alcohol concentrations were 514.24 ± 80.21 μg ml−1 in the LAB-fed group and 795.15 ± 203.45 μg ml−1 in the alcohol-treated group (P < 0.005). Serum alcohol concentrations were reduced by 48% (P < 0.01) in the LAB-fed group, but by only 4% in the alcohol-treated group (P > 0.05) 120 min after oral intake of alcohol. The blood levels of endotoxin, AST and ALT were improved in the LAB-fed group compared to the alcohol-fed group (P < 0.01). All alcohol-treated rats showed moderate to severe steatohepatitis, but the LAB-fed rats showed almost normal histology or very slight lesions only. In conclusion, LAB decreased the alcohol concentration in the blood by increasing the first-pass metabolism in both the stomach and the liver, and effectively protected against alcohol-induced gastric and liver injury. It is interesting to note that the protection was more effective in the liver.


Similar content being viewed by others
References
Caballería J (2003) Current concepts in alcohol metabolism. Ann Hepatol 2:60–68
Rao RK, Seth A, Sheth P (2004) Recent advances in alcoholic liver disease I. Role of intestinal permeability and endotoxemia in alcoholic liver disease. Am J Physiol Gastrointest Liver Physiol 286:881–884
Nanji AA, Khettry U, Sadrzadeh SM (1994) Lactobacillus feeding reduces endotoxemia and severity of experimental alcoholic liver disease. Proc Soc Exp Biol Med 205:243–247
Thurman RG (1998) Mechanisms of hepatic toxicity II. Alcoholic liver injury involves activation of Kupffer cells by endotoxin. Am J Physiol Gastrointest Liver Physiol 275:605–611
Marotta F, Barreto R, Wu CC et al (2005) Experimental acute alcohol pancreatitis-related liver damage and endotoxemia: symbiotics but not metronidazole have a protective effect. Chin J Dig Dis 6:193–197
Nosova T, Jousimies-Somer H, Jokelainen K et al (2000) Acetaldehyde production and metabolism by human indigenous and probiotic Lactobacillus and Bifidobacterium strains. Alcohol Alcohol 35:561–568
Lowry OH, Rosebrough NJ, Farr AL (1951) Protein measurement with the folin phenol reagent. J Biol Chem 193:265–275
Nanji AA, Mendenhall CL, French SW (1989) Beef fat prevents alcoholic liver disease in the rat. Alcohol Clin Exp Res 13:15–19
Julkunen RJ, di Padova C, Lieber CS (1985) First pass metabolism of ethanol: a gastrointestinal barrier against the systemic toxicity of ethanol. Life Sci 37:567–573
Roine R, Gentry RT, Hernández-Muñoz R et al (1990) Aspirin increases blood alcohol concentrations in humans after ingestion of ethanol. JAMA 264:2406–2408
Caballería J, Baraona E, Rodamilans M, Lieber CS (1989) Effects of cimetidine on gastric alcohol dehydrogenase activity and blood ethanol levels. Gastroenterology 96:388–392
Hernández-Muñoz R, Caballería J, Uppal R et al (1990) Human gastric alcohol dehydrogenase: its inhibition by H2-receptor antagonists, and its effects on the bioavailability of ethanol. Alcohol Clin Exp Res 14:946–950
Salaspuro M (1996) Bacteriocolonic pathway for ethanol oxidation. Ann Med 28:195–200
Frezza M, di Padova C, Pozzato G et al (1990) High blood alcohol levels in women. The role of decreased gastric alcohol dehydrogenase activity and first-pass metabolism. N Engl J Med 322:95–99
Chiva M, Soriano G, Rochat I et al (2002) Effect of Lactobacillus johnsonii La1 and antioxidants on intestinal flora and bacteria translocation in rats with experimental cirrhosis. J Hepatol 37:456–462
Qing L, Duan ZP, Ha DK et al (2004) Symbiotic modulation of gut flora: effect on minimal hepatic encephalopathy in patients with cirrhosis. Hepatology 39:1441–1449
Riordan SM, Skinner NA, McIver CJ et al (2007) Symbiotic-associated improvement in liver function in cirrhotic patients: relation to changes in circulating cytokine messenger RNA and protein levels. Microb Ecol Health Dis 19:7–16
Bengmark S (2005) Symbiotics and the mucosal barrier in critically ill patients. Curr Opin Gastroenterol 21:712–716
Woodcock NP, McNaught CE, Morgan DR et al (2004) An investigation into the effect of a probiotic on gut immune function in surgical patients. Clin Nutr 23:1069–1073
Tok D, Ilkgul O, Bengmark S et al (2007) Pretreatment with pro- and symbiotics reduces peritonitis-induced acute lung injury in rats. J Trauma 62:880–885
Acknowledgments
We thank Professor Jin Xueyuan, Rehabilitation Research of China, for helpful comments on the experiment, and we also thank San Zhu Corporation for funding and for providing the probiotic.
Conflict of interest statement
The authors declare that they have no commercial or financial interest in the publication of this article.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Qing, L., Wang, T. Lactic acid bacteria prevent alcohol-induced steatohepatitis in rats by acting on the pathways of alcohol metabolism. Clin Exp Med 8, 187–191 (2008). https://doi.org/10.1007/s10238-008-0002-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10238-008-0002-4