Abstract
Objective
There is an absence of data on public preferences for health states (HSs) associated with severe chronic pain. The aim of this study was to develop accurate HS descriptions for severe chronic pain requiring intrathecal (IT) therapy and to derive utility weights that describe the health-related quality of life (HRQL) impact of chronic pain associated with malignant (MP) and non-malignant (NMP) aetiologies.
Research design and method
Eight visual analogue scale pain index (VAS-PI) HSs were defined using ranges 0–40, 41–60, 61–80 and 81–100 applied to both MP and NMP. Additionally, eight HSs representing common adverse events associated with IT therapy were identified. The content and description of the HSs were ascertained by interviews with five United Kingdom clinical experts. In total, 16 HSs were compiled. These HS descriptions and HS questionnaires were administered to 102 members of the public, utilising a time trade off (TTO) approach to estimate utilities for the HSs.
Results
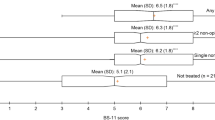
Participants generally were well matched to the general public in England and Wales, with some differences in mean age, race and education. A substantial decline in utility was observed with more severe VAS-PI values. The mean TTO utility values also decreased from mild pain to severe pain. Participants were able to differentiate between the side effects.
Conclusion
The study shows a clear decrement in utility moving from different severity levels of severe chronic pain.
Similar content being viewed by others
References
Gureje, O., Von Korff, M., Simon, G.E., Gater, R.: Persistent pain and well-being: a world health organization study in primary care. JAMA 280, 147–151 (1998)
Smith, B.H., Elliott, A.M., Chambers, W.A., Smith, W.C., Hannaford, P.C., Penny, K.: The impact of chronic pain in the community. Fam. Pract. 18, 292–299 (2001)
Elliott, A.M., Smith, B.H., Hannaford, P.C., Smith, W.C., Chambers, W.A.: The course of chronic pain in the community: results of a 4-year follow-up study. Pain 99, 299–307 (2002)
Turner, J.A., Sears, J.M., Loeser, J.D.: Programmable intrathecal opioid delivery systems for chronic noncancer pain: a systematic review of effectiveness and complications. Clin. J. Pain. 23, 180–195 (2007)
Williams, J.E., Louw, G.: Intrathecal pumps for giving opioids in chronic pain: a systematic review. Health Technol. Assess. 4, 1–65 (2000)
Dolan, P.: Modeling valuations for EuroQol health states. Med. Care 35, 1095–1108 (1997)
Brazier, J., Roberts, J., Deverill, M.: The estimation of a preference-based single index measure for health from the SF-36. J. Health Econ. 21, 271–292 (2002)
McDermott, A.M., Toelle, T.R., Rowbotham, D.J., Schaefer, C.P., Dukes, E.M.: The burden of neuropathic pain: results from a cross-sectional survey. Eur. J. Pain 10, 127–135 (2006)
Ackerman, L.L., Follet, K.A.: Long-term outcomes during treatment of chronic pain with intrathecal clonidine or clonidine/opioid combinations. J. Pain Symptom Manage. 26, 668–677 (2003)
Anderson, A., Burchiel, K.: A prospective study of long-term intrathecal morphine in the management of chronic nonmalignant pain. Neurosurgery 44, 289–300 (1999)
Nitescu, P., Dahm, P., Appelgren, L., Curelaru, I.: Continuous infusion of opioid and bupivacaine by externalized intrathecal catheters in long-term treatment of “refractory” nonmalignant pain. J. Clin. Pharm. 14, 17–28 (1998)
Sjöberg, M., Nitescu, P., Appelgren, L., Curelaru, I.: Long-term intrathecal morphine and bupivacaine in patients with refractory cancer pain. Results from a morphine:bupivacaine dose regimen of 0.5:4.75 mg/ml. Anesthesiology 80, 284–297 (1994)
Winkelmüller, M., Winkelmüller, W.: Long-term effects of continuous intrathecal opioid treatment in chronic pain of nonmalignant etiology. J. Neurosurg. 85, 458–467 (1996)
Carlsson, A.M.: Assessment of chronic pain. I. Aspects of the reliability and validity of the visual analogue scale. Pain 16, 87–101 (1997)
Price, D.D., McGrath, P.A., Rafii, A., Buckingham, B.: The validation of visual analogue scales as ratio scale measures for chronic and experimental pain. Pain 17, 45–56 (1983)
Rauck, R.L., Wallace, M.S., Leong, M.S., Minehart, M., Webster, L.R., Charapata, S.G., Abraham, J.E., Buffington, D.E., Ellis, D., Kartzinel, R.: Ziconotide 301 Study Group: a randomized, double-blind, placebo-controlled study of intrathecal ziconotide in adults with severe chronic pain. J. Pain Symptom Manage. 31, 393–406 (2006)
Staats, P.S., Yearwood, T., Charapata, S.G., Presley, R.W., Wallace, M.S., Byas-Smith, M., Fisher, R., Bryce, D.A., Mangieri, E.A., Luther, R.R., Mayo, M., McGuire, D., Ellis, D.: Intrathecal ziconotide in the treatment of refractory pain in patients with cancer or AIDS: a randomized controlled trial. JAMA 291, 63–70 (2004)
Wermeling, D., Drass, M., Ellis, D., Mayo, M., McGuire, D., O’Connell, D., Hale, V., Chao, S.: Pharmacokinetics and pharmacodynamics of intrathecal ziconotide in chronic pain patients. J. Clin. Pharmacol. 43, 624–636 (2003)
Atanassoff, P.G., Hartmannsgruber, M.W., Thrasher, J., Wermeling, D., Longton, W., Gaeta, R., Singh, T., Mayo, M., McGuire, D., Luther, R.R.: Ziconotide, a new N-type calcium channel blocker, administered intrathecally for acute postoperative pain. Reg. Anesth. Pain. Med. 25, 274–278 (2000)
Brose, W.G., Gutlove, D.P., Luther, R.R., Bowersox, S.S., McGuire, D.: Use of intrathecal SNX-111, a novel, N-type, voltage-sensitive, calcium channel blocker, in the management of intractable brachial plexus avulsion pain. Clin. J. Pain 13, 256–259 (1997)
Breivik, H., Collett, B., Ventafridda, V., Cohen, R., Gallacher, D.: Survey of chronic pain in Europe: prevalence, impact on daily life, and treatment. Eur. J. Pain 10, 287–333 (2006)
Kamran, S., Wright, B.: Complications of intrathecal drug delivery systems. Neuromodulation 4, 111–115 (2001)
Mercadante, S.: Controversies over spinal treatment in advanced cancer patients. Support. Care Cancer 6, 495–502 (1998)
Naumann, C., Erdine, S., Koulousakis, A., Van Buyten, J.P., Schuchard, M.: Drug adverse events and system complications of intrathecal opioid delivery for pain: origins, detection, manifestations, and management. Neuromodulation 2, 92–107 (2002)
Tutak, U., Doleys, D.M.: Intrathecal infusion systems for treatment of chronic low back and leg pain of noncancer origin. South Med. J. 89, 295–300 (1996)
Yang, C.Y., Wong, C.S., Chang, J.Y., Ho, S.T.: Intrathecal ketamine reduces morphine requirements in patients with terminal cancer pain. Can. J. Anaesth. 43, 379–383 (1996)
Wallace, M.S., Rauck, R., Fisher, R., Charapata, S.G., Ellis, D., Dissanayake, S., Ziconotide 98-022 Study Group: Intrathecal ziconotide for severe chronic pain: safety and tolerability results of an open-label, long-term trial. Anesth. Analg. 106, 628–637 (2008)
Wermeling, D.P., Berger, J.R.: Ziconotide infusion for severe chronic pain: case series of patients with neuropathic pain. Pharmacotherapy 26, 395–402 (2006)
Kind, P.: The EuroQoL instrument: an index of health-related quality of life. In: Spilker, B. (ed.) Quality of Life and Pharmacoeconomics in Clinical Trials, pp. 191–203. Lippincott-Raven, Philadelphia (1996)
The EuroQol Group: EuroQol–a new facility for the measurement of health-related quality of life. Health Policy 16, 199–208 (1990)
Bennett, K., Torrance, G.: Measuring health state preferences and utilities: Rating scale, time trade-off and standard gamble techniques. In: Spilker, B. (ed.) HRQL and Pharmacoeconomics in Clinical Trials, pp. 253–266. Lippincott-Rave, Philadelphia (1996)
National Institute for Clinical Excellence, Guide to the Methods of Technology Appraisal. http://www.nice.org.uk/niceMedia/pdf/TAP_Methods.pdf (2004)
National Institute for Clinical Excellence, Guide to the Methods of Technology Appraisal. http://www.nice.org.uk/media/B52/A7/TAMethodsGuideUpdatedJune2008.pdf (2008)
Lai, K.C., Provenzale, J.M., Delong, D., Mukundan, S, Jr: Assessing patient utilities for varying degrees of low back pain. Acad. Radiol. 12, 467–474 (2005)
Roland, M., Morris, R.: A study of the natural history of low-back pain. Part II: development of guidelines for trials of treatment in primary care. Spine 8, 145–150 (1983)
Roland, M., Morris, R.: A study of the natural history of back pain. Part I: development of a reliable and sensitive measure of disability in low-back pain. Spine 8, 141–144 (1983)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Eldabe, S., Lloyd, A., Verdian, L. et al. Eliciting health state utilities from the general public for severe chronic pain. Eur J Health Econ 11, 323–330 (2010). https://doi.org/10.1007/s10198-009-0178-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10198-009-0178-x